Advanced Synthetic Route for Complex 3'-Spiro Indolizidine Derivatives via Double Dearomatization
Introduction to Breakthrough Double Dearomatization Technology
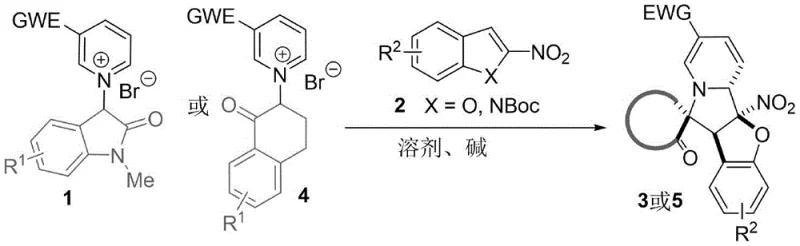
The field of organic synthesis is constantly evolving to meet the demands of complex drug molecule construction, particularly for alkaloid scaffolds found in numerous bioactive natural products. A significant advancement in this domain is detailed in patent CN115353521B, which discloses a highly efficient method for synthesizing complex 3′-spiro indolizidine derivatives. This technology addresses a long-standing challenge in synthetic chemistry: the difficulty of executing double dearomatization reactions on two distinct aromatic fragments simultaneously. Traditionally, constructing such intricate polycyclic systems required harsh conditions or multi-step sequences that compromised overall efficiency. The disclosed method leverages the reactivity of active methylene pyridinium salts, acting as precursors to pyridine ylides, which undergo a cascade reaction with electron-deficient nitro-heterocycles.
This innovative approach not only streamlines the synthetic pathway but also enhances the structural diversity accessible to medicinal chemists. By utilizing 3-position electron-deficient pyridine onium salts reacting with 2-nitroindoles or 2-nitrobenzofurans, the process facilitates an intramolecular ring-closing step that drives the formation of the spirocyclic core. The significance of this development lies in its ability to bypass the high energy barriers typically associated with bimolecular dearomatization, offering a robust platform for generating high-value pharmaceutical intermediates. As a reliable pharmaceutical intermediate supplier, understanding such pivotal technological shifts is crucial for maintaining a competitive edge in API manufacturing.
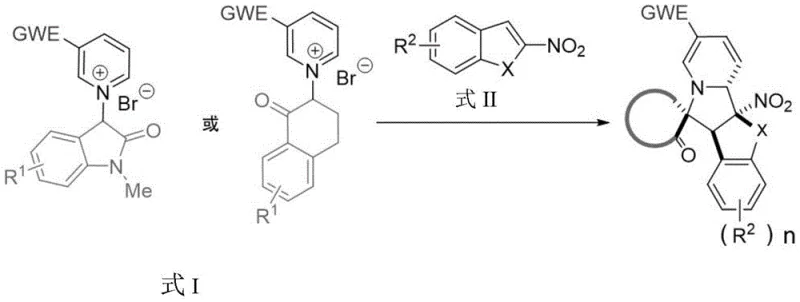
The core of this invention rests on a novel 3+2 cycloaddition strategy that effectively merges two aromatic systems into a rigid, three-dimensional spiro-framework. This transformation is particularly valuable because spiro-indolizidine motifs are prevalent in a wide array of alkaloids with potent biological activities, including anticancer and antiviral properties. The patent highlights that while single-molecule dearomatization is well-documented, the intermolecular variant involving two different aromatic partners has been rarely studied due to kinetic and thermodynamic hurdles. The successful implementation of this reaction under mild conditions represents a substantial leap forward, providing a versatile tool for the rapid assembly of molecular complexity from simple, commercially available starting materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the construction of similar spirocyclic architectures often relied on photocatalytic strategies or transition-metal catalysis, both of which present significant drawbacks for large-scale industrial application. For instance, previous literature, such as work by S.-L. You et al., demonstrated intramolecular double dearomatization under photocatalytic conditions. However, these methods necessitate the use of expensive precious metal photosensitizers, which not only inflate the raw material costs but also introduce critical purification challenges. Removing trace amounts of heavy metals to meet the stringent purity specifications required for high-purity pharmaceutical intermediates adds additional processing steps, such as scavenging or recrystallization, which inevitably reduce overall yield and increase production time.
Furthermore, conventional approaches often struggle with substrate scope and functional group tolerance. Many traditional cyclization reactions require elevated temperatures or strong acidic/basic conditions that can degrade sensitive functional groups commonly found in drug candidates. The energy barrier for effecting dearomatization on two separate molecules is significantly higher than for a single molecule, leading to sluggish reaction kinetics and poor conversion rates in non-optimized systems. This limitation restricts the utility of older methods, making them less viable for the commercial scale-up of complex polymer additives or drug substances where consistency and cost-efficiency are paramount. The reliance on specialized equipment, such as high-power LED arrays for photoredox chemistry, further complicates the engineering requirements for manufacturing plants.
The Novel Approach
In stark contrast, the method described in CN115353521B utilizes a base-mediated organocatalytic pathway that eliminates the need for precious metals entirely. By employing pyridinium salts with electron-withdrawing groups at the 3-position, the electrophilicity of the pyridine ring is enhanced, promoting the formation of the reactive pyridine ylide species in situ. This activation strategy allows the reaction to proceed smoothly at room temperature (20-40°C) using common organic bases like triethylamine. The elimination of expensive catalysts and specialized light sources translates directly into cost reduction in pharmaceutical intermediate manufacturing, as the process becomes more atom-economical and operationally simpler.
Moreover, this novel approach exhibits exceptional stereoselectivity, consistently producing the desired spiro-isomer with high diastereomeric ratios (often >19:1). The mild reaction conditions preserve sensitive functional groups, allowing for a broader substrate scope that includes various halogens, alkoxy, and alkyl substituents on both the indole/benzofuran and pyridine rings. This flexibility is essential for medicinal chemistry campaigns where rapid analog synthesis is required to optimize biological activity. The ability to achieve yields as high as 95% with minimal byproduct formation underscores the efficiency of this route, positioning it as a superior alternative for the synthesis of complex heterocyclic scaffolds needed in modern drug discovery and development pipelines.
Mechanistic Insights into Base-Mediated Double Dearomatization
The mechanistic pathway of this transformation involves a sophisticated sequence of deprotonation, nucleophilic attack, and cyclization events that collectively drive the double dearomatization process. Initially, the base abstracts an acidic proton from the active methylene position of the pyridinium salt, generating a reactive pyridine ylide intermediate. This ylide acts as a 1,3-dipole or a nucleophile depending on the specific electronic environment, attacking the electron-deficient double bond of the 2-nitrobenzofuran or 2-nitroindole. The presence of the nitro group is critical, as it significantly lowers the LUMO energy of the heterocycle, facilitating the initial addition step. Following this intermolecular coupling, the resulting intermediate undergoes an intramolecular nucleophilic attack by the nitrogen or oxygen atom of the heterocycle onto the activated pyridine ring.
This second step constitutes the actual dearomatization of the pyridine moiety, locking the structure into the rigid spiro-indolizidine framework. The stereochemical outcome is controlled by the steric interactions during the transition state of the ring-closing step, favoring the formation of the observed major diastereomer. The electron-withdrawing group (GWE) on the pyridine ring, such as a methoxycarbonyl (-CO2Me) or cyano (-CN) group, plays a dual role: it stabilizes the ylide intermediate and increases the electrophilicity of the pyridine ring to facilitate the final cyclization. Understanding these electronic nuances is vital for process chemists aiming to optimize reaction parameters for reducing lead time for high-purity pharmaceutical intermediates. The robustness of this mechanism ensures that even with varying substituents (R1, R2), the reaction maintains high fidelity and selectivity.
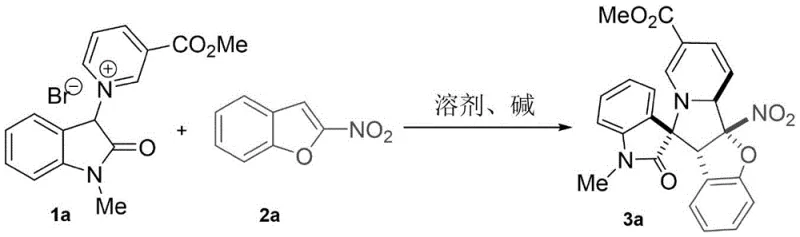
Impurity control is inherently built into this mechanism due to the high specificity of the ylide-nitroalkene interaction. Unlike radical-based photocatalytic processes that can generate diverse side products through uncontrolled radical propagation, this ionic pathway follows a defined trajectory. The primary impurities typically arise from incomplete conversion or hydrolysis of the ester groups, both of which are easily managed through standard workup procedures like silica gel chromatography. The patent data indicates that the reaction tolerates a wide range of solvents, with dichloromethane (DCM) proving optimal for balancing solubility and reaction rate. This mechanistic clarity allows for precise tuning of stoichiometry; for instance, using a slight excess of the pyridinium salt (1.2 equivalents) ensures complete consumption of the potentially more valuable nitro-heterocycle partner, maximizing material efficiency in a production setting.
How to Synthesize 3'-Spiro Indolizidine Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reagent quality and reaction monitoring to ensure optimal outcomes. The process begins with the preparation of the key pyridinium salt precursor, which is synthesized by quaternizing a 3-substituted pyridine with a brominated oxindole or naphthalenone derivative. Once the salt is secured, the cyclization reaction is straightforward: the salt and the nitro-heterocycle are dissolved in anhydrous dichloromethane, and a stoichiometric amount of base is added. The mixture is stirred at ambient temperature, avoiding the need for heating or cooling baths, which simplifies the operational setup. Reaction progress can be monitored via TLC or HPLC, with completion typically achieved within 10 to 30 hours depending on the specific substrate electronics.
- Prepare the pyridinium salt precursor by reacting a 3-position electron-deficient pyridine with 3-bromooxindole or 2-bromonaphthalenone.
- Dissolve the pyridinium salt and 2-nitrobenzofuran (or 2-nitroindole) in dichloromethane (DCM) at an equivalent ratio of approximately 1.2: 1.
- Add an organic base such as triethylamine (TEA) at 1 to 2.5 equivalents and stir at room temperature (20-40°C) for 10-30 hours to complete the cyclization.
Following the reaction, the crude product is purified using standard silica gel column chromatography, typically eluting with a petroleum ether and ethyl acetate mixture. The high stereoselectivity of the reaction means that the crude material is often quite clean, minimizing the loss of product during purification. For larger scale operations, crystallization might be explored as a more cost-effective purification method compared to chromatography. The versatility of this method allows for the introduction of diverse functional groups, enabling the synthesis of a library of derivatives for biological screening. Whether targeting a specific API intermediate or exploring new chemical space, this protocol offers a reliable and scalable entry point into the world of spiro-indolizidine chemistry.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain and procurement perspective, this synthetic methodology offers compelling advantages that align with the goals of cost efficiency and operational reliability. The elimination of precious metal catalysts is perhaps the most significant economic benefit. Traditional photocatalytic routes rely on iridium or ruthenium complexes, which are not only expensive to purchase but also subject to volatile market pricing and supply constraints. By switching to a base-mediated organocatalytic system, manufacturers can decouple their production costs from the fluctuations of the precious metals market. Furthermore, the absence of heavy metals simplifies the regulatory compliance landscape, as there is no need for rigorous testing and validation of metal residue levels in the final product, thereby accelerating the release of batches for clinical or commercial use.
- Cost Reduction in Manufacturing: The shift to room temperature conditions drastically reduces energy consumption compared to processes requiring reflux or cryogenic cooling. Additionally, the use of commodity chemicals like triethylamine and dichloromethane ensures that raw material costs remain low and predictable. The high yields reported (up to 95%) mean that less starting material is wasted, improving the overall mass balance and reducing the cost of goods sold (COGS). This efficiency is critical for maintaining margins in the competitive generic pharmaceutical market.
- Enhanced Supply Chain Reliability: The starting materials for this reaction, such as substituted pyridines and nitrobenzofurans, are widely available from multiple global suppliers, reducing the risk of single-source dependency. The robustness of the reaction conditions means that production is less susceptible to disruptions caused by equipment failure or utility fluctuations. This reliability ensures a steady flow of intermediates, supporting just-in-time manufacturing models and preventing stockouts that could delay downstream API synthesis.
- Scalability and Environmental Compliance: The simplicity of the workup and the use of common solvents make this process highly amenable to scale-up from gram to ton quantities without significant re-engineering. The reduced waste generation, owing to high atom economy and selectivity, aligns with green chemistry principles and helps facilities meet increasingly strict environmental regulations. This sustainability aspect is becoming a key differentiator for suppliers seeking to partner with environmentally conscious multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis method, derived from the detailed experimental data provided in the patent documentation. These insights are intended to clarify the practical implications of adopting this technology for your specific manufacturing needs. Understanding the nuances of substrate compatibility and reaction optimization can help R&D teams integrate this route into their existing workflows more effectively.
Q: What are the key advantages of this double dearomatization method over photocatalytic routes?
A: Unlike previous photocatalytic methods that require expensive precious metal photosensitizers, this novel approach utilizes simple organic bases under mild room temperature conditions, significantly reducing raw material costs and simplifying the removal of metal impurities.
Q: What is the stereoselectivity and yield profile of this synthesis?
A: The reaction demonstrates excellent stereocontrol, achieving diastereoselectivity greater than 19:1 in most cases, with isolated yields frequently exceeding 80% and reaching up to 95% for optimized substrates.
Q: Can this method be scaled for commercial production of API intermediates?
A: Yes, the process uses common solvents like dichloromethane and operates at ambient temperatures without specialized light equipment, making it highly suitable for scale-up from laboratory to multi-ton commercial manufacturing.
For further technical details regarding specific substrate limitations or custom optimization studies, our technical team is available to provide deeper analysis. We recognize that every project has unique constraints, and having access to granular data regarding impurity profiles and kinetic parameters can be invaluable for process validation. We encourage potential partners to review the full experimental section of the patent for comprehensive characterization data, including NMR and HRMS results that confirm the structural integrity of the synthesized spiro-compounds.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3'-Spiro Indolizidine Supplier
At NINGBO INNO PHARMCHEM, we are committed to translating cutting-edge academic research into tangible commercial solutions for the global pharmaceutical industry. Our expertise in complex organic synthesis allows us to rapidly adapt novel methodologies, such as the double dearomatization route described in CN115353521B, for large-scale production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to manufacturing plant is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications required by top-tier regulatory agencies worldwide.
We invite you to contact our technical procurement team to discuss how we can support your supply chain with high-quality spiro-indolizidine intermediates. Whether you require a Customized Cost-Saving Analysis for your current route or need specific COA data and route feasibility assessments for new projects, we are ready to provide the data-driven insights you need. Let us collaborate to optimize your synthesis strategy, reduce costs, and accelerate your time to market with reliable, high-performance chemical solutions.
