Advanced C-H Activation Strategy for Scalable Spiro Quinazolinone Pharmaceutical Intermediates Production
Advanced C-H Activation Strategy for Scalable Spiro Quinazolinone Pharmaceutical Intermediates Production
The landscape of heterocyclic chemistry is undergoing a significant transformation driven by the demand for more efficient and sustainable synthetic methodologies, particularly for privileged scaffolds like quinazolinones. Patent CN111087401B discloses a groundbreaking approach for establishing spiro rings on 2-aryl quinazoline-4(3H)-one compounds, utilizing a novel continuous C-H activation strategy mediated by cyclopropenone. This technology addresses critical bottlenecks in the synthesis of complex nitrogen-containing heterocycles, which are ubiquitous in pharmaceuticals, agrochemicals, and functional materials. By leveraging the unique reactivity of strained cyclopropenone rings, this method enables the direct formation of carbon-carbon bonds on aromatic systems without the need for pre-functionalized halides or boronic acids. For R&D directors and process chemists, this represents a paradigm shift towards step-economical synthesis, offering a robust pathway to access diverse spirocyclic libraries that were previously difficult or prohibitively expensive to construct using classical cross-coupling techniques.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the functionalization of the quinazolinone core to generate complex spirocyclic architectures has been fraught with synthetic challenges that hinder rapid drug discovery and process development. Traditional routes typically involve multi-step sequences requiring the installation of leaving groups, followed by metal-catalyzed cross-couplings or cycloadditions that often suffer from poor regioselectivity and low atom economy. These conventional protocols frequently necessitate the use of expensive palladium or platinum catalysts, stringent anhydrous conditions, and stoichiometric amounts of toxic oxidants, which complicate downstream purification and waste management. Furthermore, the construction of spiro-centers often requires specialized precursors that are not commercially available, forcing medicinal chemistry teams to dedicate valuable resources to building block synthesis rather than biological evaluation. The cumulative effect of these inefficiencies is a prolonged development timeline and inflated cost of goods, creating a significant barrier for the commercial scale-up of complex quinazolinone derivatives in the competitive pharmaceutical intermediate market.
The Novel Approach
In stark contrast to these legacy methods, the technology described in CN111087401B introduces a streamlined, one-pot cascade reaction that harnesses the power of transition metal-catalyzed C-H activation coupled with the release of ring strain from cyclopropenones. This innovative strategy allows for the direct coupling of readily available 2-aryl quinazolinones with diaryl cyclopropenones, effectively bypassing the need for pre-activated substrates. The reaction proceeds through a sophisticated mechanism where the transition metal catalyst activates the inert C-H bond on the quinazolinone aromatic ring, facilitating a sequential cyclization that constructs two new C-C bonds and one C-N bond simultaneously. 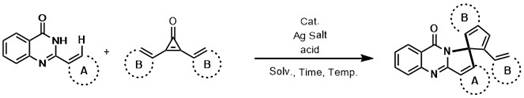 This convergence of reactivity not only simplifies the operational complexity but also significantly enhances the overall yield and purity of the final spirocyclic products. By utilizing earth-abundant metals like ruthenium, rhodium, or iridium in conjunction with silver salts and organic acids, this method provides a versatile platform that accommodates a wide range of substituents, thereby expanding the chemical space accessible to process chemists seeking reliable pharmaceutical intermediate supplier solutions.
This convergence of reactivity not only simplifies the operational complexity but also significantly enhances the overall yield and purity of the final spirocyclic products. By utilizing earth-abundant metals like ruthenium, rhodium, or iridium in conjunction with silver salts and organic acids, this method provides a versatile platform that accommodates a wide range of substituents, thereby expanding the chemical space accessible to process chemists seeking reliable pharmaceutical intermediate supplier solutions.
Mechanistic Insights into Transition Metal-Catalyzed C-H Activation and Cyclization
The mechanistic underpinning of this transformation relies on the synergistic interplay between the transition metal catalyst, the silver salt additive, and the inherent strain energy of the cyclopropenone substrate. The reaction initiates with the coordination of the quinazolinone nitrogen to the metal center, directing the activation of the ortho-C-H bond on the pendant aryl ring through a concerted metalation-deprotonation (CMD) or electrophilic substitution pathway. The presence of the silver salt is crucial, as it acts as a halide scavenger to generate the cationic active catalytic species and may also assist in the abstraction of protons during the C-H cleavage step. Once the metallacycle is formed, the highly reactive cyclopropenone inserts into the metal-carbon bond, driven by the relief of its substantial angle strain, which serves as the thermodynamic driving force for the entire cascade. This insertion is followed by a series of migratory insertions and reductive elimination steps that forge the new carbon framework, ultimately releasing the spirocyclic product and regenerating the catalyst for the next turnover. Understanding this cycle is vital for R&D teams aiming to optimize reaction parameters such as temperature and solvent polarity to maximize conversion and minimize side reactions.
From an impurity control perspective, the high regioselectivity of this C-H activation process is a distinct advantage over traditional electrophilic aromatic substitution methods, which often yield mixtures of ortho-, meta-, and para-substituted byproducts. The directing group ability of the quinazolinone nitrogen ensures that functionalization occurs exclusively at the desired position, simplifying the impurity profile and reducing the burden on purification processes. Additionally, the use of mild organic acids like adamantane carboxylic acid helps to buffer the reaction medium, preventing the decomposition of sensitive functional groups that might be present on the substrate. This level of control is essential for producing high-purity OLED material or pharmaceutical intermediates where trace impurities can have detrimental effects on downstream performance or regulatory approval. The robustness of the catalytic system against various electronic and steric environments further underscores its utility in generating diverse compound libraries with consistent quality.
How to Synthesize Spiro Quinazolinone Derivatives Efficiently
The practical implementation of this synthesis protocol is designed to be straightforward and adaptable to standard laboratory equipment, making it accessible for both academic research and industrial process development. The general procedure involves charging a pressure-resistant vessel with the 2-aryl quinazolinone substrate, a ruthenium-based catalyst such as [Ru(p-cymene)Cl2]2, a silver salt like AgSbF6, and an organic acid additive in a chlorinated solvent. The specific stoichiometry and reaction conditions, including temperature and time, can be tuned based on the electronic nature of the substrates to achieve optimal yields, as demonstrated in the patent examples where yields ranged from 71% to 84%. 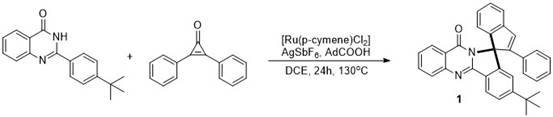 Following the reaction period, typically around 24 hours at elevated temperatures, the workup is remarkably simple, involving solvent removal and purification via silica gel column chromatography. For those interested in the precise operational details and stoichiometric ratios required to replicate these results, the detailed standardized synthesis steps are outlined in the guide below.
Following the reaction period, typically around 24 hours at elevated temperatures, the workup is remarkably simple, involving solvent removal and purification via silica gel column chromatography. For those interested in the precise operational details and stoichiometric ratios required to replicate these results, the detailed standardized synthesis steps are outlined in the guide below.
- Charge a pressure-resistant vessel with 2-aryl quinazolinone, ruthenium catalyst ([Ru(p-cymene)Cl2]2), silver salt (AgSbF6), and organic acid (AdCOOH) in DCE solvent.
- Add diphenylcyclopropenone to the mixture, seal the vessel, and stir in an oil bath at 130°C for 24 hours to facilitate C-H activation and cyclization.
- Upon completion, remove the solvent under reduced pressure and purify the crude residue via silica gel column chromatography to isolate the high-purity spiro product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic methodology offers tangible benefits that extend beyond mere chemical elegance, directly impacting the bottom line and operational resilience. The shift towards a one-pot cascade reaction eliminates several unit operations associated with traditional multi-step syntheses, such as intermediate isolation, drying, and additional reactor charges, which translates to significant reductions in labor, energy consumption, and solvent usage. Furthermore, the reliance on commercially available and relatively inexpensive starting materials, such as simple quinazolinones and cyclopropenones, mitigates the risk of supply chain disruptions often associated with exotic or custom-synthesized building blocks. This accessibility ensures a more stable and predictable supply of critical intermediates, allowing manufacturing teams to plan production schedules with greater confidence and reduce lead times for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The economic viability of this process is heavily bolstered by the use of cost-effective transition metal catalysts, particularly ruthenium complexes, which are generally more affordable than the palladium or rhodium catalysts often employed in similar transformations. By eliminating the need for pre-functionalized substrates like aryl halides or boronic acids, the raw material costs are substantially lowered, as these precursors often require additional synthetic steps to prepare. Moreover, the high atom economy of the reaction, where the majority of the reactant atoms are incorporated into the final product, minimizes waste disposal costs and maximizes the value derived from each kilogram of input material. These factors combine to create a manufacturing process that is not only chemically efficient but also financially optimized for cost reduction in API manufacturing.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions and the broad substrate scope contribute to a more resilient supply chain capable of adapting to varying demands. Since the method tolerates a wide array of functional groups and substituents, it allows for the flexible production of different analogues using a common set of reagents and equipment, reducing the need for specialized infrastructure. This flexibility is crucial for maintaining continuity of supply in the face of fluctuating market demands or raw material availability issues. Additionally, the simplified purification process reduces the dependency on complex chromatographic resins or specialized filtration media, further streamlining the logistics of production and ensuring that delivery timelines are met consistently for clients seeking a reliable agrochemical intermediate supplier.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, this technology aligns well with modern green chemistry principles by reducing the number of synthetic steps and the associated solvent waste. The ability to perform the reaction in standard pressure vessels at moderate temperatures facilitates easy scale-up from gram to kilogram quantities without the need for specialized high-pressure equipment. The use of less hazardous solvents and the generation of fewer byproducts simplify the waste treatment process, helping manufacturers meet increasingly stringent environmental regulations. This ease of commercial scale-up of complex polymer additives or pharmaceutical intermediates ensures that the technology can be rapidly deployed to meet industrial volume requirements while maintaining a low environmental footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this spiro-ring establishment method, providing clarity for stakeholders evaluating its potential integration into their existing workflows. These insights are derived directly from the experimental data and technical disclosures within the patent, ensuring that the information provided is accurate and relevant to real-world application scenarios. Understanding these nuances is key to making informed decisions about process adoption and resource allocation.
Q: What are the primary limitations of traditional quinazolinone functionalization methods?
A: Traditional methods often rely on pre-functionalized substrates requiring multiple synthetic steps, harsh reaction conditions, and stoichiometric oxidants, leading to poor atom economy and significant waste generation compared to direct C-H activation strategies.
Q: How does the novel cyclopropenone coupling method improve process efficiency?
A: This method utilizes the high ring strain of cyclopropenone to drive the reaction, enabling the simultaneous construction of two C-C bonds and one C-N bond in a single pot, thereby drastically simplifying the synthetic route and improving overall yield.
Q: Is this catalytic system suitable for large-scale industrial manufacturing?
A: Yes, the protocol employs commercially available and relatively inexpensive ruthenium catalysts alongside simple workup procedures like silica gel chromatography, making it highly amenable to scale-up for commercial production of complex heterocycles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Spiro Quinazolinone Supplier
The technological advancements detailed in CN111087401B represent a significant opportunity for the pharmaceutical and fine chemical industries to access complex heterocyclic scaffolds with unprecedented efficiency. At NINGBO INNO PHARMCHEM, we recognize the potential of such innovative synthetic routes and have positioned ourselves as a leader in translating cutting-edge academic and patent literature into viable commercial processes. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory methods are successfully adapted for industrial manufacturing. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of spiro quinazolinone intermediate performs reliably in your downstream applications.
We invite you to collaborate with us to leverage this advanced C-H activation technology for your specific project needs. Whether you require custom synthesis of novel derivatives or scale-up of existing protocols, our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can drive value and efficiency in your supply chain.
