Advanced Oxidative Sulfonylation for Scalable Cycloenamine Sulfone Production
The pharmaceutical and fine chemical industries are constantly seeking efficient, cost-effective routes to access complex heterocyclic scaffolds that serve as critical building blocks for bioactive molecules. Patent CN112341376B discloses a groundbreaking preparation method for cycloenamine sulfone derivatives, a class of compounds renowned for their valuable structural utility in drug discovery and agrochemical development. This innovative protocol leverages a metal-free oxidative sulfonylation strategy, utilizing elemental iodine and tert-butyl hydroperoxide (TBHP) as the oxidant system. By employing readily available piperidine compounds and sulfonyl hydrazides as starting materials, this technology addresses the longstanding challenges of high cost and operational complexity associated with traditional alkenyl sulfone synthesis. The resulting cycloenamine sulfone derivatives, characterized by the general structure shown below, represent a versatile platform for generating diverse libraries of biologically active candidates.

Furthermore, the method described in this patent offers a significant strategic advantage for supply chain managers and procurement specialists by relying on commodity chemicals rather than specialized, high-cost catalysts. The ability to synthesize these high-value intermediates in a single step under relatively mild conditions translates directly into improved process economics and reduced environmental footprint. As the demand for functionalized sulfone motifs continues to grow in the development of next-generation therapeutics, adopting this robust synthetic methodology positions manufacturers to meet market needs with greater agility and reliability. The following analysis details the technical merits and commercial implications of this novel oxidative transformation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alkenyl sulfone compounds has relied on classical condensation reactions such as the Knoevenagel condensation or the Horner-Emmons reaction, which often necessitate stringent reaction conditions and generate stoichiometric amounts of waste. More recent methodologies involving the direct reaction of alkenes or alkynes with sodium sulfinates have gained traction, yet they frequently suffer from limited substrate scope or the requirement for expensive transition metal catalysts that complicate downstream processing. The presence of heavy metal residues in the final product is a critical concern for pharmaceutical applications, often mandating additional, costly purification steps to meet regulatory standards for elemental impurities. Moreover, many existing protocols require inert atmospheres or specialized equipment to handle sensitive reagents, which increases the capital expenditure and operational complexity for commercial-scale manufacturing facilities.
The Novel Approach
In stark contrast to these conventional limitations, the method disclosed in patent CN112341376B introduces a streamlined, one-pot oxidative sulfonylation that bypasses the need for transition metals entirely. As illustrated in the general reaction scheme below, the process couples a piperidine derivative with a sulfonyl hydrazide using a simple iodine/TBHP oxidant system in the presence of a mild base. This approach not only simplifies the synthetic route by merging oxidation and sulfonylation into a single operational step but also utilizes inexpensive and stable reagents that are easily sourced from global chemical suppliers. The reaction proceeds efficiently under air atmosphere, eliminating the need for rigorous exclusion of oxygen or moisture, which significantly lowers the barrier for implementation in standard chemical production plants.
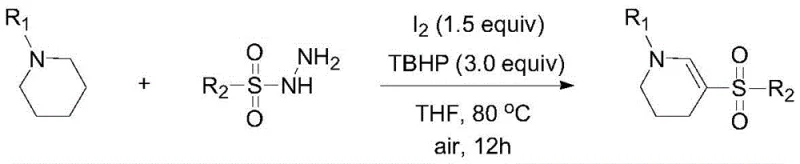
The versatility of this novel approach is further evidenced by its tolerance for a wide range of substituents on both the piperidine and sulfonyl hydrazide components, allowing for the rapid generation of diverse chemical space. The use of elemental iodine as a promoter is particularly advantageous from a cost perspective, as it is significantly cheaper than palladium, copper, or rhodium catalysts typically employed in C-H functionalization reactions. Additionally, the mild reaction conditions, typically ranging from 50°C to 100°C, ensure energy efficiency and minimize the risk of thermal runaway or decomposition of sensitive functional groups. This combination of operational simplicity, cost efficiency, and broad applicability makes the new method a superior choice for the industrial production of cycloenamine sulfone derivatives.
Mechanistic Insights into Iodine-Mediated Oxidative Sulfonylation
The mechanistic pathway of this transformation involves a radical-mediated process initiated by the interaction between elemental iodine and the sulfonyl hydrazide substrate. Under the oxidative conditions provided by TBHP, the sulfonyl hydrazide undergoes homolytic cleavage to generate a reactive sulfonyl radical species, which serves as the key sulfur-containing intermediate. Simultaneously, the piperidine ring undergoes oxidative dehydrogenation, likely facilitated by the iodine/TBHP system, to form an enamine or iminium intermediate in situ. The convergence of these two reactive species leads to the formation of the carbon-sulfur bond, ultimately yielding the desired cycloenamine sulfone product after elimination or rearomatization steps. This radical mechanism avoids the high-energy barriers associated with ionic pathways, allowing the reaction to proceed smoothly at moderate temperatures without the need for strong acids or bases that could degrade the product.
Impurity control is a critical aspect of this mechanism, particularly given the potential for over-oxidation or polymerization of the reactive intermediates. The selection of sodium bicarbonate as the preferred base plays a pivotal role in maintaining the optimal pH environment to facilitate radical generation while suppressing side reactions. Stronger bases might lead to premature decomposition of the sulfonyl hydrazide or unwanted elimination reactions, whereas weaker bases may fail to neutralize acidic byproducts effectively. Furthermore, the use of 1,4-dioxane as the solvent of choice provides a stable medium that solubilizes both organic substrates and the inorganic base, ensuring homogeneous reaction kinetics. The careful balance of oxidant equivalents, typically around 3.0 equivalents of TBHP, ensures complete conversion of the starting materials while minimizing the formation of sulfone over-oxidation byproducts, thereby enhancing the overall purity profile of the crude reaction mixture.
How to Synthesize Cycloenamine Sulfone Derivatives Efficiently
To implement this synthesis effectively in a laboratory or pilot plant setting, operators should adhere to the optimized parameters identified in the patent examples, which highlight the importance of reagent ratios and temperature control. The standard procedure involves dissolving the piperidine derivative and sulfonyl hydrazide in 1,4-dioxane, followed by the sequential addition of iodine, sodium bicarbonate, and TBHP. The reaction mixture is then heated to approximately 100°C and stirred for 3 to 12 hours under air, monitoring progress via TLC or HPLC until the starting materials are fully consumed. Detailed standardized synthesis steps see the guide below.
- Combine piperidine derivative and sulfonyl hydrazide in an organic solvent such as 1,4-dioxane.
- Add iodine and sodium bicarbonate as the base to the reaction mixture under air atmosphere.
- Introduce TBHP as the oxidant and heat the mixture to 80-100°C for 3 to 12 hours to complete the reaction.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this iodine-mediated synthesis route offers tangible benefits that extend beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the raw material portfolio, as the process relies on commodity chemicals like iodine, TBHP, and sodium bicarbonate, which are available in bulk quantities from multiple global vendors. This diversification of supply sources mitigates the risk of production delays caused by shortages of specialized catalysts or ligands, ensuring a more resilient and continuous manufacturing operation. Furthermore, the elimination of transition metals removes the necessity for expensive scavenging resins or complex filtration systems designed to reduce metal content to ppm levels, directly translating into lower operational expenditures and reduced waste disposal costs.
- Cost Reduction in Manufacturing: The economic impact of replacing precious metal catalysts with elemental iodine cannot be overstated, as it fundamentally alters the cost structure of the synthesis. Iodine is a abundant halogen with a stable market price, whereas palladium or rhodium prices are subject to extreme volatility and geopolitical supply constraints. By removing these expensive inputs, the variable cost per kilogram of the final intermediate is significantly reduced, improving the overall margin profile for the manufacturing entity. Additionally, the one-pot nature of the reaction reduces solvent consumption and labor hours associated with multi-step isolation procedures, contributing to substantial cost savings in utilities and personnel.
- Enhanced Supply Chain Reliability: The robustness of this chemical process enhances supply chain reliability by minimizing sensitivity to environmental variables such as moisture or oxygen levels. Since the reaction proceeds efficiently under air atmosphere, there is no need for specialized nitrogen blanketing infrastructure or glovebox operations, which simplifies facility requirements and reduces maintenance overhead. The stability of the starting materials, particularly the sulfonyl hydrazides and piperidines, allows for long-term storage without significant degradation, enabling manufacturers to maintain strategic stockpiles and respond quickly to fluctuating market demand without risking raw material quality.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this method aligns well with green chemistry principles by reducing the E-factor through higher atom economy and simpler workups. The absence of heavy metals simplifies wastewater treatment protocols, as effluent streams do not require specialized processing to remove toxic metal ions before discharge. This compliance advantage accelerates regulatory approvals for new manufacturing sites and reduces the liability associated with hazardous waste management. The mild thermal conditions also imply lower energy consumption for heating and cooling cycles, supporting corporate sustainability goals and reducing the carbon footprint of the production process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxidative sulfonylation technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a reliable basis for decision-making.
Q: What are the key advantages of this iodine-catalyzed method over traditional transition metal catalysis?
A: This method eliminates the need for expensive and toxic transition metal catalysts, utilizing inexpensive iodine instead. This significantly simplifies downstream purification, reduces heavy metal residue risks in pharmaceutical products, and lowers overall raw material costs.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process utilizes stable, commercially available raw materials and operates under mild conditions (air atmosphere, moderate temperatures). The simplicity of the workup and the robustness of the reaction conditions make it highly amenable to scale-up from kilogram to multi-ton production.
Q: What is the typical purity profile of the resulting cycloenamine sulfone derivatives?
A: The reaction demonstrates high selectivity with minimal byproduct formation when optimized bases like sodium bicarbonate are used. Standard purification techniques such as silica gel chromatography yield high-purity products suitable for further pharmaceutical development.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cycloenamine Sulfone Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of adopting innovative synthetic routes like the one described in CN112341376B to enhance our clients' competitive edge in the pharmaceutical market. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We are committed to delivering high-purity cycloenamine sulfone derivatives that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch.
We invite potential partners to engage with our technical procurement team to discuss how this cost-effective synthesis method can be integrated into your specific supply chain. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the potential economic benefits of switching to this iodine-mediated protocol for your projects. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your unique molecular targets, ensuring a reliable and high-quality supply of these critical pharmaceutical intermediates.
