Advanced Heck Reaction Strategy for Scalable Production of HIV Drug Intermediate IDX-899 Precursors
The pharmaceutical industry continuously seeks robust synthetic pathways for critical antiretroviral agents, particularly non-nucleoside reverse transcriptase inhibitors (NNRTIs) like IDX-899. Patent CN102115452A discloses a groundbreaking preparation method for (E)-3-(3-substituted-5-methylphenyl)acrylonitrile, a pivotal intermediate in the synthesis of this next-generation HIV therapeutic. Unlike traditional approaches that rely on hazardous organometallic reagents, this invention leverages a palladium-catalyzed Heck reaction to construct the carbon-carbon double bond with high efficiency. The strategic shift from Wittig-type olefinations to cross-coupling chemistry represents a significant technological leap, offering superior control over stereochemistry and operational safety. As a reliable pharmaceutical intermediate supplier, understanding these mechanistic nuances is essential for ensuring the consistent quality required for clinical applications. The following analysis details how this novel route addresses the critical pain points of cost, safety, and scalability in modern drug manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods, such as those disclosed in US2008213217A1, typically employ a Wittig reaction sequence involving n-butyllithium and diethyl cyanomethyl phosphate. This conventional pathway presents severe logistical and economic challenges for large-scale production. The use of n-butyllithium necessitates stringent anhydrous and oxygen-free environments, often requiring cryogenic temperatures to manage exothermic risks and prevent reagent decomposition. Furthermore, diethyl cyanomethyl phosphate is not only costly but also generates significant phosphorus-containing waste, complicating environmental compliance and wastewater treatment. The sensitivity of these reagents demands specialized reactor infrastructure and highly trained personnel, driving up the capital expenditure and operational costs significantly. Consequently, the conventional route is ill-suited for the high-volume manufacturing demands of the global HIV treatment market, where cost reduction in API manufacturing is paramount.
The Novel Approach
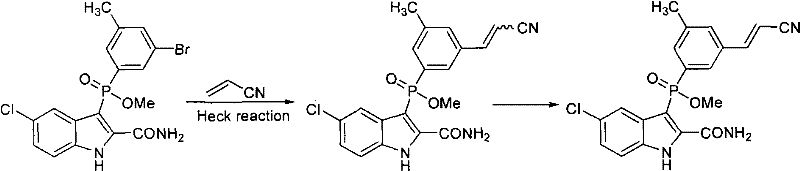
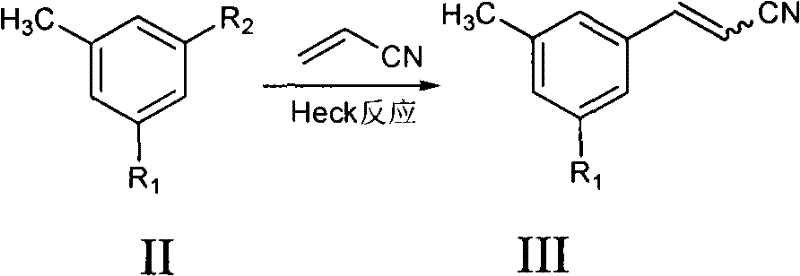
The patented methodology replaces the fragile Wittig sequence with a robust Heck coupling between a 3,5-disubstituted toluene derivative (Formula II) and acrylonitrile. This transformation proceeds under much milder conditions, typically ranging from 20°C to 180°C, eliminating the need for cryogenic cooling. The reaction utilizes readily available palladium catalysts and common organic bases, drastically simplifying the supply chain logistics. By avoiding pyrophoric reagents, the process enhances workplace safety and reduces the complexity of hazard management protocols. Moreover, the Heck reaction exhibits excellent atom economy and generates fewer by-products compared to phosphonate-based olefinations. This streamlined approach not only lowers the raw material costs but also simplifies the downstream purification processes, making it an ideal candidate for cost reduction in pharmaceutical intermediate manufacturing. The versatility of this method allows for various substituents (R1 = NO2, NH2, Br, PG), providing a flexible platform for synthesizing diverse analogues.
Mechanistic Insights into Palladium-Catalyzed Heck Coupling
The core of this innovation lies in the palladium-catalyzed cross-coupling mechanism, which facilitates the formation of the styrene-like double bond with high regioselectivity. The catalytic cycle initiates with the oxidative addition of the aryl halide (Formula II) to the active Pd(0) species, forming an aryl-palladium(II) complex. Subsequent migratory insertion of acrylonitrile into the palladium-carbon bond generates a new alkyl-palladium intermediate. The stereochemical outcome is largely determined during the beta-hydride elimination step, which preferentially yields the thermodynamically more stable trans (E) isomer due to steric factors. The patent specifies that the reaction can utilize various homogeneous catalysts such as Pd(PPh3)4, PdCl2, or Pd(OAc)2, often in conjunction with ligands like triphenylphosphine. The choice of base, ranging from inorganic carbonates to organic amines like triethylamine, plays a crucial role in regenerating the active catalyst and neutralizing the acid by-product. This mechanistic robustness ensures consistent performance across different batches, a critical factor for maintaining stringent purity specifications in GMP environments.
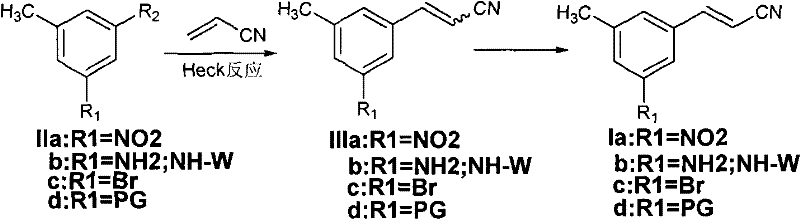
Impurity control is another critical aspect addressed by the patent's mechanistic understanding. While the Heck reaction predominantly yields the trans isomer, minor amounts of the cis (Z) isomer may form depending on the substrate electronics. The patent outlines sophisticated purification strategies to manage this impurity profile. For substrates where the E/Z ratio is favorable (e.g., R1 = NO2 or NH2), simple recrystallization or slurring effectively removes the cis isomer. However, for challenging substrates like the dibromo derivative, where the initial ratio might be closer to 1:1, a double bond configuration transformation is employed. This involves a bromination-elimination sequence using liquid bromine followed by zinc-mediated dehalogenation in acetic acid. This chemical editing step converts the unwanted cis isomer into the desired trans configuration, thereby maximizing overall yield and minimizing material loss. Such precise control over the impurity spectrum demonstrates a deep understanding of reaction engineering, ensuring the final product meets the rigorous quality standards required for subsequent coupling steps in the synthesis of IDX-899.
How to Synthesize (E)-3-(3-Substituted-5-Methylphenyl)Acrylonitrile Efficiently
The synthesis protocol described in the patent offers a practical guide for laboratory and pilot-scale execution. The process begins with the selection of appropriate starting materials, such as 3-bromo-5-nitrotoluene or 3,5-dibromotoluene, which are commercially accessible. The reaction is typically conducted in polar aprotic solvents like DMF or NMP, which facilitate the solubility of both organic substrates and inorganic bases. Temperature control is maintained within the specified range to optimize reaction kinetics while preventing thermal degradation of the acrylonitrile. Post-reaction workup involves standard aqueous extraction and drying procedures, followed by crystallization to isolate the pure E-isomer. For specific derivatives requiring isomerization, the bromination step must be carefully monitored to avoid over-bromination of the aromatic ring. The detailed standardized synthesis steps see the guide below.
- Perform a Heck reaction between a 3,5-disubstituted toluene derivative (Formula II) and acrylonitrile using a palladium catalyst and base.
- Separate the resulting cis/trans isomer mixture (Formula III) via recrystallization or slurring to isolate the trans (E) isomer.
- If necessary, convert unwanted cis isomers to trans via double bond configuration transformation using bromination and zinc elimination.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented Heck reaction route offers transformative benefits that directly impact the bottom line and operational resilience. The elimination of specialized, high-cost reagents like n-butyllithium and cyanomethyl phosphates removes a significant variable from the raw material budget. These legacy reagents often suffer from price volatility and limited supplier availability, creating bottlenecks in production planning. By switching to commodity chemicals like acrylonitrile and standard palladium catalysts, manufacturers can secure more stable pricing contracts and diversify their supplier base. This shift significantly reduces the risk of supply chain disruptions, ensuring continuous availability of critical intermediates for downstream API production. Furthermore, the simplified reaction conditions reduce the dependency on specialized cryogenic infrastructure, allowing production to be transferred to a wider range of contract manufacturing organizations (CMOs) without extensive capital investment.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the replacement of expensive organolithium reagents with economical palladium catalysts and acrylonitrile. Organolithiums require careful handling, storage under inert atmosphere, and often come with high disposal costs due to their hazardous nature. In contrast, the Heck catalysts can often be recovered or used in low loadings, and the waste stream is less hazardous. The avoidance of cryogenic operations also leads to substantial energy savings, as refrigeration systems are among the most energy-intensive units in a chemical plant. Additionally, the higher selectivity of the Heck reaction reduces the burden on purification units, lowering solvent consumption and waste treatment costs. These cumulative effects result in a significantly leaner cost structure for the final intermediate.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials enhances the robustness of the supply chain. 3-Substituted toluenes and acrylonitrile are produced on a massive industrial scale for various sectors, ensuring a steady supply even during market fluctuations. This contrasts sharply with niche reagents like diethyl cyanomethyl phosphate, which may have limited production capacity and longer lead times. By aligning the synthesis with bulk chemical availability, procurement teams can negotiate better terms and maintain lower safety stocks. The operational simplicity also means that lead times for production campaigns can be shortened, as there is less need for extensive equipment preparation and safety checks associated with pyrophoric materials. This agility allows for faster response to market demand changes.
- Scalability and Environmental Compliance: Scaling chemical processes often reveals hidden complexities, but this Heck reaction is inherently scalable due to its thermal stability and lack of sensitive intermediates. The reaction can be performed in standard stainless steel reactors without the need for glass-lined vessels specifically for corrosion resistance against strong bases or cryogens. From an environmental standpoint, the process generates less hazardous waste, simplifying compliance with increasingly strict environmental regulations. The reduction in phosphorus waste and the ability to recycle solvents contribute to a greener manufacturing footprint. This alignment with green chemistry principles not only mitigates regulatory risk but also enhances the corporate sustainability profile, which is increasingly important for partnerships with major pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific experimental data and beneficial effects outlined in the patent documentation. Understanding these details helps stakeholders evaluate the feasibility of adopting this route for their specific supply chain needs. The answers reflect the practical realities of running this chemistry at scale, focusing on yield, purity, and operational parameters.
Q: What are the advantages of the Heck reaction route over the conventional Wittig method for this intermediate?
A: The Heck reaction avoids the use of expensive and hazardous reagents like n-butyllithium and diethyl cyanomethyl phosphate. It operates under milder conditions without strict anhydrous or oxygen-free requirements, significantly reducing operational costs and safety risks associated with large-scale manufacturing.
Q: How is high E-isomer selectivity achieved in this process?
A: The palladium-catalyzed Heck reaction inherently favors the formation of the thermodynamically stable trans (E) isomer. For substrates where the E/Z ratio is lower, the process employs a specific double bond configuration transformation involving bromination followed by zinc-mediated elimination to enrich the desired E-isomer purity to over 99%.
Q: Is this synthesis method suitable for industrial scale-up?
A: Yes, the method is specifically designed for industrial applicability. It utilizes commercially available raw materials, standard reaction equipment capable of handling temperatures between 20-180°C, and avoids cryogenic conditions, making it highly adaptable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (E)-3-(3-Substituted-5-Methylphenyl)Acrylonitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthetic routes in the development of life-saving antiretroviral therapies. Our technical team has extensively analyzed the Heck reaction methodology described in CN102115452A and is fully equipped to translate this laboratory-scale innovation into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot plant to full-scale manufacturing is seamless. Our facilities are designed to handle palladium-catalyzed reactions with rigorous safety protocols and advanced containment systems. We maintain stringent purity specifications through our rigorous QC labs, utilizing state-of-the-art HPLC and NMR instrumentation to verify the E/Z ratio and overall chemical purity of every batch. This commitment to quality ensures that the intermediates we supply meet the exacting standards required for GMP API synthesis.
We invite potential partners to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. Our technical procurement team is ready to provide specific COA data and route feasibility assessments to demonstrate how this optimized Heck reaction can enhance your supply chain efficiency. By leveraging our expertise in process chemistry and scale-up engineering, we can help you secure a reliable source of high-quality intermediates while optimizing your overall production costs. Contact us today to discuss how we can support your HIV drug development programs with superior manufacturing solutions.
