Advanced Metal-Free Synthesis of Sulfur-Containing Polysubstituted Pyrrole Compounds for Commercial Scale-Up
Advanced Metal-Free Synthesis of Sulfur-Containing Polysubstituted Pyrrole Compounds for Commercial Scale-Up
The development of efficient synthetic routes for nitrogen-containing heterocycles remains a cornerstone of modern pharmaceutical chemistry, particularly for constructing complex bioactive scaffolds. Five-membered nitrogen heterocycles, specifically pyrrolidine and pyrrole nuclei, serve as critical structural motifs in a vast array of natural products and therapeutic agents, ranging from alkaloids like (-)-slaframine to advanced clinical candidates such as ABBV-3221. 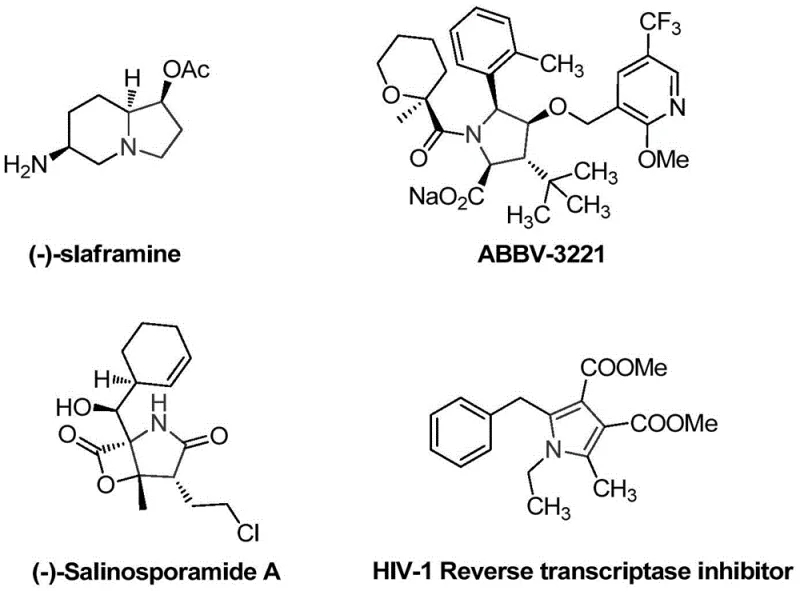 . Recognizing the immense value of these structures, recent intellectual property disclosures, specifically patent CN112574090A, have introduced a groundbreaking preparation method for sulfur-containing polysubstituted pyrrole compounds. This technology addresses long-standing challenges in heterocycle synthesis by offering a pathway that is not only operationally simple but also remarkably robust in terms of functional group tolerance and environmental impact. For R&D directors and procurement strategists alike, this represents a significant opportunity to streamline the production of high-value intermediates without relying on costly transition metal catalysis.
. Recognizing the immense value of these structures, recent intellectual property disclosures, specifically patent CN112574090A, have introduced a groundbreaking preparation method for sulfur-containing polysubstituted pyrrole compounds. This technology addresses long-standing challenges in heterocycle synthesis by offering a pathway that is not only operationally simple but also remarkably robust in terms of functional group tolerance and environmental impact. For R&D directors and procurement strategists alike, this represents a significant opportunity to streamline the production of high-value intermediates without relying on costly transition metal catalysis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of polysubstituted pyrrole skeletons has relied heavily on classical named reactions such as the Hantzsch pyrrole synthesis, Knorr pyrrole synthesis, and Paal-Knorr synthesis. While these methods are foundational, they often suffer from significant limitations when applied to modern drug discovery and process chemistry. Many contemporary variations of these syntheses depend on transition metal catalysis, utilizing expensive and potentially toxic metals like palladium, copper, or rhodium to facilitate cyclization or cross-coupling steps. From a supply chain perspective, the reliance on noble metals introduces volatility in raw material costs and necessitates rigorous downstream processing to meet stringent regulatory limits on residual heavy metals in active pharmaceutical ingredients (APIs). Furthermore, traditional methods frequently require harsh reaction conditions, including high temperatures or strong acidic/basic environments, which can compromise sensitive functional groups and lead to complex impurity profiles that are difficult to separate.
The Novel Approach
In stark contrast to these conventional hurdles, the methodology disclosed in patent CN112574090A presents a transformative approach by utilizing a base-promoted cyclization of sulfonyl-protected β-amino ketone derivatives with prop-2-ynylsulfonium salt compounds. This metal-free strategy operates under exceptionally mild conditions, typically between 0°C and 50°C, with a preferred temperature of 30°C, thereby drastically reducing energy consumption and safety risks associated with exothermic runaway reactions. 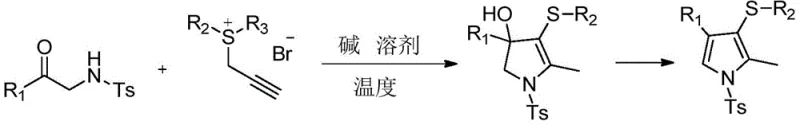 . The reaction proceeds efficiently in common organic solvents such as tetrahydrofuran (THF), avoiding the need for specialized or hazardous media. By eliminating the dependence on transition metals, this novel route simplifies the purification process, often requiring only filtration and silica gel column chromatography to achieve high-purity products. This shift not only enhances the economic viability of the process but also aligns perfectly with green chemistry principles, making it an attractive option for sustainable manufacturing.
. The reaction proceeds efficiently in common organic solvents such as tetrahydrofuran (THF), avoiding the need for specialized or hazardous media. By eliminating the dependence on transition metals, this novel route simplifies the purification process, often requiring only filtration and silica gel column chromatography to achieve high-purity products. This shift not only enhances the economic viability of the process but also aligns perfectly with green chemistry principles, making it an attractive option for sustainable manufacturing.
Mechanistic Insights into Base-Promoted Cyclization
The core of this synthetic innovation lies in the unique reactivity of the prop-2-ynylsulfonium salt acting as an electrophilic partner towards the nucleophilic nitrogen of the sulfonyl-protected β-amino ketone. Under the influence of a base such as triethylamine (TEA), the reaction initiates through a nucleophilic attack that triggers a cascade of intramolecular cyclization events. The mechanism likely involves the formation of an intermediate species, specifically a sulfur-containing polysubstituted pyrroline derivative (Structure A in the patent), which can subsequently undergo an elimination reaction to yield the fully aromatic pyrrole system (Structure B). This mechanistic pathway is highly advantageous because it allows for the precise installation of sulfur substituents at the 3-position of the pyrrole ring, a modification that is notoriously difficult to achieve with high regioselectivity using other methods. The presence of the tosyl (Ts) protecting group on the nitrogen atom plays a crucial role in stabilizing the intermediate and directing the cyclization, ensuring that the reaction proceeds cleanly to the desired polysubstituted product.
From an impurity control standpoint, the mildness of the reaction conditions contributes significantly to a cleaner crude profile. The use of THF as a solvent and TEA as a base minimizes side reactions such as polymerization of the alkyne or hydrolysis of the sulfonium salt, which are common pitfalls in more aggressive protocols. Experimental data from the patent indicates that optimizing the molar ratio of reactants is key to maximizing yield; for instance, using a 1:9 ratio of the ketone derivative to the sulfonium salt, along with 3.0 equivalents of base, has been shown to drive the reaction to completion with yields often exceeding 60%. This level of control over the reaction parameters allows process chemists to fine-tune the synthesis for specific substrates, ensuring consistent quality and minimizing the formation of hard-to-remove byproducts that could otherwise complicate the supply chain.
How to Synthesize Sulfur-Containing Polysubstituted Pyrroles Efficiently
To implement this synthesis effectively in a laboratory or pilot plant setting, operators should follow a standardized protocol that emphasizes precise stoichiometry and temperature control. The process begins by dissolving the sulfonyl-protected β-amino ketone derivative in dry tetrahydrofuran to ensure a homogeneous reaction mixture. Subsequently, the prop-2-ynylsulfonium salt is added slowly under vigorous stirring to manage any potential exotherm, although the reaction is generally mild. Finally, the base is introduced to initiate the cyclization. Detailed standard operating procedures and specific stoichiometric guidelines for scaling this reaction are provided in the technical section below.
- Dissolve the sulfonyl-protected beta-aminoketone derivative in a suitable solvent such as tetrahydrofuran (THF).
- Slowly add the prop-2-ynylsulfonium salt compound (e.g., dimethyl(prop-2-yn-1-yl)sulfonium bromide) to the solution under stirring.
- Add a base such as triethylamine (TEA) at a controlled temperature (preferably 30°C) and stir until the reaction is complete, followed by filtration and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metal-free synthesis route offers tangible strategic benefits that extend beyond simple yield metrics. The primary advantage lies in the drastic simplification of the raw material portfolio. By removing the requirement for precious metal catalysts, manufacturers can avoid the price volatility and supply constraints often associated with palladium or rhodium sourcing. This shift directly translates to cost reduction in pharmaceutical intermediate manufacturing, as the expense of purchasing expensive catalysts and the specialized scavenger resins needed for their removal is entirely eliminated. Furthermore, the simplified workup procedure—often involving just filtration and concentration—reduces the consumption of solvents and silica gel, lowering both material costs and waste disposal fees.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts removes a significant cost center from the production budget. Without the need for expensive metals or complex purification steps like charcoal treatment or metal scavenging, the overall cost of goods sold (COGS) is significantly lowered. Additionally, the high atom economy of the reaction and the ability to use inexpensive bases like triethylamine further enhance the economic efficiency of the process, allowing for competitive pricing in the global market for fine chemical intermediates.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals such as THF, TEA, and readily available sulfonium salts ensures a stable and resilient supply chain. Unlike specialized catalysts that may have long lead times or single-source dependencies, the reagents for this process are widely produced and easily accessible. This availability reduces the risk of production delays due to raw material shortages, ensuring consistent delivery schedules for downstream API manufacturers. The robustness of the reaction also means that batch-to-batch variability is minimized, fostering trust and long-term partnerships with clients.
- Scalability and Environmental Compliance: The mild reaction conditions (0-50°C) and the absence of toxic heavy metals make this process inherently safer and easier to scale from gram to ton quantities. The reduced environmental footprint, characterized by lower E-factors and the absence of heavy metal waste streams, simplifies regulatory compliance and environmental permitting. This alignment with green chemistry standards not only mitigates regulatory risk but also enhances the corporate sustainability profile, which is increasingly important for multinational pharmaceutical partners seeking eco-friendly suppliers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these sulfur-containing pyrrole compounds. These answers are derived directly from the experimental data and claims within the patent documentation to provide accurate guidance for potential partners.
Q: Does this synthesis method require transition metal catalysts?
A: No, the method described in patent CN112574090A is metal-free. It utilizes a base-promoted cyclization strategy, eliminating the need for expensive palladium or copper catalysts and the subsequent heavy metal removal steps.
Q: What are the optimal reaction conditions for high yield?
A: The patent data indicates that using tetrahydrofuran (THF) as the solvent and triethylamine (TEA) as the base at a temperature of 30°C provides excellent yields, often exceeding 60% for various substrates.
Q: Can this method tolerate diverse functional groups on the substrate?
A: Yes, the method demonstrates broad substrate scope. It successfully tolerates various substituents on the phenyl ring, including electron-withdrawing groups like fluorine, chlorine, bromine, and trifluoromethyl, as well as electron-donating groups like methoxy and methyl.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sulfur-Containing Polysubstituted Pyrrole Supplier
The technological breakthroughs detailed in patent CN112574090A highlight the immense potential of sulfur-containing polysubstituted pyrroles as versatile building blocks for next-generation therapeutics. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate this innovative chemistry from the laboratory bench to commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from clinical trials to market launch. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch meets the highest international standards for pharmaceutical intermediates.
We invite you to collaborate with us to leverage this efficient, metal-free synthesis for your specific drug development programs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this route can optimize your budget. Please contact us today to request specific COA data for our available inventory or to discuss route feasibility assessments for your custom synthesis projects. Let us be your partner in delivering high-quality, cost-effective chemical solutions.
