Scalable Production of High-Purity Dihydroquinazolinone Intermediates Using Recyclable Ionic Liquids
Scalable Production of High-Purity Dihydroquinazolinone Intermediates Using Recyclable Ionic Liquids
The pharmaceutical industry constantly seeks robust synthetic routes that balance high purity with environmental sustainability and cost efficiency. A significant breakthrough in this domain is detailed in patent CN112979563B, which discloses a novel preparation method for the medical intermediate 2,3-diaryl-2,3-dihydroquinazoline-4[1H]-ketone. This technology leverages the unique properties of alkaline ionic liquids to overcome the longstanding limitations of traditional solid-base catalysis. By shifting from harsh, non-recyclable conditions to a mild, recyclable ionic liquid system, manufacturers can achieve superior atom economy and drastically simplify purification workflows. For R&D directors and procurement managers alike, this represents a pivotal opportunity to optimize the supply chain for complex heterocyclic intermediates, ensuring reliable access to high-quality materials while adhering to stricter environmental regulations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2,3-diaryl-2,3-dihydroquinazolin-4[1H]-ones has relied on methods such as the solvent-free reaction of isatoic anhydride and Schiff bases using sodium hydroxide (NaOH) as a catalyst. While earlier literature suggested benefits like high yield, practical industrial application revealed severe drawbacks. The solid alkali catalyst cannot be recycled, leading to substantial chemical waste and increased disposal costs that erode profit margins. Furthermore, these conventional routes often generate complex by-product profiles, necessitating rigorous and multi-step purification processes involving water washing and recrystallization. This complexity not only extends production lead times but also introduces variability in product quality, making it difficult to guarantee the stringent purity specifications required for pharmaceutical grade intermediates on a continuous large scale.
The Novel Approach
The innovative methodology described in the patent introduces a basic ionic liquid catalyst that fundamentally transforms the reaction landscape. Unlike traditional solid bases, this ionic liquid functions dually as both a highly active catalyst and a polar reaction medium, facilitating rapid cyclization under mild reflux conditions. The process utilizes a mixed solvent system of ethanol and dimethylformamide, which enhances the solubility of reactants while maintaining poor miscibility with the lipophilic product, thereby promoting spontaneous precipitation. This physical property allows for simple filtration to isolate the crude product, bypassing the need for complex chromatography or extensive recrystallization. The result is a streamlined workflow that delivers high-purity intermediates with minimal operational friction, perfectly aligning with the needs of a reliable pharmaceutical intermediate supplier aiming for efficiency.
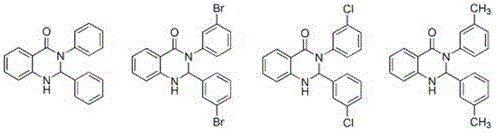
Mechanistic Insights into Basic Ionic Liquid-Catalyzed Cyclization
The core of this technological advancement lies in the specific molecular architecture of the basic ionic liquid catalyst. As illustrated in the structural data, the catalyst incorporates basic functional groups directly into the ionic liquid framework, combining the high activity of liquid alkalis with the separation ease of solid bases. This dual functionality accelerates the nucleophilic attack of the Schiff base nitrogen on the carbonyl carbon of the isatoic anhydride, driving the ring-closing reaction to completion within a remarkably short timeframe of 12 to 18 minutes. The strong polarity of the ionic liquid stabilizes the transition states involved in the cyclization, ensuring high conversion rates even at relatively low catalyst loadings of 3-7% relative to the Schiff base substrate.
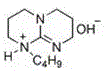
Beyond kinetics, the mechanism offers profound advantages in impurity control and product isolation. The basic ionic liquid exhibits high selectivity, effectively suppressing side reactions that typically plague traditional base-catalyzed condensations. Moreover, the distinct polarity difference between the ionic liquid catalyst system and the fat-soluble quinazolinone product creates a natural phase separation upon cooling. This phenomenon allows the product to precipitate out as a solid while the catalyst remains dissolved in the mother liquor. Consequently, the filtrate containing the catalyst and solvent can be directly recycled for subsequent batches, significantly reducing raw material consumption and waste generation without compromising the purity profile of the final API intermediate.
How to Synthesize 2,3-Diaryl-2,3-dihydroquinazolin-4(1H)-one Efficiently
Implementing this synthesis route requires precise control over solvent ratios and reaction parameters to maximize the benefits of the ionic liquid system. The process begins by dissolving isatoic anhydride and the specific Schiff base derivative in a mixed solvent of ethanol and DMF (volume ratio 9:1 to 9:1.5), followed by the addition of the basic ionic liquid. The mixture is then heated to reflux, where the reaction proceeds rapidly to completion. Upon cooling, the product crystallizes out and is isolated via vacuum filtration, washed with the same mixed solvent to remove residual impurities, and dried. For a comprehensive understanding of the operational parameters and safety considerations, the detailed standardized synthesis steps are provided in the guide below.
- Mix isatoic anhydride and Schiff base in an ethanol/DMF solvent system, then add the basic ionic liquid catalyst.
- Heat the reaction mixture to reflux for 12-18 minutes to complete the cyclization.
- Cool the mixture to precipitate the solid product, filter, wash with the mixed solvent, and dry to obtain high-purity intermediates.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this ionic liquid-catalyzed process offers tangible strategic benefits that extend beyond mere chemical yield. The ability to recycle the catalytic system multiple times without significant loss of activity translates directly into substantial cost savings on raw materials and waste treatment. By eliminating the need for expensive transition metal catalysts or harsh mineral acids, the process reduces the burden on downstream purification infrastructure, allowing for faster batch turnover and improved facility utilization rates. This efficiency is critical for maintaining competitive pricing in the global market for fine chemical intermediates while adhering to increasingly stringent environmental compliance standards.
- Cost Reduction in Manufacturing: The recyclability of the basic ionic liquid catalyst is a primary driver for cost optimization. Since the catalyst remains in the solution phase after product precipitation, the mother liquor can be replenished with fresh reactants and reused for multiple cycles. This eliminates the recurring cost of purchasing stoichiometric amounts of base for every batch and significantly lowers the expense associated with neutralizing and disposing of alkaline waste streams. Furthermore, the simplified purification protocol reduces solvent consumption and energy usage during drying, contributing to a leaner and more cost-effective manufacturing operation overall.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures consistent product quality and availability, which is paramount for securing long-term supply contracts. The use of readily available solvents like ethanol and DMF, combined with a catalyst that does not degrade rapidly, minimizes the risk of production delays caused by raw material shortages or complex reagent synthesis. The short reaction time of approximately 15 minutes allows for high throughput in existing reactor vessels, enabling suppliers to respond agilely to fluctuating market demands and reducing lead times for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: Scaling this process from laboratory to industrial production is straightforward due to the absence of hazardous reagents and the simplicity of the workup procedure. The reduction in by-product formation means less chemical waste is generated per kilogram of product, easing the load on wastewater treatment facilities and helping manufacturers meet green chemistry goals. The process inherently supports sustainable manufacturing practices by maximizing atom economy and minimizing the environmental footprint, making it an attractive option for companies aiming to enhance their corporate social responsibility profiles through greener supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ionic liquid catalysis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on how this method compares to legacy processes. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for commercial production of heterocyclic scaffolds.
Q: What are the primary advantages of using basic ionic liquids over traditional NaOH catalysts?
A: Basic ionic liquids offer superior recyclability, eliminating the waste associated with single-use solid alkalis. They also provide higher selectivity, resulting in fewer by-products and simplifying the downstream purification process significantly.
Q: How does this method improve supply chain reliability for pharmaceutical intermediates?
A: By utilizing a recyclable catalytic system and a simplified solvent recovery process, the method reduces raw material consumption and waste disposal costs. This stability ensures consistent production capacity and shorter lead times for bulk orders.
Q: Is this synthesis method suitable for large-scale industrial manufacturing?
A: Yes, the process operates under mild reflux conditions with short reaction times (12-18 minutes) and uses common solvents like ethanol and DMF. The ease of product isolation via simple filtration makes it highly adaptable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3-Diaryl-2,3-dihydroquinazolin-4(1H)-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the evolving needs of the global pharmaceutical sector. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the ionic liquid-catalyzed synthesis of dihydroquinazolinones can be seamlessly transferred to industrial scale. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of intermediate supplied meets the exacting standards required for downstream API synthesis and drug development projects.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this greener, more efficient process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our capability as a trusted partner in delivering high-value chemical solutions for your supply chain.
