Advanced One-Pot Palladium Catalysis for Scalable 2-(3-Furyl)acetamide Production
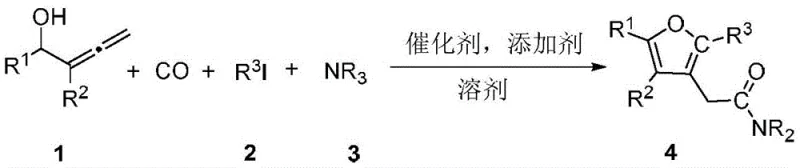
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to access bioactive heterocyclic scaffolds, particularly those containing furan moieties which are prevalent in numerous drug candidates. Patent CN108794437B, published in late 2022, introduces a groundbreaking synthetic methodology for preparing 2-(3-furyl)acetamide compounds through a highly atom-economical one-pot tandem reaction. This innovation addresses critical bottlenecks in traditional organic synthesis by merging carbonylation, coupling, and cyclization into a single operational sequence. By utilizing readily available 2-substituted-2,3-butadien-1-ols, carbon monoxide, aryl iodides, and amines, this protocol eliminates the need for pre-functionalized intermediates that often plague legacy manufacturing routes. For R&D directors and process chemists, this represents a significant leap forward in constructing complex molecular architectures with minimal step count, thereby enhancing the overall feasibility of producing high-purity pharmaceutical intermediates on a commercial scale.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-(3-furyl)acetamide derivatives has relied on several distinct but inefficient strategies that impose heavy burdens on both cost and timeline. Traditional routes often involve the direct amination of 2-(3-furyl)acetic acid, which requires harsh reaction conditions and generates significant waste streams during activation and coupling. Alternatively, palladium-catalyzed cross-coupling reactions between 1-bromo-2-amidoethylenes or chloroacetamides and 3-furanboronic acids have been employed; however, these methods suffer from the high cost and limited availability of specialized boronic acid reagents and halogenated amide precursors. Furthermore, condensation reactions involving 2-en-4-yne amides and 1,3-diketones introduce additional complexity regarding regioselectivity and purification. These conventional approaches are characterized by繁琐 (cumbersome) multi-step procedures, stringent temperature requirements, and difficult-to-source starting materials, all of which severely limit their practical utility in a fast-paced industrial environment where speed-to-market is paramount.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN108794437B leverages a sophisticated palladium-catalyzed carbonylative cyclization strategy that streamlines the entire synthesis into a single vessel. This novel approach utilizes inexpensive and commercially abundant allene alcohols as the core building block, reacting them directly with carbon monoxide gas, aryl iodides, and simple amines. The reaction proceeds smoothly under mild thermal conditions, typically between 60°C and 100°C, which drastically reduces energy consumption compared to high-temperature alternatives. By integrating the formation of the furan ring and the amide bond simultaneously, this method bypasses the isolation of unstable intermediates, thereby minimizing material loss and exposure to hazardous reagents. The versatility of this system is demonstrated by its tolerance to a wide range of substituents on the aromatic rings, allowing for the rapid generation of diverse chemical libraries essential for drug discovery programs without the need for extensive process re-optimization.
Mechanistic Insights into Pd-Catalyzed Carbonylative Cyclization
The success of this transformation hinges on a carefully orchestrated catalytic cycle initiated by the oxidative addition of the aryl iodide to the active palladium(0) species generated in situ. Following this activation, the coordination and subsequent migratory insertion of carbon monoxide into the palladium-carbon bond creates an acyl-palladium intermediate, which is the key precursor for amide bond formation. Simultaneously, the hydroxyl group of the allene alcohol substrate participates in a nucleophilic attack or coordination event that facilitates the construction of the furan ring system through an intramolecular cyclization pathway. The presence of additives such as pivalic acid or phosphine ligands plays a critical role in stabilizing the catalytic center and promoting the turnover frequency, ensuring that the reaction reaches completion within a reasonable timeframe, typically around 8 hours. This mechanistic elegance allows for the precise assembly of the 2-(3-furyl)acetamide core while maintaining high levels of regioselectivity, which is crucial for avoiding the formation of difficult-to-remove isomeric impurities that could compromise the quality of the final API intermediate.
From an impurity control perspective, the mild reaction conditions and the one-pot nature of the process significantly reduce the generation of side products associated with multi-step syntheses. In traditional multi-step routes, each isolation and purification stage introduces opportunities for degradation, oxidation, or hydrolysis of sensitive functional groups. By contrast, this tandem reaction minimizes the exposure of reactive intermediates to the external environment, thereby preserving the integrity of the molecular structure throughout the synthesis. The use of common solvents like acetonitrile or DMF further simplifies the downstream processing, as these solvents are well-understood in terms of removal and recycling. For quality assurance teams, this translates to a cleaner crude reaction profile, which eases the burden on purification columns and crystallization steps, ultimately leading to a final product that meets stringent purity specifications required for pharmaceutical applications with greater consistency and reliability.
How to Synthesize 2-(3-Furyl)acetamide Efficiently
To implement this robust synthetic route in a laboratory or pilot plant setting, operators must adhere to specific stoichiometric ratios and environmental controls to maximize yield and reproducibility. The patent details a optimized protocol where the molar ratio of the allene alcohol, aryl iodide, amine, catalyst, and additive is carefully balanced, typically ranging from 1:1-2:3-5:0.05-0.2:0.2-0.6. This precise formulation ensures that the catalytic cycle is sustained without excessive accumulation of unreacted starting materials that could complicate purification. The reaction is conducted under a carbon monoxide atmosphere, usually at 1 atm pressure, which is safe and manageable in standard high-pressure reactors equipped with appropriate gas handling systems. Detailed standardized synthesis steps for executing this transformation are provided in the guide below, ensuring that technical teams can replicate the high yields reported in the patent examples, such as the 70% yield achieved in the model reaction producing compound 4a.
- Dissolve 2-substituted-2,3-butadien-1-ol, aryl iodide, and amine in a solvent such as acetonitrile.
- Add palladium catalyst (e.g., PdCl2) and additive (e.g., PivOH) to the reaction mixture under a CO atmosphere.
- Stir the reaction at 60-100°C for approximately 8 hours, then quench and purify via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic methodology offers profound strategic benefits that extend beyond mere technical novelty. The shift towards a one-pot process fundamentally alters the cost structure of manufacturing these valuable intermediates by eliminating the need for multiple reactor vessels, extensive solvent swaps, and intermediate quality control testing. This consolidation of steps leads to a drastic simplification of the production workflow, which inherently reduces the labor hours and facility occupancy time required per batch. Furthermore, the reliance on commodity chemicals like carbon monoxide and simple aryl iodides, rather than exotic boronic acids or halogenated amides, insulates the supply chain from the volatility often seen in the market for specialized fine chemical reagents. This stability in raw material sourcing is critical for maintaining continuous production schedules and meeting the demanding delivery timelines of downstream pharmaceutical clients.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven primarily by the elimination of expensive transition metal catalysts often required in alternative cross-coupling strategies and the reduction of waste disposal costs associated with multi-step purifications. By avoiding the use of costly boronic acid derivatives and minimizing the number of unit operations, the overall cost of goods sold (COGS) is significantly lowered. The process utilizes catalytic amounts of palladium, which can potentially be recovered and recycled, further enhancing the economic viability of the route. Additionally, the high atom economy of the carbonylation reaction ensures that a maximum proportion of the raw material mass is incorporated into the final product, reducing the financial loss associated with byproduct formation and improving the overall material efficiency of the manufacturing campaign.
- Enhanced Supply Chain Reliability: The use of widely available starting materials such as 2-substituted-2,3-butadien-1-ols and common amines ensures a robust and resilient supply chain that is less susceptible to disruptions. Unlike specialized reagents that may have single-source suppliers or long lead times, the inputs for this reaction are produced by multiple chemical manufacturers globally, providing procurement teams with greater flexibility and negotiating power. The simplified operational protocol also reduces the risk of batch failures due to complex handling requirements, thereby increasing the reliability of supply. This dependability is essential for securing long-term contracts with major pharmaceutical companies that require guaranteed continuity of supply for their critical drug development programs and commercial launches.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this green chemistry approach aligns perfectly with modern sustainability goals by reducing solvent consumption and waste generation. The one-pot nature of the reaction means fewer work-up procedures and less aqueous waste, which lowers the burden on wastewater treatment facilities and reduces the environmental footprint of the manufacturing site. The mild reaction temperatures (60-100°C) also contribute to lower energy consumption compared to high-temperature processes, supporting corporate carbon reduction targets. These factors make the technology highly scalable, allowing for seamless transition from kilogram-scale laboratory synthesis to ton-scale commercial production without the need for major engineering modifications or safety overhauls, ensuring compliance with increasingly strict environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on reaction conditions, substrate scope, and operational parameters. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their specific project needs and for procurement professionals assessing the potential impact on their supply chain strategy.
Q: What are the key advantages of this one-pot synthesis method?
A: The method avoids cumbersome intermediate separation, utilizes readily available raw materials like allene alcohols and CO, and operates under mild conditions (60-100°C), significantly reducing environmental burden and operational complexity.
Q: What catalysts and additives are optimal for this transformation?
A: The patent specifies palladium chloride (PdCl2) or palladium acetate as effective catalysts, with pivalic acid (PivOH), L-proline, or phosphines serving as crucial additives to facilitate the tandem carbonylation and cyclization process.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is designed for industrial production due to its one-pot nature, wide substrate scope, and use of common solvents like acetonitrile, which simplifies scale-up and waste management compared to multi-step traditional routes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(3-Furyl)acetamide Supplier
As the global demand for complex heterocyclic intermediates continues to rise, partnering with a technically proficient CDMO is essential for navigating the challenges of modern drug development. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from benchtop discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped to handle the specific requirements of palladium-catalyzed reactions, including rigorous QC labs that enforce stringent purity specifications to guarantee the quality of every batch. We understand the critical nature of timeline and cost in the pharmaceutical industry, and our team is dedicated to optimizing processes like the one described in CN108794437B to deliver maximum value to our partners.
We invite you to engage with our technical procurement team to discuss how this advanced synthetic technology can be tailored to your specific molecule requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this efficient one-pot route for your supply chain. We encourage you to contact us today to obtain specific COA data for similar structures and to receive comprehensive route feasibility assessments that will empower your decision-making process. Let us help you accelerate your development timeline and secure a reliable supply of high-quality intermediates for your next breakthrough therapy.
