Advanced Solvent-Free Synthesis of Indolizine Derivatives for Commercial Pharmaceutical Manufacturing
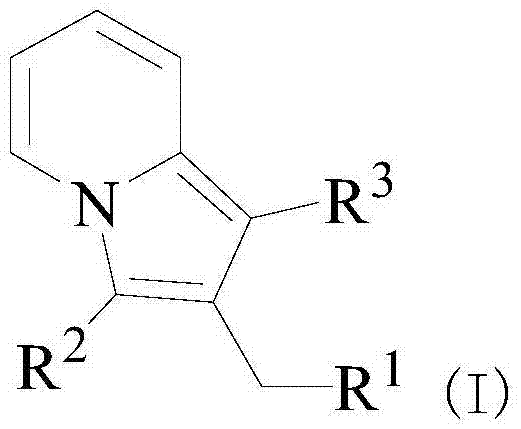
In the rapidly evolving landscape of oncology drug development, the efficient and sustainable construction of nitrogen-containing heterocyclic scaffolds remains a pivotal challenge for process chemists and supply chain strategists alike. Patent CN103641827A introduces a groundbreaking methodology for the synthesis of a novel class of indolizine derivatives, which have demonstrated significant potential as antitumor agents, particularly against gastric cancer cell lines such as MGC80-3. This technology leverages samarium trifluoromethanesulfonate as a potent Lewis acid catalyst to drive the cyclization of propargyl alcohols and 2-alkylpyridine compounds under strictly solvent-free conditions. By eliminating the need for volatile organic compounds (VOCs) during the reaction phase, this approach not only addresses critical environmental compliance issues but also streamlines the purification workflow, offering a compelling value proposition for manufacturers seeking reliable pharmaceutical intermediates supplier partnerships. The structural versatility of the resulting indolizine core, as depicted in the general formula below, allows for extensive derivatization to optimize biological activity and pharmacokinetic profiles.
The strategic implementation of this solvent-free protocol represents a paradigm shift from traditional synthetic routes that heavily rely on bulk solvents to mediate reaction kinetics and heat transfer. Historically, the synthesis of complex heterocycles like indolizines has been plagued by the necessity for large volumes of organic solvents, which not only inflate raw material costs but also generate substantial hazardous waste streams requiring expensive disposal or energy-intensive recovery processes. Conventional methods often suffer from poor atom economy and require rigorous drying of reagents to prevent catalyst deactivation, adding layers of operational complexity and time consumption to the manufacturing schedule. Furthermore, the presence of residual solvents in the final active pharmaceutical ingredient (API) intermediate necessitates additional drying steps and stringent quality control testing to meet regulatory limits, thereby extending the overall lead time for high-purity pharmaceutical intermediates. In contrast, the novel approach detailed in the patent utilizes the intrinsic reactivity of the substrates in a neat state, significantly reducing the reaction mass intensity and simplifying the thermal management of the process.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic pathways for constructing the indolizine ring system typically involve multi-step sequences or transition metal-catalyzed couplings that demand anhydrous conditions and inert atmospheres, driving up capital expenditure and operational overhead. The reliance on solvents such as dichloromethane, toluene, or DMF creates a significant environmental burden, contributing to the facility's carbon footprint and complicating waste management logistics. Moreover, the removal of these solvents post-reaction often requires high-vacuum distillation or chromatographic purification, which can lead to product degradation or loss of yield, especially for thermally sensitive intermediates. The accumulation of solvent residues can also interfere with downstream crystallization processes, resulting in inconsistent particle size distribution and purity profiles that fail to meet the rigorous specifications required for clinical-grade materials. These inefficiencies cumulatively erode profit margins and introduce supply chain vulnerabilities, as fluctuations in solvent availability or pricing can directly impact production continuity and cost stability.
The Novel Approach
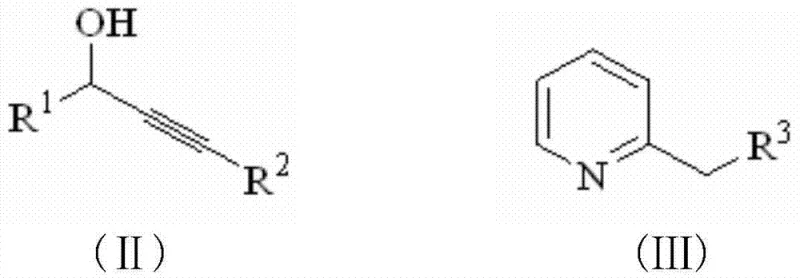
The innovative methodology disclosed in CN103641827A overcomes these hurdles by employing a direct condensation reaction between propargyl alcohol derivatives and 2-alkylpyridines in the absence of any external solvent medium. As illustrated in the reaction scheme below, the process initiates with the mixing of the solid or liquid reactants in a stoichiometric ratio, followed by the addition of a catalytic amount of samarium trifluoromethanesulfonate. The reaction mixture is then heated to a moderate temperature range of 100 to 160 degrees Celsius, where the molten reactants serve as both the substrate and the reaction medium, ensuring excellent molecular contact and heat distribution. This solvent-free environment promotes a cleaner reaction profile with fewer side products, facilitating easier isolation of the target indolizine derivatives through simple aqueous workup and extraction. The elimination of solvent recovery units and the reduction in reactor volume requirements translate directly into substantial cost reduction in pharmaceutical intermediates manufacturing, while the simplified operation enhances overall process safety and scalability.
Mechanistic Insights into Sm(OTf)3-Catalyzed Cyclization
The efficacy of this transformation hinges on the unique Lewis acidic properties of samarium trifluoromethanesulfonate, which acts as a robust activator for the hydroxyl group of the propargyl alcohol moiety. Upon coordination with the samarium center, the hydroxyl group becomes a superior leaving group, facilitating the generation of a propargylic cation or a highly electrophilic intermediate that is susceptible to nucleophilic attack by the nitrogen atom of the 2-alkylpyridine. This initial N-alkylation step is followed by an intramolecular cyclization, likely proceeding through a 6-endo-dig or 5-exo-dig pathway depending on the specific substitution pattern, to close the five-membered ring characteristic of the indolizine skeleton. The lanthanide catalyst is particularly advantageous due to its high tolerance towards moisture and oxygen compared to other Lewis acids like aluminum chloride or boron trifluoride, allowing the reaction to proceed efficiently even without rigorous exclusion of atmospheric elements. This stability ensures consistent catalytic turnover and minimizes the formation of tarry by-products that often complicate the purification of heterocyclic compounds synthesized via harsher acidic conditions.
From an impurity control perspective, the mildness of the samarium catalyst and the absence of aggressive solvents contribute to a cleaner crude reaction mixture, which is critical for maintaining high purity specifications in API intermediates. The reaction conditions are tuned to favor the thermodynamic product, minimizing the occurrence of regioisomers or polymerization side reactions that are common in uncatalyzed thermal cycloadditions. Post-reaction, the catalyst can be effectively separated during the aqueous wash steps, as the samarium salts are generally water-soluble, leaving the organic product in the extraction phase. This inherent separability reduces the risk of heavy metal contamination in the final product, a key concern for regulatory compliance in pharmaceutical manufacturing. The ability to fine-tune the substituent groups R1, R2, and R3 on the indolizine core allows chemists to modulate the electronic and steric properties of the molecule, optimizing its interaction with biological targets while maintaining a streamlined synthetic route that supports commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize Indolizine Derivatives Efficiently
The practical execution of this synthesis involves a straightforward protocol where the propargyl alcohol and 2-alkylpyridine are combined in a reaction vessel equipped with a mechanical stirrer and heating mantle. The catalyst is added, and the temperature is ramped to the optimal range of 120 to 140 degrees Celsius, where the reaction progress is monitored via thin-layer chromatography (TLC) to ensure complete conversion of the starting materials. Upon completion, the reaction mass is cooled to ambient temperature and quenched by pouring into water, followed by extraction with ethyl acetate or diethyl ether to isolate the organic layer. The combined organic extracts are washed with brine to remove residual inorganic salts and dried over anhydrous sodium sulfate before concentration under reduced pressure. The resulting crude solid or oil is then purified using silica gel column chromatography with a gradient of ethyl acetate in petroleum ether to afford the pure indolizine derivative. For a detailed, step-by-step guide including specific molar ratios and purification parameters, please refer to the standardized synthesis protocol below.
- Mix propargyl alcohol derivatives and 2-alkylpyridine compounds in a stoichiometric ratio without additional solvents.
- Add samarium trifluoromethanesulfonate catalyst (5-20 mol%) to the reaction mixture.
- Heat the mixture to 100-160°C and stir for 20-48 hours until TLC indicates completion, followed by standard aqueous workup and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this solvent-free technology offers transformative benefits that extend beyond mere chemical efficiency, impacting the bottom line through reduced operational expenditures and enhanced logistical flexibility. The elimination of bulk solvents removes a significant variable cost component from the bill of materials, while simultaneously negating the need for expensive solvent recovery infrastructure and the associated energy consumption for distillation. This simplification of the process flow reduces the turnaround time between batches, allowing for higher throughput within existing manufacturing facilities without the need for major capital investment in new equipment. Furthermore, the reduced generation of hazardous waste lowers disposal costs and mitigates regulatory risks associated with environmental compliance, making the supply chain more resilient to changing legislative landscapes regarding industrial emissions and waste management.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the complete removal of organic solvents from the reaction phase, which traditionally accounts for a substantial portion of raw material expenses and waste treatment fees. By operating under neat conditions, the process intensity is maximized, meaning more product can be produced per unit volume of reactor space, effectively lowering the fixed cost allocation per kilogram of output. Additionally, the use of a recoverable or low-loading lanthanide catalyst minimizes the consumption of precious metal resources, further contributing to a leaner cost structure that supports competitive pricing strategies in the global market for high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The simplicity of the reagent profile, consisting primarily of readily available propargyl alcohols and alkylpyridines, reduces dependency on specialized or scarce chemical inputs that often cause supply bottlenecks. The robustness of the reaction conditions, which do not require stringent anhydrous environments or inert gas blanketing, simplifies the operational requirements for contract manufacturing organizations (CMOs), broadening the pool of qualified suppliers capable of executing the synthesis. This flexibility ensures a more stable supply of critical intermediates, reducing the risk of production delays caused by raw material shortages or equipment failures related to complex solvent handling systems.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the absence of solvent-related safety hazards such as flash points and vapor pressure buildup, allowing for safer operation in larger reactors. The green chemistry credentials of the method align with corporate sustainability goals, enhancing the brand value of the final pharmaceutical product by demonstrating a commitment to environmentally responsible manufacturing practices. The ease of waste treatment, primarily consisting of aqueous washes and minimal organic residue, simplifies the permitting process for new production lines and ensures long-term operational continuity in regions with strict environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these indolizine derivatives, providing clarity for stakeholders evaluating this technology for integration into their development pipelines. The answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, ensuring accuracy and relevance for decision-making processes. Understanding these nuances is essential for assessing the feasibility of adopting this method for large-scale production and for anticipating potential challenges in process validation and regulatory filing.
Q: What are the primary advantages of the solvent-free method described in CN103641827A?
A: The primary advantages include significant reduction in chemical waste due to the elimination of organic solvents, lower operational costs associated with solvent recovery, and simplified downstream processing, which collectively enhance the environmental profile and economic viability of the manufacturing process.
Q: What is the role of Samarium Trifluoromethanesulfonate in this synthesis?
A: Samarium trifluoromethanesulfonate acts as a highly efficient Lewis acid catalyst that facilitates the cyclization reaction between propargyl alcohols and 2-alkylpyridines under mild, solvent-free conditions, enabling high conversion rates without the need for harsh reagents.
Q: Can this synthesis method be scaled for industrial production of API intermediates?
A: Yes, the method is highly amenable to scale-up due to its solvent-free nature which reduces reactor volume requirements and safety hazards, alongside the use of a robust catalyst system that maintains efficiency at larger batch sizes, making it suitable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indolizine Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating innovative academic research into robust, commercially viable manufacturing processes that meet the exacting standards of the global pharmaceutical industry. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial supply is seamless and efficient. We are committed to delivering high-purity indolizine derivatives that adhere to stringent purity specifications, utilizing our state-of-the-art rigorous QC labs to verify every batch against comprehensive analytical standards. Our dedication to quality and reliability makes us the preferred partner for companies seeking to secure a stable supply of advanced anticancer intermediates.
We invite you to engage with our technical procurement team to discuss how this solvent-free synthesis technology can be tailored to your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this greener manufacturing route for your supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive innovation and efficiency in your drug development programs.
