Advanced Synthesis of Polycyclic Pyridine Compounds for Commercial Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing complex heterocyclic scaffolds, particularly polycyclic pyridine derivatives which serve as critical cores in numerous bioactive molecules. Patent CN109575041B introduces a groundbreaking approach to synthesizing these valuable compounds through a novel silver-catalyzed radical reaction sequence. This technology represents a significant leap forward in synthetic efficiency, utilizing a one-pot two-step strategy that bypasses the laborious isolation of intermediates typically required in conventional pathways. By leveraging silver nitrate as a catalyst in conjunction with potassium persulfate, the process achieves high conversion rates under remarkably mild thermal conditions, specifically maintaining temperatures between 40-50°C during the initial coupling phase. This innovation not only streamlines the synthetic route but also opens new avenues for the cost-effective production of high-purity pharmaceutical intermediates, addressing the growing demand for scalable and environmentally conscious manufacturing processes in the global supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for accessing 5H-chromano[4,3-b]pyridine and related polycyclic structures have long been hindered by significant operational complexities and inefficiencies that impact overall production viability. Historically, these methods rely heavily on the pre-synthesis and isolation of 1,5-dicarbonyl compounds as essential precursors, a step that is notoriously difficult due to the instability and reactivity of these intermediates. The requirement to isolate and purify these 1,5-dicarbonyl species before the final cyclization adds substantial time, labor, and solvent consumption to the manufacturing process, thereby inflating the cost of goods sold. Furthermore, conventional condensation reactions often necessitate harsh conditions or expensive reagents that can lead to the formation of difficult-to-remove impurities, compromising the purity profile required for pharmaceutical applications. These legacy methods struggle to meet the rigorous demands of modern supply chains, where speed, cost-efficiency, and consistent quality are paramount for maintaining competitive advantage in the market.
The Novel Approach
In stark contrast to these legacy limitations, the methodology disclosed in CN109575041B offers a streamlined and highly efficient alternative that fundamentally restructures the synthetic logic for polycyclic pyridine construction. This novel approach utilizes a direct radical coupling between o-allyloxybenzaldehyde derivatives and 1,3-dicarbonyl compounds, effectively generating the necessary 1,5-dicarbonyl intermediate in situ without the need for isolation. By eliminating the intermediate purification step, the process drastically reduces solvent usage and operational time, translating directly into substantial cost savings and a reduced environmental footprint. The use of dimethyl sulfoxide (DMSO) as a solvent combined with a silver nitrate catalyst system allows for precise control over the reaction trajectory, ensuring high selectivity and yield. This one-pot strategy not only simplifies the workflow for chemists but also enhances the scalability of the process, making it an ideal candidate for reliable polycyclic pyridine supplier operations aiming to optimize their manufacturing capabilities.
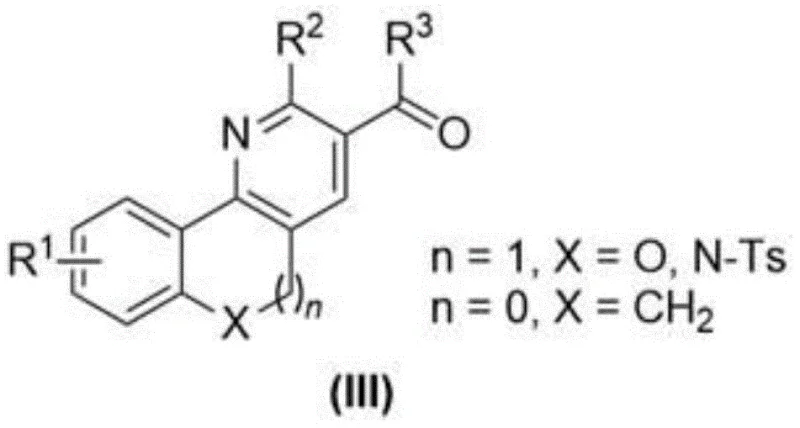
Mechanistic Insights into AgNO3-Catalyzed Radical Cyclization
The core of this technological advancement lies in the sophisticated mechanistic pathway driven by the silver nitrate and potassium persulfate catalytic system, which facilitates a unique radical cascade reaction. The process initiates with the generation of radical species from the 1,3-dicarbonyl compound, which then undergoes a selective addition to the alkene moiety of the o-allyloxybenzaldehyde derivative. This radical addition is critical as it constructs the carbon-carbon bonds necessary for the polycyclic framework while maintaining the integrity of the aldehyde functionality for the subsequent cyclization step. The mild temperature range of 40-50°C is crucial here, as it provides sufficient energy to drive the radical propagation without triggering unwanted side reactions or decomposition of sensitive functional groups. This precise control over reaction kinetics ensures that the intermediate 1,5-dicarbonyl structure is formed with high fidelity, setting the stage for the final ring-closing event.
Following the initial radical coupling, the reaction mixture undergoes a cyclization phase upon the addition of ammonium acetate and acetic acid at elevated temperatures of 120°C. This second step involves a condensation reaction where the aldehyde group reacts with the ammonium source to form the pyridine ring, completing the polycyclic architecture. The beauty of this mechanism is its tolerance to various substituents, as indicated by the general formula where R1 can be fluorine, chlorine, bromine, or methyl, allowing for significant structural diversity. This mechanistic robustness is essential for impurity control, as the specific reaction conditions minimize the formation of by-products that often plague traditional acid-catalyzed cyclizations. For R&D directors, understanding this mechanism highlights the potential for developing a wide range of analogues with consistent quality, ensuring that the final high-purity pharmaceutical intermediates meet stringent regulatory specifications.
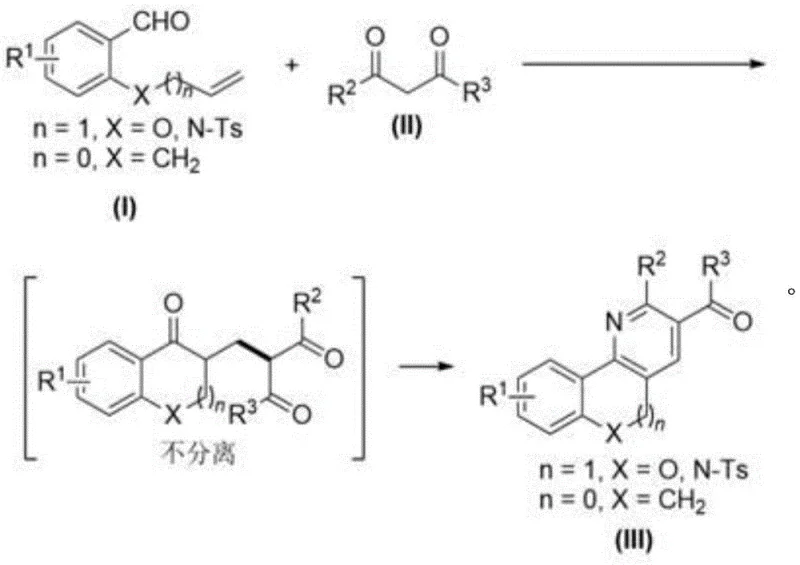
How to Synthesize Polycyclic Pyridine Compounds Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to the specific stoichiometric ratios and thermal profiles outlined in the patent data to ensure optimal results. The process begins by charging a reaction vessel with the o-allyloxybenzaldehyde derivative and the 1,3-dicarbonyl compound in DMSO, followed by the precise addition of the silver nitrate catalyst and potassium persulfate oxidant. Maintaining the temperature strictly between 40-50°C for a duration of 24-30 hours is critical to allow the radical coupling to reach completion without degrading the reactants. Once the initial coupling is finished, the crude mixture is concentrated and directly subjected to the cyclization conditions without purification, which is a key operational advantage of this method. The detailed standardized synthesis steps see the guide below for specific molar ratios and workup procedures that guarantee reproducibility.
- Mix o-allyloxybenzaldehyde derivatives and 1,3-dicarbonyl compounds in DMSO with AgNO3 and K2S2O8 at 40-50°C.
- Concentrate the crude reaction mixture without intermediate purification to retain the 1,5-dicarbonyl structure.
- Add ammonium acetate and acetic acid, then reflux at 120°C for 3 hours to cyclize into the final polycyclic pyridine.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented synthesis method offers profound advantages for procurement managers and supply chain heads looking to optimize their sourcing strategies for complex heterocyclic intermediates. The elimination of the intermediate isolation step fundamentally alters the cost structure of the manufacturing process, leading to significantly reduced operational expenditures associated with solvent procurement, waste disposal, and labor hours. By utilizing readily available and inexpensive raw materials such as silver nitrate and common aldehydes, the process mitigates the risk of supply chain disruptions often caused by reliance on exotic or proprietary reagents. Furthermore, the mild reaction conditions contribute to enhanced safety profiles in the manufacturing facility, reducing the energy consumption required for heating and cooling compared to traditional high-temperature processes. These factors collectively contribute to a more resilient and cost-effective supply chain, ensuring consistent availability of high-purity polycyclic pyridine compounds for downstream drug development.
- Cost Reduction in Manufacturing: The one-pot nature of this synthesis eliminates the need for intermediate purification, which drastically reduces solvent consumption and waste generation, leading to substantial cost savings in raw material and disposal expenses. By avoiding the isolation of the unstable 1,5-dicarbonyl intermediate, the process minimizes material loss and labor costs associated with multiple workup steps. The use of cheap and abundant catalysts like silver nitrate further lowers the reagent cost profile compared to precious metal catalysts often used in similar transformations. This economic efficiency allows for more competitive pricing structures without compromising on the quality or purity of the final pharmaceutical intermediate product.
- Enhanced Supply Chain Reliability: The reliance on commercially available starting materials such as o-allyloxybenzaldehyde derivatives and 1,3-dicarbonyl compounds ensures a stable and secure supply chain that is less susceptible to market volatility. The robustness of the reaction conditions means that production can be scaled up with minimal risk of batch failure, ensuring consistent lead times for customers requiring large quantities of material. Additionally, the simplified process flow reduces the complexity of manufacturing scheduling, allowing for faster turnaround times from order to delivery. This reliability is crucial for pharmaceutical clients who depend on uninterrupted supply of key intermediates to maintain their own production timelines and regulatory filings.
- Scalability and Environmental Compliance: The mild thermal conditions and reduced solvent usage align perfectly with modern green chemistry principles, making the process easier to scale while meeting stringent environmental regulations. The absence of harsh reagents and the minimization of waste streams simplify the effluent treatment process, reducing the environmental burden and compliance costs for the manufacturing facility. This scalability ensures that the transition from laboratory gram-scale to commercial ton-scale production can be achieved smoothly without significant re-engineering of the process. Consequently, this method supports the commercial scale-up of complex pharmaceutical intermediates while maintaining a sustainable and eco-friendly manufacturing footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this novel polycyclic pyridine synthesis technology. These answers are derived directly from the technical specifications and beneficial effects described in the patent documentation to provide clarity for potential partners. Understanding these details is essential for evaluating the feasibility of integrating this route into existing manufacturing portfolios or sourcing strategies.
Q: What are the key advantages of the AgNO3 catalyzed route over traditional methods?
A: The novel method avoids the complex synthesis and separation of 1,5-dicarbonyl precursors required in traditional routes, utilizing a direct one-pot radical coupling strategy that significantly simplifies the workflow.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the process utilizes readily available raw materials and operates under mild temperature conditions (40-50°C for the first step), which enhances safety and scalability for industrial manufacturing.
Q: What is the purity profile of the resulting polycyclic pyridine compounds?
A: The method employs a specific cyclization step with ammonium acetate and acetic acid followed by column chromatography, ensuring high structural integrity and minimizing impurity formation common in harsher condensation reactions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polycyclic Pyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of advanced synthetic methodologies in driving the next generation of pharmaceutical innovations, and we are uniquely positioned to leverage this patented technology for our clients. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to industrial manufacturing is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of polycyclic pyridine compound meets the highest standards of quality and consistency required by global regulatory bodies. Our commitment to technical excellence allows us to offer a reliable polycyclic pyridine supplier partnership that combines cutting-edge chemistry with robust manufacturing capabilities.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project requirements and cost targets. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this streamlined manufacturing process for your supply chain. We encourage you to contact us to obtain specific COA data and route feasibility assessments that demonstrate the viability of this technology for your commercial needs. Let us collaborate to optimize your intermediate sourcing strategy and accelerate your drug development timelines with our superior chemical solutions.
