Scalable Asymmetric Synthesis of (S,S)-2,8-Diazabicyclo[4,3,0]nonane for Commercial API Production
The pharmaceutical industry continuously seeks robust pathways for generating high-value chiral building blocks, particularly for fourth-generation quinolone antibiotics like Moxifloxacin. Patent CN110317201B introduces a groundbreaking asymmetric synthesis method for (S,S)-2,8-diazabicyclo[4,3,0]nonane, a critical chiral intermediate that dictates the biological efficacy of the final drug substance. This technology represents a significant paradigm shift from traditional resolution-based approaches to direct asymmetric organocatalysis, utilizing a chiral squaramide catalyst to drive a highly stereoselective [2+4] cycloaddition. For R&D directors and procurement strategists, this patent offers a compelling solution to the longstanding challenges of yield loss and waste generation associated with racemic synthesis. By leveraging bifunctional catalysis, the process achieves exceptional optical purity and operational simplicity, positioning it as a superior candidate for reliable pharmaceutical intermediate supplier networks aiming to optimize their API supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of (S,S)-2,8-diazabicyclo[4,3,0]nonane has relied heavily on resolution techniques or multi-step sequences starting from pyridine derivatives, as documented in prior art such as WO9415938 and various enzymatic resolution studies. These conventional methodologies suffer from inherent thermodynamic limitations, specifically the maximum theoretical yield of 50% for the desired enantiomer when starting from racemic mixtures. This inefficiency necessitates the disposal or complex recycling of the unwanted (R,R) isomer, leading to substantial increases in Process Mass Intensity (PMI) and waste treatment costs. Furthermore, many existing routes involve harsh reaction conditions, expensive chiral resolving agents like D-mandelic acid, or lengthy synthetic sequences that accumulate impurities and reduce overall throughput. The environmental burden and economic inefficiency of discarding half the synthesized material make these legacy processes increasingly untenable for modern, sustainability-focused chemical manufacturing.
The Novel Approach
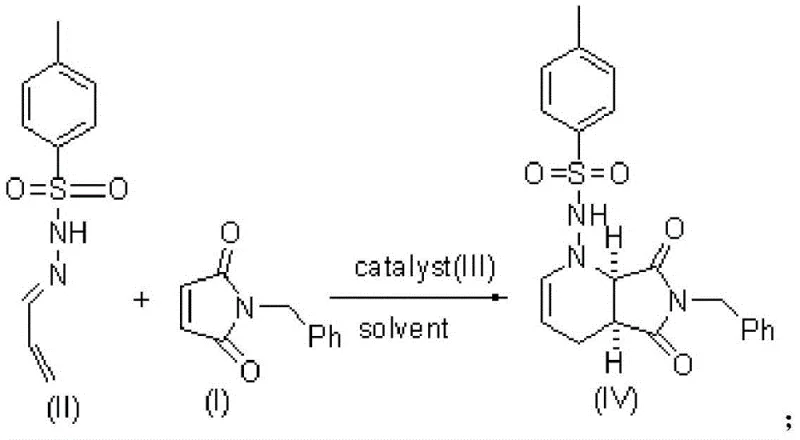
In stark contrast, the novel approach detailed in CN110317201B employs a direct asymmetric [2+4] cycloaddition between N-benzyl-maleimide and an N-p-toluenesulfonyl-N'-propenylidenehydrazine derivative. This strategy bypasses the need for resolution entirely by constructing the chiral centers directly during the ring-forming step. The process utilizes a chiral squaramide catalyst to induce high stereoselectivity, achieving crude enantiomeric excess values exceeding 90%, which can be further upgraded to over 99% ee through straightforward recrystallization. This route not only dramatically improves the total yield to above 60% across four steps but also simplifies the purification workflow. By eliminating the resolution step, the method significantly reduces solvent consumption and raw material usage, offering a streamlined pathway that is far more amenable to cost reduction in API manufacturing compared to traditional resolution-based protocols.
![Overall reaction scheme showing the four-step synthesis from N-benzyl-maleimide to (S,S)-2,8-diazabicyclo[4,3,0]nonane](/insights/img/s-s-2-8-diazabicyclo-nonane-synthesis-supplier-20260308093030-07.webp)
Mechanistic Insights into Squaramide-Catalyzed Asymmetric Cycloaddition
The core innovation of this synthesis lies in the sophisticated mechanism of the chiral squaramide catalyst, which operates through a dual-activation mode typical of organocatalysis. The catalyst functions as a bifunctional entity, simultaneously activating both the electrophilic maleimide and the nucleophilic hydrazine species through a network of hydrogen bonds. Specifically, the squaramide moiety acts as a double hydrogen bond donor to activate the carbonyl groups of the maleimide, while the basic tertiary amine group within the catalyst framework deprotonates or coordinates with the hydrazine nitrogen. This precise spatial arrangement creates a rigid chiral environment that directs the approach of the reactants, ensuring that the [2+4] cycloaddition occurs exclusively to form the desired (1S,6R) configuration. This level of stereocontrol is critical for preventing the formation of diastereomeric impurities that are notoriously difficult to separate in later stages of synthesis.
Furthermore, the robustness of this catalytic system allows for excellent impurity control throughout the synthetic sequence. The high selectivity in the initial step means that downstream processing does not need to account for significant amounts of the wrong enantiomer, thereby simplifying the crystallization and filtration steps. The subsequent reduction and hydrolysis steps proceed with high fidelity, preserving the stereochemical integrity established in the first step. For quality assurance teams, this implies a much cleaner impurity profile in the final active pharmaceutical ingredient (API). The ability to tune the catalyst structure, as shown by variants IIIa, IIIb, and IIIc, allows for further optimization of reaction kinetics and selectivity, providing a versatile platform for adapting the process to different scales or specific purity requirements without compromising the fundamental mechanistic advantages of the squaramide scaffold.
How to Synthesize (S,S)-2,8-Diazabicyclo[4,3,0]nonane Efficiently
The synthesis protocol outlined in the patent provides a clear, four-step roadmap for producing high-purity (S,S)-2,8-diazabicyclo[4,3,0]nonane suitable for commercial applications. The process begins with the critical asymmetric cycloaddition in a controlled temperature range of -20°C to 50°C, followed by a reduction step using hydride sources like LAH or NaBH4. Subsequent acid-catalyzed hydrolysis removes the protecting group, and a final hydrogenation step yields the target diamine. This sequence is designed to maximize yield while minimizing operational complexity, making it an ideal candidate for technology transfer.
- Perform asymmetric [2+4] addition of N-benzyl-maleimide and hydrazine derivative using chiral squaramide catalyst at -20 to 50°C.
- Reduce the resulting dioxo-intermediate using Lithium Aluminum Hydride or Sodium Borohydride systems.
- Execute acid-catalyzed hydrolysis to remove the p-toluenesulfonyl protecting group.
- Finalize via catalytic hydrogenation and debenzylation to obtain the target diamine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this asymmetric synthesis route offers transformative benefits regarding cost structure and supply security. The elimination of the resolution step fundamentally alters the economics of production by removing the 50% yield penalty associated with discarding unwanted enantiomers. This improvement in atom economy translates directly into lower raw material costs per kilogram of finished product. Additionally, the use of readily available starting materials like N-benzyl-maleimide reduces dependency on specialized, high-cost chiral pools. The mild reaction conditions, which do not require cryogenic temperatures below -20°C or extreme pressures, further contribute to cost reduction in API manufacturing by lowering energy consumption and allowing the use of standard stainless steel reactor equipment rather than specialized Hastelloy vessels.
- Cost Reduction in Manufacturing: The primary economic driver of this technology is the drastic improvement in overall yield compared to resolution methods. By avoiding the loss of half the material stream, the process effectively doubles the output from the same amount of input precursors. This efficiency gain is compounded by the simplified purification steps; since the crude product already possesses high optical purity (>90% ee), the need for expensive chiral chromatography or multiple recrystallizations is minimized. Consequently, the total cost of goods sold (COGS) is significantly reduced, allowing for more competitive pricing in the global market for Moxifloxacin intermediates without sacrificing margin.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the scarcity of chiral resolving agents or the bottlenecks associated with recycling unwanted isomers. This new method mitigates those risks by relying on achiral, commodity-grade starting materials that are widely available from multiple global suppliers. The robustness of the squaramide catalyst, which can be used in relatively low loadings (0.05 to 1 equivalent), ensures that catalyst supply is not a limiting factor. Furthermore, the shortened synthetic route reduces the number of unit operations, thereby decreasing the probability of batch failures and ensuring more consistent lead times for high-purity intermediates, which is crucial for maintaining just-in-time inventory levels for downstream API manufacturers.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this process aligns perfectly with green chemistry principles. The reduction in waste generation—specifically the elimination of the unwanted enantiomer waste stream—lowers the burden on wastewater treatment facilities and reduces the carbon footprint of the manufacturing site. The solvents employed, such as dichloromethane, ethanol, and ethyl acetate, are well-understood and easily recoverable through distillation, facilitating solvent recycling loops. This environmental efficiency not only aids in meeting stringent regulatory compliance standards but also enhances the commercial scale-up of complex heterocycles by simplifying the permitting process for new production lines and reducing the costs associated with hazardous waste disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric synthesis technology. These insights are derived directly from the experimental data and process descriptions provided in the patent documentation, ensuring accuracy for technical evaluation. Understanding these details is essential for assessing the feasibility of integrating this route into existing manufacturing portfolios.
Q: What is the primary advantage of this synthesis over resolution methods?
A: Unlike traditional resolution methods which theoretically discard 50% of the material as the unwanted enantiomer, this asymmetric catalytic route utilizes chiral induction to directly synthesize the desired (S,S) configuration, significantly improving atom economy and reducing raw material costs.
Q: What level of enantiomeric excess (ee) can be achieved?
A: The process achieves an initial enantioselectivity of over 90% ee in the crude asymmetric addition step. Through a simple recrystallization procedure, the optical purity can be enhanced to greater than 99% ee, meeting stringent requirements for pharmaceutical intermediates.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly highlights the method's suitability for industrialization due to mild reaction conditions ranging from -20°C to 50°C, the use of common solvents like dichloromethane and ethanol, and a robust total yield exceeding 60% across four steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S,S)-2,8-Diazabicyclo[4,3,0]nonane Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient chiral synthesis in the modern pharmaceutical landscape. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes like the one described in CN110317201B can be successfully translated into robust industrial operations. Our state-of-the-art facilities are equipped to handle the specific requirements of organocatalytic reactions, including precise temperature control and inert atmosphere handling, while our rigorous QC labs guarantee that all batches meet stringent purity specifications required for GMP manufacturing. We are committed to delivering high-quality intermediates that support the global supply of life-saving antibiotics.
We invite pharmaceutical partners to collaborate with us to leverage this advanced synthesis technology for your Moxifloxacin supply chain. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this asymmetric route for your specific volume requirements. We encourage you to contact us today to discuss specific COA data, route feasibility assessments, and how we can tailor our production capabilities to meet your unique timeline and quality objectives, ensuring a secure and cost-effective supply of this critical chiral building block.
