Advanced One-Step Synthesis of Discotic Liquid Crystal Intermediates for Commercial Scale-Up
Advanced One-Step Synthesis of Discotic Liquid Crystal Intermediates for Commercial Scale-Up
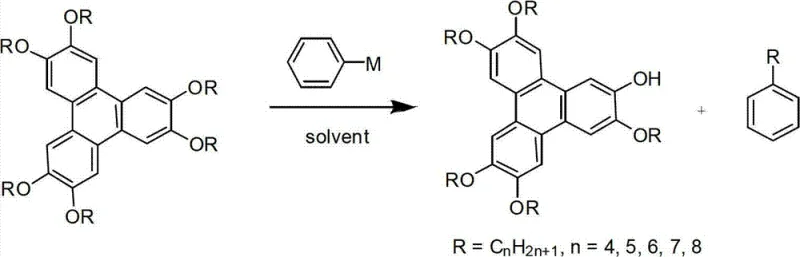
The rapid evolution of organic electronics demands robust synthetic pathways for high-performance materials, particularly discotic liquid crystals used in next-generation displays and photovoltaics. Patent CN102701921B introduces a groundbreaking methodology for the synthesis of 2-hydroxy-3,6,7,10,11-penta(alkoxy)triphenylene, a critical intermediate for columnar phase liquid crystals. This innovation addresses long-standing challenges in the field by employing a selective dealkylation strategy using phenyl metal reagents, which not only streamlines the production of the target molecule but also concurrently generates linear alkyl benzene as a valuable co-product. For R&D directors and procurement specialists seeking a reliable OLED material supplier, this technology represents a paradigm shift from low-yield, multi-step processes to a highly efficient, one-pot transformation that enhances both economic viability and supply chain stability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of mono-hydroxy triphenylene derivatives has been plagued by inefficient methodologies that hinder commercial adoption. Traditional routes, such as the partial alkylation of hexaacetoxytriphenylene, often result in abysmal total yields of less than 5%, accompanied by difficult-to-control alkylation patterns and a plethora of by-products that complicate purification. Alternative oxidative coupling methods using ferric chloride or molybdenum pentachloride require complex precursor synthesis via Suzuki or Ullman couplings, driving the total yield down to approximately 10% while generating significant hazardous waste. Furthermore, existing selective dealkylation techniques relying on bulky Lewis acids like 9-bromo-9-borabicyclo[3.3.1]nonane are prohibitively expensive, extremely sensitive to atmospheric conditions, and suffer from poor selectivity, making them unsuitable for the rigorous demands of industrial scale-up in the electronic chemical sector.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes phenyl metal reagents—such as phenyllithium or phenylmagnesium halides—as selective dealkylation agents to cleave a single alkyl chain from the hexaalkoxytriphenylene core. This ingenious strategy transforms a typically destructive process into a constructive synthesis, achieving high selectivity without the need for exotic catalysts. The reaction proceeds under standard reflux conditions in common organic solvents, offering a dramatic improvement in operational simplicity and safety. By converting the removed alkyl group into a linear alkyl benzene, the process effectively turns a waste stream into a revenue-generating co-product, thereby optimizing the atom economy and reducing the overall cost burden. This dual-output capability makes the method exceptionally attractive for manufacturers aiming for cost reduction in electronic chemical manufacturing.
Mechanistic Insights into Phenyl Metal-Mediated Selective Dealkylation
The core of this technological breakthrough lies in the unique reactivity of phenyl metal species towards the ether linkages of the triphenylene scaffold. Unlike traditional Lewis acids which coordinate broadly with oxygen atoms leading to random demethylation or decomposition, the phenyl anion acts as a potent nucleophile that selectively attacks the alkyl carbon of the ether bond. This nucleophilic substitution mechanism facilitates the cleavage of the C-O bond at a specific position, likely driven by steric and electronic factors inherent to the crowded triphenylene core. The use of a substantial excess of the phenyl metal reagent, typically in a molar ratio ranging from 1:10 to 1:20, ensures that the equilibrium is driven towards the formation of the mono-hydroxy product while suppressing potential side reactions such as nucleophilic attack on the aromatic ring itself. This precise control over the reaction trajectory is what enables the high purity levels observed in the final product.
From an impurity control perspective, the mechanism inherently limits the formation of poly-hydroxylated by-products which are common in acid-catalyzed hydrolysis. The reaction environment, maintained under inert gas protection, prevents the oxidation of sensitive organometallic intermediates, ensuring a clean reaction profile. Post-reaction workup involves a straightforward acid quench which protonates the phenoxide intermediate to yield the target hydroxy-triphenylene, while the displaced alkyl group combines with the phenyl moiety to form the stable linear alkyl benzene. This distinct separation of products based on polarity allows for efficient isolation via standard column chromatography and recrystallization, significantly reducing the burden on quality control laboratories and ensuring that the final material meets the stringent purity specifications required for high-mobility organic semiconductors.
How to Synthesize 2-Hydroxy-3,6,7,10,11-penta(alkoxy)triphenylene Efficiently
The implementation of this synthesis route requires careful attention to stoichiometry and reaction conditions to maximize the recovery of the valuable liquid crystal intermediate. The process begins with the dissolution of the hexaalkoxy precursor and the phenyl metal reagent in an anhydrous organic solvent such as tetrahydrofuran or diethyl ether, followed by a prolonged heating period to ensure complete conversion. Detailed standardized operating procedures regarding temperature gradients, addition rates, and quenching protocols are essential for maintaining batch-to-batch consistency. For technical teams looking to implement this chemistry, the following guide outlines the critical operational parameters derived from the patent examples to ensure optimal results.
- Dissolve 2,3,6,7,10,11-hexa(alkoxy)triphenylene and phenyl metal reagent in an organic solvent under inert gas protection at a molar ratio of 1: (10-20).
- Heat the mixture to perform a reflux reaction for 24 to 36 hours, then distill off the solvent to isolate the crude residue.
- Quench the residue in ice water, adjust pH to less than 2 with dilute hydrochloric acid, extract with dichloromethane, and purify via column chromatography and recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers profound strategic benefits beyond mere technical feasibility. The elimination of expensive and air-sensitive boron-based reagents drastically simplifies the raw material sourcing strategy, allowing for the utilization of commodity chemicals that are readily available in the global market. This shift reduces dependency on niche suppliers and mitigates the risk of supply disruptions caused by the limited production capacity of specialized catalysts. Furthermore, the ability to recover and recycle unreacted starting material significantly enhances the overall material efficiency of the process, ensuring that precious intermediates are not lost to waste streams. This circular approach to material usage aligns perfectly with modern sustainability goals and provides a buffer against volatility in raw material pricing.
- Cost Reduction in Manufacturing: The economic model of this process is strengthened by the co-production of linear alkyl benzene, a high-volume industrial chemical used in detergent manufacturing. This dual-output system effectively subsidizes the cost of producing the high-value liquid crystal intermediate, creating a natural hedge against market fluctuations. By avoiding the use of costly transition metal catalysts and complex purification steps associated with oxidative coupling, the overall operational expenditure is significantly lowered. The simplified workup procedure, which relies on standard extraction and crystallization techniques rather than preparative HPLC or complex distillation, further reduces utility consumption and labor costs, delivering substantial cost savings without compromising on product quality.
- Enhanced Supply Chain Reliability: The reliance on robust, well-established organometallic chemistry ensures that the manufacturing process is resilient to external shocks. Phenyl metal reagents and common ether solvents are produced by a wide network of chemical suppliers globally, ensuring a continuous and reliable supply chain. The process tolerance to standard industrial equipment, without the need for specialized corrosion-resistant reactors required for strong Lewis acids like HF or AlCl3, means that production can be easily transferred between different manufacturing sites. This flexibility is crucial for maintaining supply continuity for downstream clients in the display and semiconductor industries, where downtime can result in significant financial losses.
- Scalability and Environmental Compliance: Scaling this reaction from laboratory to commercial production is facilitated by the absence of highly toxic by-products and the use of manageable reaction exotherms. The waste profile is significantly cleaner compared to traditional methods that generate heavy metal sludge or halogenated waste, simplifying compliance with increasingly strict environmental regulations. The ability to recycle unreacted hexaalkoxytriphenylene minimizes the generation of solid waste, contributing to a greener manufacturing footprint. This environmental advantage not only reduces disposal costs but also enhances the brand reputation of the manufacturer as a responsible partner in the green electronics supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this selective dealkylation technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on performance metrics and operational requirements. Understanding these details is vital for stakeholders evaluating the feasibility of integrating this intermediate into their existing product portfolios or R&D pipelines.
Q: What are the primary advantages of using phenyl metal reagents over traditional Lewis acids for triphenylene dealkylation?
A: Unlike traditional Lewis acids such as boron tribromide which are highly sensitive to air and moisture, phenyl metal reagents offer superior selectivity and operational stability. This method avoids the formation of complex by-products and eliminates the need for hazardous quenching procedures associated with strong Lewis acids, resulting in a cleaner reaction profile and simplified downstream processing.
Q: How does this synthesis method impact the overall yield and purity of discotic liquid crystal intermediates?
A: The patented process achieves significantly higher yields compared to conventional oxidative coupling or partial alkylation methods, which often suffer from yields below 10%. By utilizing a selective dealkylation strategy, the reaction minimizes side reactions, allowing for the recovery of unreacted starting materials and ensuring the final product meets stringent purity specifications required for organic semiconductor applications.
Q: Can this process be scaled for industrial production of electronic chemicals?
A: Yes, the process is specifically designed for scalability. It utilizes common organic solvents and readily available phenyl metal reagents, avoiding exotic catalysts. Furthermore, the co-production of linear alkyl benzene adds economic value, making the large-scale manufacturing of these high-performance display materials more commercially viable and sustainable.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Hydroxy-3,6,7,10,11-penta(alkoxy)triphenylene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity intermediates play in the advancement of organic electronics and display technologies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering materials that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 2-hydroxy-3,6,7,10,11-penta(alkoxy)triphenylene performs consistently in your final device applications. Our infrastructure is designed to support the complex synthesis requirements of advanced functional materials while maintaining the highest standards of safety and quality assurance.
We invite you to collaborate with us to leverage this innovative synthetic route for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this efficient methodology can optimize your bill of materials. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in fine chemical synthesis can accelerate your time to market for next-generation electronic devices.
