Advanced Green Synthesis of Benzocyclohexanone Compounds for Scalable Pharmaceutical Manufacturing
Advanced Green Synthesis of Benzocyclohexanone Compounds for Scalable Pharmaceutical Manufacturing
The pharmaceutical industry is constantly seeking robust, environmentally sustainable, and cost-effective pathways for the production of critical intermediates. A significant breakthrough in this domain is detailed in Chinese Patent CN112094182A, which discloses a novel green synthesis method for benzocyclohexanone compounds. These structures serve as pivotal scaffolds in the design of numerous active pharmaceutical ingredients (APIs), playing a vital role in drug discovery and functional group modification. The patented methodology addresses long-standing challenges in organic synthesis by replacing hazardous homogeneous catalysts with a recyclable, metal-doped modified molecular sieve system. This innovation not only streamlines the operational workflow but also aligns with modern green chemistry principles, offering a compelling value proposition for R&D directors and procurement managers alike who are tasked with optimizing supply chains for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzocyclohexanone derivatives has relied heavily on classical Friedel-Crafts acylation strategies utilizing harsh reagents. Traditional protocols often employ polyphosphoric acid (PPA), phosphorus pentoxide, or Eaton's reagent to effect dehydration and cyclization. While effective in converting 4-phenylbutyric acid precursors, these methods necessitate the use of massive excesses of strong acids, creating severe safety hazards for operators and causing significant corrosion to reactor equipment. Furthermore, the post-treatment phase generates substantial quantities of highly acidic wastewater, posing a heavy burden on environmental compliance and waste management systems. Alternative approaches utilizing Lewis acids such as aluminum trichloride or tin tetrachloride mitigate some issues but introduce new complexities, including the formation of intractable colloidal solutions during workup and the requirement for stoichiometric or near-stoichiometric amounts of catalyst, which drastically increases raw material costs and complicates product isolation.
The Novel Approach
In stark contrast to these legacy methods, the technology described in Patent CN112094182A introduces a sophisticated two-step sequence that prioritizes efficiency and sustainability. The process begins with the activation of the 4-phenylbutyric acid starting material using oxalyl chloride in dichloromethane to generate the corresponding acid chloride intermediate in situ. This activated species is then subjected to an intramolecular cyclization catalyzed by a metal-doped modified molecular sieve, specifically Fe-Al-MCM-41. This heterogeneous catalytic system operates under mild conditions, typically between 0°C and 110°C, and allows for the catalyst to be recovered via simple filtration. The elimination of corrosive liquid acids and the ability to recycle the solid catalyst represent a paradigm shift in manufacturing efficiency. As illustrated in the general reaction scheme below, this method accommodates a wide variety of substituents, ensuring versatility for diverse drug synthesis programs.
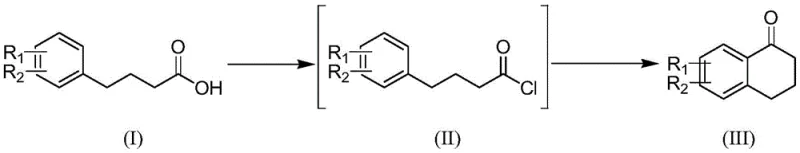
Mechanistic Insights into Fe-Al-MCM-41 Catalyzed Cyclization
The core of this technological advancement lies in the unique properties of the Fe-Al-MCM-41 molecular sieve catalyst. Unlike traditional homogeneous Lewis acids that dissolve into the reaction medium and require quenching, this solid acid catalyst provides accessible Lewis acidic sites on its surface and within its mesoporous structure. The iron doping enhances the Lewis acidity of the aluminum sites, facilitating the activation of the acyl chloride carbonyl group towards electrophilic attack by the aromatic ring. This intramolecular Friedel-Crafts acylation proceeds through a concerted mechanism where the proximity of the acyl group to the ortho-position of the phenyl ring drives the formation of the six-membered ketone ring. The mesoporous nature of the MCM-41 support ensures high surface area and efficient mass transfer, allowing the reaction to proceed to completion with high conversion rates even at moderate temperatures such as 60°C.
From an impurity control perspective, this heterogeneous system offers distinct advantages. The absence of water-sensitive homogeneous catalysts minimizes the risk of hydrolysis of the acid chloride intermediate back to the starting acid, a common side reaction in traditional methods. Moreover, the solid nature of the catalyst prevents the formation of emulsions or gels that often trap product during aqueous workups with AlCl3. The specific example shown below demonstrates the efficacy of this mechanism with a dimethyl-substituted substrate, achieving an exceptional isolated yield of 98.8%. Such high purity and yield are critical for reducing downstream purification costs and ensuring the quality of the final API intermediate.
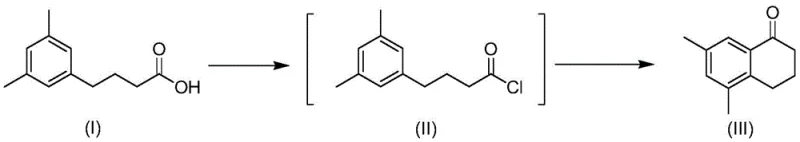
How to Synthesize Benzocyclohexanone Efficiently
Implementing this green synthesis route requires careful attention to reaction parameters to maximize the benefits of the molecular sieve catalyst. The process is designed to be operationally simple, avoiding the need for specialized high-pressure equipment or cryogenic conditions. The initial activation step with oxalyl chloride must be conducted under anhydrous conditions to prevent premature hydrolysis. Following the formation of the acid chloride, the choice of solvent for the cyclization step is crucial; toluene has been identified as the optimal medium, providing the right balance of solubility and boiling point for the 60°C reaction temperature. The standardized protocol ensures reproducibility across different batch sizes, making it an ideal candidate for technology transfer from laboratory to pilot plant.
- React 4-phenylbutyric acid derivative with oxalyl chloride in dichloromethane under reflux to form the crude acid chloride intermediate.
- Dissolve the crude acid chloride in toluene and add Fe-Al-MCM-41 molecular sieve catalyst to initiate the cyclization reaction.
- Stir the mixture at 60°C for 18 hours, then filter to recover the catalyst, evaporate solvent, and purify the residue via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this molecular sieve-catalyzed process offers tangible strategic benefits beyond mere technical elegance. The primary advantage lies in the drastic simplification of the workup procedure. By replacing corrosive liquid acids with a filterable solid catalyst, the process eliminates the need for complex neutralization steps and the associated generation of salt waste. This reduction in processing complexity translates directly into shorter cycle times and lower utility consumption, contributing to significant cost reduction in pharmaceutical intermediates manufacturing. Furthermore, the robustness of the catalyst system enhances supply chain reliability by reducing the dependency on volatile reagent markets for stoichiometric Lewis acids.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven primarily by catalyst recyclability and waste minimization. Since the Fe-Al-MCM-41 catalyst can be recovered, washed, and reused for multiple cycles without significant loss of activity, the effective cost per kilogram of catalyst consumed is drastically lowered compared to single-use aluminum trichloride. Additionally, the avoidance of strong mineral acids reduces the expenditure on corrosion-resistant equipment maintenance and hazardous waste disposal fees. The high yields observed across various substrates mean less raw material is wasted, further optimizing the cost of goods sold (COGS) for high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Operational simplicity is a key driver of supply security. The protocol's tolerance for a broad range of reaction temperatures and its use of common solvents like dichloromethane and toluene ensure that production is not bottlenecked by exotic reagent availability. The ability to filter the catalyst rather than perform an aqueous quench reduces the risk of batch failures due to emulsion formation or incomplete phase separation. This reliability allows for more accurate forecasting of lead times for high-purity pharmaceutical intermediates, enabling downstream API manufacturers to maintain leaner inventory levels without risking stockouts.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies environmental challenges, but this green synthesis method mitigates those risks inherently. The heterogeneous nature of the reaction facilitates easier scale-up, as heat transfer and mixing are more manageable without viscous acid slurries. From a regulatory standpoint, the reduction in E-factor (mass of waste per mass of product) supports corporate sustainability goals and simplifies compliance with increasingly stringent environmental regulations. The potential for commercial scale-up of complex pharmaceutical intermediates is thus greatly enhanced, offering a future-proof manufacturing solution.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis method is essential for stakeholders evaluating its adoption. The following questions address common inquiries regarding catalyst performance, substrate scope, and operational safety. These insights are derived directly from the experimental data provided in the patent documentation, ensuring that the information reflects the actual capabilities of the technology.
Q: What are the advantages of using metal-doped molecular sieves over traditional Lewis acids?
A: Metal-doped molecular sieves like Fe-Al-MCM-41 act as heterogeneous catalysts that can be easily filtered and recycled, eliminating the generation of large volumes of acidic wastewater and colloidal sludge associated with traditional AlCl3 or PPA methods.
Q: Can the catalyst be reused without significant loss of activity?
A: Yes, the patent data demonstrates that the Fe-Al-MCM-41 catalyst can be recovered by filtration, washed, and dried for reuse in subsequent batches while maintaining high catalytic activity and product yields over multiple cycles.
Q: What types of substituents are tolerated in this synthetic route?
A: The method exhibits broad substrate scope, successfully accommodating various substituents including alkyl groups (methyl, ethyl), alkoxy groups (methoxy), halogens (bromo), and cycloalkyl groups on the aromatic ring.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzocyclohexanone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the evolving demands of the global pharmaceutical market. Our team of expert chemists has extensively evaluated the green synthesis pathway described in Patent CN112094182A and confirmed its viability for large-scale production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to manufacturing plant is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of benzocyclohexanone intermediate meets the highest industry standards for impurity profiles and physical properties.
We invite potential partners to collaborate with us to leverage this innovative technology for their specific drug development programs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to reach out today to discuss specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific excellence and commercial reliability.
