Advanced Chiral Ferrocene P,N,N-Ligands for Scalable Pyrethroid Intermediate Manufacturing
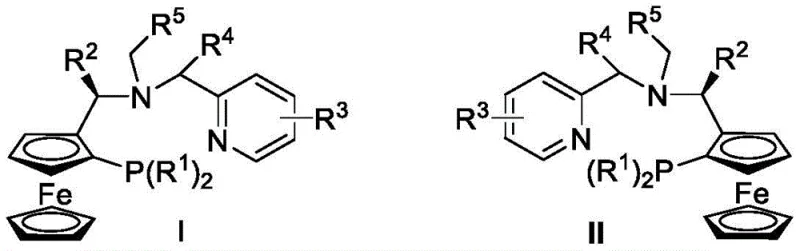
The landscape of asymmetric catalysis is undergoing a significant transformation driven by the need for more robust and versatile chiral ligands, as evidenced by the technological breakthroughs detailed in patent CN115873047A. This intellectual property introduces a novel class of chiral ferrocene P,N,N-ligands equipped with functional side arms, representing a strategic evolution from traditional rigid ligand architectures. The core innovation lies in the synergistic combination of a ferrocene backbone with a pyridine-nitrogen donor and a tunable side arm, which collectively create a highly defined chiral environment around the metal center. For R&D directors and process chemists, this development offers a compelling solution to the longstanding challenges of balancing catalytic activity with stereocontrol in complex molecular transformations. The patent explicitly highlights the utility of these ligands in copper-catalyzed asymmetric cyclopropanation, a reaction of paramount importance for constructing the three-membered rings found in numerous bioactive molecules.
Furthermore, the practical implications of this technology extend deeply into the supply chain strategies for agrochemical and pharmaceutical manufacturers. By enabling the efficient synthesis of chiral first chrysanthemic acid, a pivotal intermediate for pyrethroid insecticides, this ligand system addresses critical bottlenecks in the production of high-volume crop protection agents. The ability to achieve substantial yields and high optical purity under relatively mild conditions suggests a pathway toward more sustainable and cost-effective manufacturing processes. As a reliable agrochemical intermediate supplier, understanding the nuances of such enabling technologies is essential for maintaining a competitive edge in the global market. The following analysis dissects the mechanistic advantages, synthetic accessibility, and commercial viability of this novel ligand class.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the asymmetric cyclopropanation of olefins has relied heavily on ligands such as Schiff bases, hemicorrins, bisoxazolines, and bipyridines. While these traditional systems have served the industry well, they often suffer from inherent structural rigidity that limits their adaptability to diverse substrate scopes. In many industrial scenarios, conventional catalysts exhibit narrow substrate tolerance, requiring extensive optimization for each new olefin feedstock, which drastically increases development timelines and costs. Moreover, achieving high diastereoselectivity and enantioselectivity simultaneously often necessitates harsh reaction conditions or expensive metal precursors that complicate downstream purification and waste management. The inability to finely tune the steric and electronic properties of these rigid frameworks without complete redesign poses a significant barrier to process intensification and cost reduction in agrochemical intermediate manufacturing.
The Novel Approach
The methodology presented in the patent overcomes these limitations through the strategic design of a hybrid P,N,N-ligand featuring a dynamic side arm. Unlike static ligand systems, the incorporation of a side arm—such as a benzyl or chloromethyl oxazole group—introduces a secondary coordination sphere that can adapt to the transition state geometry. This structural flexibility allows the catalyst to accommodate a broader range of olefins, from simple styrenes to complex dienes like 2,5-dimethyl-2,4-hexadiene, while maintaining rigorous stereocontrol. The synthetic route to these ligands is notably streamlined, utilizing readily available ferrocenyl chiral amines and pyridine aldehydes. This accessibility translates directly into supply chain resilience, as the reliance on exotic or hard-to-source chiral building blocks is minimized. The result is a catalytic system that delivers high performance, exemplified by yields reaching 80% and enantioselectivity up to 90% for key pyrethroid precursors, making it a viable candidate for industrial adoption.
Mechanistic Insights into Copper-Catalyzed Asymmetric Cyclopropanation
The efficacy of the chiral ferrocene P,N,N-ligand stems from its ability to form a well-defined chiral copper carbene intermediate upon reaction with diazo compounds. In this catalytic cycle, the phosphorus atom of the ferrocene moiety and the nitrogen atom of the pyridine ring coordinate with the copper metal precursor, creating a chiral pocket. The side arm plays a critical role in shielding one face of the reactive carbene species, thereby directing the approach of the olefin substrate to favor the formation of a specific enantiomer. This steric differentiation is crucial for minimizing the formation of unwanted isomers, which are often difficult and costly to separate on a large scale. The patent data indicates that the catalyst system is robust enough to handle various diazoacetates, including methyl phenyldiazoacetate and ethyl diazoacetate, demonstrating versatility in reagent selection.
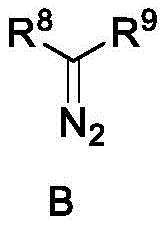
Impurity control is another significant advantage of this mechanistic pathway. The high diastereoselectivity observed, with dr values exceeding 19:1 in certain examples, implies that the formation of trans-isomers or other stereoisomeric byproducts is effectively suppressed. This high level of selectivity reduces the burden on purification units, such as crystallization or chromatography columns, leading to improved overall process mass intensity (PMI). For quality assurance teams, this means a cleaner impurity profile in the final high-purity chiral cyclopropanes, which is essential for meeting the stringent regulatory standards required for agrochemical active ingredients. The stability of the copper-ligand complex under the reaction conditions further ensures consistent performance over extended reaction times, mitigating the risk of catalyst decomposition which can lead to metal contamination in the product.
How to Synthesize Chiral Ferrocene P,N,N-Ligand Efficiently
The synthesis of these advanced ligands is designed for operational simplicity, avoiding the need for specialized equipment or extreme conditions. The process begins with a reductive amination step where a ferrocenyl chiral amine reacts with a pyridine aldehyde in methanol at room temperature. This is followed by the addition of sodium borohydride to reduce the intermediate imine to a stable amine. The second stage involves the installation of the side arm via an alkylation reaction in anhydrous acetonitrile using a base like potassium carbonate. This modular approach allows for the rapid generation of ligand libraries by simply varying the aldehyde or the alkylating agent. Detailed standardized synthesis steps for this protocol are provided in the guide below.
- React ferrocenyl chiral amine with pyridine aldehyde in methanol at room temperature, followed by reduction with NaBH4 to form the amine-pyridine intermediate.
- Dissolve the intermediate in anhydrous acetonitrile under nitrogen protection and react with a side-arm substance like benzyl bromide and dry K2CO3 under reflux.
- Filter the reaction mixture, concentrate, and purify the crude product via column chromatography to obtain the final chiral P,N,N-ligand.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the adoption of this ligand technology offers substantial strategic benefits centered around cost efficiency and supply security. The raw materials required for the ligand synthesis, such as ferrocenyl amines and pyridine derivatives, are commodity chemicals with established global supply chains, reducing the risk of sourcing bottlenecks. Furthermore, the reaction conditions employed—primarily room temperature stirring and standard reflux—are energy-efficient compared to processes requiring cryogenic cooling or high-pressure reactors. This reduction in energy consumption and equipment complexity contributes to a lower overall cost of goods sold (COGS) for the final intermediates. The elimination of expensive transition metals like rhodium or ruthenium in favor of abundant copper also represents a significant cost reduction in manufacturing, aligning with green chemistry principles.
Enhanced supply chain reliability is achieved through the robustness of the catalytic system. The ability to achieve high yields and selectivity across a range of substrates means that a single ligand platform can potentially service multiple product lines, simplifying inventory management. For supply chain heads, this versatility reduces the need to qualify and stock multiple specialized catalysts, thereby streamlining logistics. Additionally, the scalability of the process is supported by the use of common solvents like methanol, acetonitrile, and dichloroethane, which are easily handled in large-scale reactor trains. The commercial scale-up of complex pyrethroid intermediates is thus facilitated by a chemistry that is both forgiving and high-performing, ensuring consistent delivery schedules to downstream formulators.
Scalability and environmental compliance are further strengthened by the simplified workup procedures described in the patent. The reactions can be quenched with standard aqueous solutions and purified via conventional silica gel chromatography or crystallization, avoiding the generation of hazardous heavy metal waste streams associated with other catalytic systems. This ease of waste treatment lowers the environmental compliance burden and associated disposal costs. Moreover, the high atom economy of the cyclopropanation reaction, combined with the high selectivity of the ligand, minimizes the generation of organic waste, supporting corporate sustainability goals. Reducing lead time for high-purity chiral cyclopropanes becomes feasible when the synthesis is this direct and efficient, allowing manufacturers to respond more agilely to market demand fluctuations.
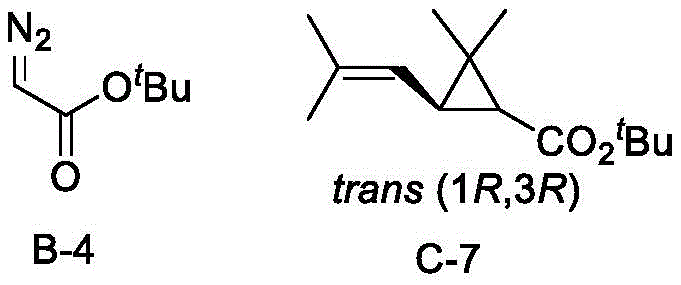
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral ligand technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making. Understanding these details is crucial for evaluating the feasibility of integrating this chemistry into existing production workflows.
Q: What level of enantioselectivity can be achieved with this ligand system?
A: The patent data demonstrates that the chiral copper catalyst generated in situ can achieve up to 95% enantiomeric excess (ee) and diastereomeric ratios greater than 20:1 depending on the specific olefin substrate used.
Q: Is this catalytic system suitable for industrial scale-up?
A: Yes, the synthesis utilizes mild reaction conditions such as room temperature reductive amination and standard reflux alkylation, avoiding cryogenic temperatures or ultra-high pressures, which facilitates commercial scale-up.
Q: What represents the primary application of this technology?
A: The primary application is the asymmetric synthesis of chiral cyclopropane structures, specifically serving as a key route for producing chiral first chrysanthemic acid, a critical intermediate for pyrethroid insecticides.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Ferrocene P,N,N-Ligand Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating advanced academic research into commercial reality, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is adept at optimizing the synthesis of complex chiral ligands and their subsequent application in catalytic processes to meet stringent purity specifications. We understand that the transition from bench-scale success to plant-scale production requires rigorous QC labs and a deep understanding of process safety and thermodynamics. Our infrastructure is designed to support the demanding requirements of chiral intermediate manufacturing, ensuring that every batch meets the high standards expected by global agrochemical and pharmaceutical partners.
We invite potential partners to engage with our technical procurement team to discuss how this technology can be tailored to your specific needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this copper-catalyzed system. We encourage you to contact us for specific COA data and route feasibility assessments to validate the performance of these ligands in your own process environments. Together, we can drive innovation and efficiency in the production of next-generation chiral intermediates.
