Advanced Pd-Catalyzed Synthesis of 1,4-Diene Scaffolds for Commercial Pharmaceutical Applications
Advanced Pd-Catalyzed Synthesis of 1,4-Diene Scaffolds for Commercial Pharmaceutical Applications
The development of efficient synthetic routes for 1,4-diene compounds represents a critical advancement in the field of organic chemistry, particularly for the construction of complex bioactive molecular skeletons. As detailed in the recent patent CN112047842A, a novel preparation method has been established that addresses significant limitations in traditional synthesis protocols. This technology leverages a palladium-catalyzed coupling strategy that directly utilizes allylic alcohols and olefins, bypassing the need for hazardous pre-activated substrates. For R&D directors and procurement specialists in the pharmaceutical sector, this innovation offers a pathway to high-purity 1,4-diene compounds with superior atom economy. The method is particularly relevant for synthesizing scaffolds found in potent mitochondrial complex I inhibitors like Piericidin A and other natural products such as Jerangolid, which require precise stereochemical control and high structural fidelity.
The strategic importance of this patent lies in its ability to streamline the supply chain for reliable pharmaceutical intermediate suppliers. By shifting away from multi-step activation processes, manufacturers can reduce the cumulative environmental footprint and operational complexity associated with producing these valuable intermediates. The reaction conditions described are robust, utilizing common solvents and commercially accessible catalysts, which ensures that the transition from laboratory scale to industrial production is seamless. This report analyzes the technical merits of this approach, highlighting its potential to drive cost reduction in fine chemical manufacturing while maintaining the rigorous quality standards required for drug substance production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,4-diene frameworks has relied heavily on the use of highly reactive allylic substrates, such as allylic halides, carbonates, or acetates, which must be prepared in separate preliminary steps. These conventional pathways often suffer from poor atom economy because the activation of the hydroxyl group generates stoichiometric amounts of salt waste or toxic by-products that require extensive downstream processing to remove. Furthermore, the use of strong bases or harsh activating agents can lead to side reactions, including polymerization or isomerization, which compromise the purity of the final product. For supply chain managers, these inefficiencies translate into higher raw material costs, longer lead times, and increased burden on waste treatment facilities. The reliance on sensitive intermediates also introduces stability issues during storage and transport, creating bottlenecks in the commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN112047842A introduces a direct coupling strategy that utilizes unactivated allylic alcohols as the primary nucleophilic partners. This breakthrough eliminates the pre-activation step entirely, meaning that the only by-product generated during the reaction is water, aligning perfectly with green chemistry principles. The process employs a specific catalytic system comprising tetrakis(triphenylphosphine)palladium alongside calcium bisimide and potassium hexafluorophosphate additives, which work synergistically to activate the C-OH bond under mild thermal conditions. As illustrated in the reaction scheme below, this approach allows for the efficient coupling of diverse allylic alcohols with electron-deficient olefins to yield 1,4-diene structures with high stereoselectivity.
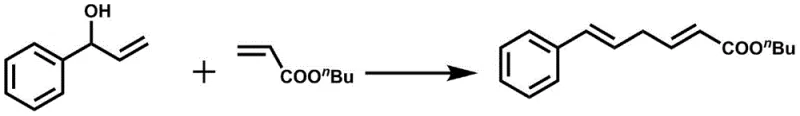
The operational simplicity of this novel approach cannot be overstated; reactions proceed smoothly in common solvents like ethylene glycol dimethyl ether or tetrahydrofuran at temperatures between 90°C and 110°C. This robustness significantly lowers the barrier for entry for manufacturers looking to integrate these scaffolds into their pipelines. By removing the need for hazardous activating reagents and simplifying the workup procedure to basic solvent removal and chromatography, the method offers a streamlined route that enhances overall process safety and efficiency. This directly supports the goal of reducing lead time for high-purity 1,4-diene compounds in a competitive market environment.
Mechanistic Insights into Pd-Catalyzed Allylic Substitution
The core of this technological advancement lies in the unique catalytic cycle facilitated by the palladium complex and the specific additive package. Mechanistically, the reaction initiates with the oxidative addition of the Pd(0) species into the allylic C-OH bond, a step that is typically energetically unfavorable without assistance. The presence of calcium bisimide and potassium hexafluorophosphate is believed to coordinate with the hydroxyl group, effectively increasing its leaving group ability and stabilizing the resulting pi-allyl palladium intermediate. This activation allows the reaction to proceed without the need for converting the alcohol into a better leaving group beforehand. Following the formation of the pi-allyl complex, the olefin substrate undergoes migratory insertion, followed by beta-hydride elimination to release the conjugated diene product and regenerate the active Pd(0) catalyst. This cycle is highly efficient, as evidenced by the low catalyst loading (0.006 to 0.4 mmol relative to substrate) required to achieve high conversions.
From an impurity control perspective, this mechanism offers distinct advantages over traditional acid- or base-promoted couplings. The mild conditions minimize the risk of double-bond isomerization, ensuring that the desired (E,E)-stereochemistry is preserved, which is critical for the biological activity of the final drug candidates. Furthermore, the absence of strong acids or bases reduces the formation of polymeric by-products that often plague diene syntheses. The specificity of the catalyst system ensures that functional groups such as esters, nitriles, and even sensitive heterocycles remain intact during the transformation. This high level of chemoselectivity simplifies the purification process, allowing for the isolation of products with purity levels suitable for subsequent biological testing or further synthetic elaboration without extensive recrystallization or distillation steps.
How to Synthesize 1,4-Diene Compounds Efficiently
The practical implementation of this synthesis route is designed to be accessible for both laboratory research and pilot-scale production. The protocol involves a straightforward one-pot procedure where all reagents are combined sequentially in a reaction vessel under an inert atmosphere. The specific molar ratios of allyl alcohol to olefin are optimized to drive the equilibrium towards the product, typically using a slight excess of the olefin component. The reaction temperature is maintained strictly within the 90°C to 110°C range to balance reaction rate with thermal stability of the catalyst. Upon completion, monitored by TLC, the workup is remarkably simple, involving only solvent evaporation and standard chromatographic purification. Detailed standardized synthesis steps for replicating this high-efficiency process are provided in the guide below.
- Combine allyl alcohol, olefin, Pd(PPh3)4 catalyst, calcium bisimide, and potassium hexafluorophosphate in a reaction solvent such as ethylene glycol dimethyl ether.
- Stir the reaction mixture under an inert argon atmosphere at temperatures between 90°C and 110°C for a duration of 12 to 24 hours.
- Remove the solvent via rotary evaporation and purify the crude residue using thin-layer chromatography with a petroleum ether and ethyl acetate system.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic methodology presents a compelling value proposition centered on cost efficiency and operational reliability. The primary driver of cost reduction is the elimination of the substrate activation step, which traditionally requires additional reagents, solvents, and processing time. By purchasing readily available allylic alcohols and acrylates directly, companies can significantly lower their raw material expenditure and reduce the inventory complexity associated with storing unstable intermediates. Additionally, the generation of water as the sole by-product drastically simplifies waste management protocols, leading to substantial savings in environmental compliance and disposal costs. This green profile not only reduces the financial burden but also enhances the corporate sustainability metrics of the manufacturing entity.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived from the streamlined reaction design which removes the need for expensive activating agents like thionyl chloride or carbonyl diimidazole. By condensing what was previously a two-step sequence into a single catalytic event, labor costs and energy consumption are inherently reduced. The use of earth-abundant additives like calcium salts further keeps reagent costs low compared to specialized ligands or exotic metals. This efficiency translates directly into a more competitive pricing structure for the final 1,4-diene intermediates, allowing buyers to optimize their bill of materials without sacrificing quality.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as cinnamyl alcohol, acrylic esters, and common solvents ensures a stable and resilient supply chain. Unlike proprietary reagents that may be subject to shortages or long lead times, the inputs for this reaction are widely produced by multiple global vendors. This diversification of supply sources mitigates the risk of production stoppages due to raw material scarcity. Furthermore, the robustness of the reaction conditions means that the process is less sensitive to minor variations in reagent quality, providing a buffer against supply chain fluctuations and ensuring consistent delivery schedules for downstream customers.
- Scalability and Environmental Compliance: Scaling this reaction from gram to kilogram quantities is facilitated by the use of standard heating and stirring equipment, with no requirement for high-pressure reactors or cryogenic conditions. The benign nature of the by-products means that effluent treatment is straightforward, helping facilities meet increasingly stringent environmental regulations. The high atom economy of the transformation ensures that maximum value is extracted from every kilogram of input material, minimizing waste generation per unit of product. This scalability makes the technology ideal for meeting the growing demand for complex diene scaffolds in the pharmaceutical and agrochemical sectors.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented synthesis method. These answers are derived directly from the experimental data and specifications outlined in the patent documentation to provide clarity for technical teams evaluating this route. Understanding these nuances is essential for successful technology transfer and process optimization.
Q: What are the primary advantages of using allylic alcohols directly in this synthesis?
A: Direct use of allylic alcohols eliminates the need for pre-activation steps such as converting alcohols to halides or carbonates, significantly improving atom economy and reducing waste generation to only water.
Q: Which catalysts and additives are essential for this transformation?
A: The process relies on tetrakis(triphenylphosphine)palladium as the catalyst, supported by calcium bisimide and potassium hexafluorophosphate as critical additives to facilitate the activation of the hydroxyl leaving group.
Q: Is this method suitable for large-scale pharmaceutical intermediate production?
A: Yes, the method utilizes commercially available raw materials like cinnamyl alcohol and common solvents, operates under relatively mild thermal conditions, and generates non-toxic by-products, making it highly scalable.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,4-Diene Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic strategies described in patent CN112047842A for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess the technical expertise and infrastructure to adapt these innovative catalytic protocols to meet your specific project requirements. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to full-scale manufacturing is executed with precision. We maintain stringent purity specifications across all our operations, supported by rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify the structural integrity and stereochemical purity of every batch of 1,4-diene compounds we produce.
We invite you to collaborate with us to leverage this green and efficient synthesis technology for your next drug development program. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating how this route can optimize your overall production budget. Please contact our technical procurement team today to request specific COA data for our existing diene inventory or to discuss route feasibility assessments for your custom synthesis projects. Let us help you secure a reliable supply of critical intermediates while advancing your sustainability goals.
