Revolutionizing Phosphoryl Azo Synthesis: A Mild, Scalable Route for High-Value Intermediates
The landscape of fine chemical manufacturing is undergoing a significant transformation driven by the urgent need for safer, more sustainable, and cost-effective synthetic methodologies. A pivotal advancement in this domain is detailed in patent CN112028933B, which discloses a novel, highly efficient method for preparing phosphoryl azo compounds. These versatile intermediates are critical building blocks in the synthesis of organic dyes, pharmaceutical agents, polymer additives, and agrochemicals. Traditionally, the synthesis of such P-N bonded structures has been plagued by the use of corrosive reagents and苛刻 reaction conditions. However, this patented technology introduces a groundbreaking approach utilizing aryldiazonium tetrafluoroborate salts and P(O)-H compounds catalyzed by simple pyridine bases. By shifting the paradigm from hazardous phosphorus halides to stable diazonium salts, this innovation not only enhances operational safety but also dramatically improves the selectivity and yield of the target molecules, positioning it as a cornerstone technology for modern reliable phosphoryl azo compound supplier networks aiming to serve high-end pharmaceutical and material science sectors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the phosphorus-nitrogen double bond or P-N single bond frameworks found in phosphoryl azo derivatives has relied heavily on the reaction of anilines with phosphorus oxyhalides in the presence of alkyl nitrites and hydrochloric acid at low temperatures. This conventional pathway presents severe logistical and safety challenges for industrial scale-up. The requirement for strict temperature control often necessitates energy-intensive cryogenic cooling systems, while the use of phosphorus oxyhalides introduces significant corrosion risks to reactor vessels and piping infrastructure. Furthermore, these traditional reagents are notoriously air-sensitive and moisture-sensitive, demanding inert atmosphere operations that complicate process engineering and increase capital expenditure. The atom economy of such routes is frequently poor, generating substantial amounts of inorganic salt waste and acidic by-products that require complex downstream separation and neutralization processes, thereby inflating the overall environmental footprint and production costs associated with cost reduction in fine chemical manufacturing.
The Novel Approach
In stark contrast, the methodology outlined in the patent data leverages the stability and reactivity of aryldiazonium tetrafluoroborates coupled with P(O)-H compounds under remarkably mild conditions. This new route operates effectively at room temperature and, crucially, tolerates the presence of air, eliminating the need for expensive inert gas purging and specialized Schlenk line techniques typically required for sensitive organometallic chemistry. The use of pyridine as a readily available, inexpensive organic base simplifies the catalyst system, removing the dependency on transition metals that often leave toxic residues in the final product. This shift allows for a streamlined workflow where substrates with diverse functional groups—including halogens, ethers, and alkyl chains—can be coupled with high fidelity. The result is a process that delivers target products with selectivity approaching 100% and excellent yields, fundamentally resolving the purity and stability issues that have long hindered the domestic production of high-end phosphoryl azo compounds.
Mechanistic Insights into Pyridine-Catalyzed P-N Bond Formation
The core of this technological breakthrough lies in the elegant simplicity of its base-catalyzed mechanism, which facilitates the nucleophilic attack of the phosphorus center onto the diazonium nitrogen. In this catalytic cycle, pyridine acts as a proton shuttle, deprotonating the P(O)-H compound to generate a highly nucleophilic phosphorus anion species in situ. This activated phosphorus intermediate then attacks the electrophilic terminal nitrogen of the aryldiazonium cation. The stability of the tetrafluoroborate counter-ion plays a crucial role here, preventing premature decomposition of the diazonium salt while allowing for a controlled release of nitrogen gas or stabilization of the transition state leading to the azo linkage. Unlike radical pathways that often lead to indiscriminate side reactions and complex impurity profiles, this ionic mechanism ensures a clean conversion path. The reaction proceeds smoothly in chlorobenzene, a solvent chosen for its ability to dissolve both organic substrates while maintaining thermal stability throughout the 12-hour reaction window, ensuring consistent kinetics across different batch sizes.
From an impurity control perspective, this mechanism offers distinct advantages for R&D teams focused on high-purity phosphoryl azo intermediates. Because the reaction avoids the generation of strong acids like HCl during the coupling step, acid-sensitive functional groups on the aromatic rings remain intact, preventing degradation or rearrangement side products. The high selectivity observed, often nearing quantitative conversion, implies that the formation of homocoupling by-products or reduced phosphine oxides is minimized. This inherent cleanliness of the reaction profile significantly reduces the burden on downstream purification units. For process chemists, this means that standard column chromatography or even recrystallization techniques are sufficient to achieve pharmaceutical-grade purity, bypassing the need for preparative HPLC or complex distillation setups that would otherwise erode profit margins in commercial production scenarios.
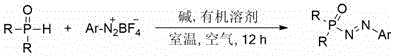
How to Synthesize Phosphoryl Azo Compounds Efficiently
The practical implementation of this synthesis route is designed for ease of adoption in both laboratory and pilot plant settings. The protocol involves simply combining the stoichiometric amounts of the aryldiazonium tetrafluoroborate, the specific P(O)-H substrate, and pyridine in chlorobenzene. The mixture is then stirred at ambient temperature for a fixed duration, typically around 12 hours, allowing the reaction to reach completion without external heating or cooling. This operational simplicity is a key factor in its potential for widespread industrial adoption. For detailed standard operating procedures and specific molar ratios optimized for different substrates, please refer to the standardized synthesis steps provided below.
- Mix aryldiazonium tetrafluoroborate, P(O)-H compound, pyridine base, and chlorobenzene solvent in a reaction vessel under air atmosphere.
- Stir the reaction mixture at room temperature for approximately 12 hours to ensure complete conversion.
- Purify the resulting phosphoryl azo compound using column chromatography to achieve high purity specifications.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel synthetic route represents a strategic opportunity to optimize sourcing strategies and mitigate risk. The elimination of hazardous phosphorus oxyhalides and the move towards stable, shelf-stable diazonium salts drastically simplifies raw material logistics. Suppliers no longer need to manage the complex storage and transportation requirements associated with corrosive liquids or air-sensitive reagents. This shift inherently reduces the risk of supply disruptions caused by regulatory changes on hazardous chemical transport or accidents during shipping. Furthermore, the use of commodity chemicals like pyridine and chlorobenzene ensures that the supply chain remains robust and less susceptible to the volatility seen with specialized catalysts or rare earth metals.
- Cost Reduction in Manufacturing: The economic implications of this technology are profound, primarily driven by the simplification of the process infrastructure. By removing the need for cryogenic cooling systems and corrosion-resistant alloy reactors, capital expenditure for new production lines can be significantly lowered. Additionally, the high atom economy and near-quantitative selectivity mean that raw material waste is minimized, directly improving the yield per kilogram of input. The absence of heavy metal catalysts also eliminates the costly and time-consuming steps required for metal scavenging and residual metal testing, which are mandatory for pharmaceutical grade materials. These factors combine to create a leaner manufacturing process that offers substantial cost savings without compromising on quality standards.
- Enhanced Supply Chain Reliability: The reliance on stable solid reagents like aryldiazonium tetrafluoroborates enhances the predictability of the supply chain. Unlike liquid reagents that may degrade over time or require cold chain logistics, these salts can be stored for extended periods under standard conditions, allowing manufacturers to maintain strategic stockpiles against market fluctuations. The tolerance of the reaction to air and moisture further reduces the risk of batch failures due to minor environmental excursions during handling. This robustness ensures a consistent flow of commercial scale-up of complex phosphoryl azo compounds, enabling suppliers to meet tight delivery schedules and maintain continuity of supply for downstream customers in the drug discovery and agrochemical sectors.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method aligns perfectly with modern green chemistry principles. The reduction in hazardous waste generation and the avoidance of toxic by-products simplify wastewater treatment and废气 handling protocols. Scaling this reaction from gram to ton scale does not introduce new safety hazards, as the exotherm is manageable at room temperature. This ease of scale-up facilitates reducing lead time for high-purity phosphoryl azo compounds by shortening the process development timeline. Companies can move from bench-scale validation to commercial production much faster, responding agilely to market demands while maintaining full compliance with increasingly stringent environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for stakeholders evaluating this technology for their supply chains.
Q: What are the primary advantages of this new synthesis method over traditional routes?
A: The new method eliminates the need for hazardous phosphorus oxyhalides and strict low-temperature controls, offering a safer, more atom-economical process with near 100% selectivity.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the use of stable aryldiazonium salts and common solvents like chlorobenzene, combined with ambient temperature operation, makes it highly scalable for industrial manufacturing.
Q: What types of substrates are compatible with this catalytic system?
A: The system demonstrates broad substrate scope, tolerating various functional groups on both the aryl diazonium and phosphine oxide components, including halogens, methoxy, and alkyl groups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phosphoryl Azo Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this base-catalyzed synthesis route for the global fine chemicals market. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in practical manufacturing. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of phosphoryl azo intermediates meets the exacting standards required for pharmaceutical and advanced material applications. We are committed to leveraging this innovative chemistry to deliver superior value to our partners.
We invite you to collaborate with us to explore how this technology can optimize your specific project requirements. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how we can become your trusted partner in delivering high-quality chemical solutions efficiently and sustainably.
