Advanced Photocatalytic Synthesis of Benzocoumarins for High-Purity Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries are constantly seeking more sustainable and efficient synthetic routes for complex heterocyclic scaffolds. Patent CN110862368A introduces a groundbreaking photocatalytic oxidation method for the synthesis of benzocoumarin compounds, a class of molecules with significant potential in medicinal chemistry and optoelectronic materials. Unlike traditional thermal methods that often rely on harsh conditions and toxic reagents, this innovative approach utilizes visible light irradiation to drive the reaction under mild conditions. By employing 2-aryl-arylcarboxylic acids as readily available starting materials, the process achieves cyclization through a C-H functionalization and C-O lactonization sequence. This technology represents a paradigm shift towards greener manufacturing, addressing critical pain points regarding energy consumption and environmental impact while maintaining high yields and selectivity for the target benzocoumarin structures.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzocoumarins has relied heavily on transition metal-catalyzed C-H activation strategies involving palladium, copper, or silver systems. These conventional protocols typically require stoichiometric amounts of expensive and hazardous oxidants such as hypervalent iodine reagents or persulfates to regenerate the active catalytic species. A major drawback of these metal-mediated processes is the inevitable contamination of the final product with trace heavy metals, which poses severe challenges for pharmaceutical applications where strict purity limits are enforced. Furthermore, the removal of these metal residues often necessitates additional downstream purification steps, such as scavenging or recrystallization, which drastically increase production costs and reduce overall process efficiency. The reliance on high temperatures and aggressive oxidants also raises significant safety concerns and environmental liabilities, making these traditional routes less attractive for modern sustainable manufacturing standards.
The Novel Approach
In stark contrast, the method disclosed in CN110862368A leverages a metal-free photocatalytic system driven by organic catalysts DDQ and TBN under blue LED irradiation. This approach completely circumvents the use of transition metals, thereby eliminating the risk of metal leaching and simplifying the purification workflow significantly. The reaction proceeds at room temperature and atmospheric pressure using molecular oxygen as the terminal oxidant, which is not only cost-effective but also generates water as the only byproduct, aligning perfectly with green chemistry principles. The use of visible light provides a clean energy source that activates the catalytic cycle without the need for thermal input, resulting in substantial energy savings. This novel pathway offers a robust and environmentally benign alternative for producing high-value benzocoumarin intermediates with exceptional purity profiles.
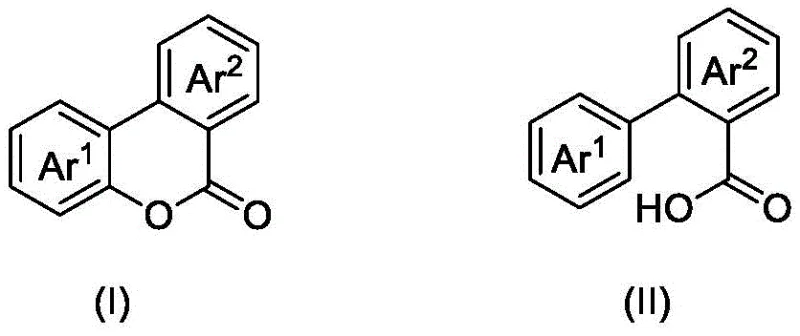
Mechanistic Insights into DDQ/TBN-Catalyzed Photocyclization
The core of this synthetic breakthrough lies in the synergistic interaction between 2,3-dichloro-5,6-dicyano-1,4-benzoquinone (DDQ) and tert-butyl nitrite (TBN) under photo-irradiation. Upon exposure to blue light, the catalytic system facilitates the generation of reactive radical species that initiate the C-H bond cleavage on the aryl ring of the carboxylic acid substrate. The DDQ acts as a potent electron acceptor and oxidant, while TBN serves as a radical initiator that helps propagate the oxidative cycle. Molecular oxygen plays a crucial role as the terminal electron acceptor, regenerating the active catalytic species and driving the thermodynamic equilibrium towards the formation of the lactone ring. This mechanism ensures a continuous turnover of the catalysts without the accumulation of inactive metal species, allowing the reaction to proceed efficiently over extended periods. The precise control over radical generation minimizes side reactions, leading to high selectivity for the desired benzocoumarin scaffold over potential over-oxidized byproducts.
From an impurity control perspective, the absence of transition metals fundamentally changes the impurity profile of the final product. In traditional methods, metal complexes can coordinate with the product or intermediates, creating difficult-to-remove impurities that require specialized chromatography. In this photocatalytic system, the primary impurities are likely derived from unreacted starting materials or minor oxidative degradation products, which are generally easier to separate using standard silica gel chromatography. The use of oxygen as the oxidant further ensures that no heavy metal salts or toxic organic oxidant residues remain in the reaction mixture. This clean reaction profile is particularly advantageous for the production of pharmaceutical intermediates, where regulatory compliance regarding residual solvents and elemental impurities is stringent. The robustness of the catalytic cycle allows for consistent quality across different batches, ensuring reliable supply for downstream drug synthesis.
How to Synthesize Benzocoumarins Efficiently
To implement this advanced synthesis in a laboratory or pilot plant setting, operators must carefully control the reaction parameters to maximize yield and reproducibility. The process begins by dissolving the 2-aryl-arylcarboxylic acid substrate in a suitable organic solvent, with 1,2-dichloroethane being the preferred medium due to its optimal solubility and stability under irradiation. Catalytic amounts of DDQ and TBN are added in a specific molar ratio to ensure efficient turnover without excessive reagent consumption. The reaction vessel is then purged with oxygen to create an aerobic environment essential for the oxidative cyclization. Exposure to an 18W blue LED lamp at room temperature initiates the photocatalytic cycle, which typically requires between 7 to 48 hours depending on the electronic nature of the substrate substituents. Detailed standardized synthesis steps are provided in the guide below.
- Dissolve 2-aryl-arylcarboxylic acid substrate in 1,2-dichloroethane and add catalytic amounts of DDQ and TBN.
- Replace air with oxygen and irradiate the mixture with an 18W blue LED lamp at room temperature for 7 to 48 hours.
- Remove solvent under reduced pressure and purify the crude product via column chromatography to obtain high-purity benzocoumarins.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this photocatalytic technology offers transformative benefits in terms of cost structure and operational reliability. The elimination of precious metal catalysts such as palladium or rhodium removes a significant variable cost component from the bill of materials, as these metals are subject to volatile market pricing and supply constraints. Furthermore, the simplified workup procedure reduces the consumption of auxiliary materials like metal scavengers and specialized filtration media, leading to direct savings in processing costs. The use of commodity chemicals like DDQ and TBN, combined with ambient air or bottled oxygen, ensures a stable and predictable supply chain that is less susceptible to geopolitical disruptions. This stability allows for better long-term planning and inventory management, reducing the risk of production stoppages due to raw material shortages.
- Cost Reduction in Manufacturing: The most significant economic advantage stems from the complete removal of transition metal catalysts, which are often the most expensive line item in fine chemical synthesis. By replacing these with inexpensive organic catalysts and oxygen, the direct material costs are drastically lowered. Additionally, the energy efficiency of running reactions at room temperature with LED lighting instead of heating mantles or oil baths results in lower utility bills. The simplified purification process also reduces labor hours and solvent usage during workup, contributing to a leaner and more cost-effective manufacturing operation overall.
- Enhanced Supply Chain Reliability: Sourcing high-purity transition metal catalysts can be challenging due to limited global suppliers and strict export controls. This new method relies on widely available organic reagents and gases that can be sourced from multiple vendors, diversifying the supply base and mitigating single-source risks. The robustness of the reaction conditions means that the process is less sensitive to minor variations in raw material quality, ensuring consistent output even with standard grade reagents. This reliability translates to shorter lead times and more dependable delivery schedules for customers requiring critical pharmaceutical intermediates.
- Scalability and Environmental Compliance: Scaling photochemical reactions has historically been difficult, but advancements in LED reactor technology have made this highly feasible for industrial production. The modular nature of LED arrays allows for easy expansion of capacity without the engineering complexities associated with high-pressure or high-temperature reactors. Moreover, the green nature of the process, utilizing oxygen and generating minimal waste, simplifies environmental permitting and waste disposal compliance. This alignment with sustainability goals enhances the corporate image and meets the increasing demand for eco-friendly manufacturing practices from end-users.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic synthesis method. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on process capabilities and limitations. Understanding these details is crucial for R&D teams evaluating the feasibility of this route for their specific projects and for procurement teams assessing the total cost of ownership. The information below highlights the versatility of the substrate scope and the practical advantages of the metal-free protocol.
Q: What are the primary advantages of this photocatalytic method over traditional thermal synthesis?
A: This method eliminates the need for transition metal catalysts like palladium or copper, thereby avoiding costly metal removal steps and ensuring higher product purity. Additionally, it utilizes molecular oxygen as a green terminal oxidant and operates at room temperature, significantly reducing energy consumption compared to high-temperature thermal processes.
Q: Is this process scalable for commercial production of pharmaceutical intermediates?
A: Yes, the process is highly scalable due to the use of standard blue LED light sources and common organic solvents like 1,2-dichloroethane. The absence of sensitive transition metal catalysts simplifies the supply chain and reduces the risk of batch-to-batch variability caused by metal contamination, making it ideal for large-scale manufacturing.
Q: What is the substrate scope for this benzocoumarin synthesis?
A: The method demonstrates excellent tolerance for a wide range of functional groups, including halogens (F, Cl, Br), alkyl groups (methyl, tert-butyl), and electron-withdrawing groups (CF3). It is applicable to various 2-aryl-arylcarboxylic acids, allowing for the synthesis of diverse benzocoumarin derivatives required in drug discovery.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzocoumarins Supplier
At NINGBO INNO PHARMCHEM, we recognize the immense potential of this photocatalytic technology to redefine the production landscape for benzocoumarins and related heterocyclic intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped with advanced photochemical reactors and rigorous QC labs capable of meeting stringent purity specifications required by global pharmaceutical clients. We are committed to delivering high-quality intermediates that adhere to the highest standards of safety and efficacy, leveraging our technical expertise to optimize every step of the value chain.
We invite you to collaborate with us to explore how this metal-free synthesis can enhance your supply chain resilience and reduce your overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact us to request specific COA data for our benzocoumarin portfolio and to discuss route feasibility assessments for your custom synthesis projects. Together, we can accelerate the development of next-generation therapeutics while maintaining a commitment to sustainable and efficient chemical manufacturing.
