Scalable Metal-Free Synthesis of 2,2-Bissulfonyl-2H-Aziridines for Advanced Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable pathways to access complex nitrogen-containing heterocycles, which serve as critical scaffolds for drug discovery. Patent CN110590631B introduces a groundbreaking methodology for the synthesis of 2,2-bissulfonyl-2H-aziridines, a class of compounds with significant potential in medicinal chemistry. Unlike traditional approaches that often rely on harsh conditions or unstable precursors, this invention utilizes a novel oxidative decarboxylation tandem cyclization strategy. By employing arylpropiolic acids and sodium sulfinates as starting materials, the process constructs two C-S bonds, one C-N bond, and one C=N bond in a single operational step. This metal-free protocol not only streamlines the synthetic route but also addresses key purity concerns associated with residual metal catalysts, positioning it as a highly attractive option for reliable pharmaceutical intermediate supplier networks aiming for greener and more efficient production standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2H-aziridine derivatives has frequently depended on the use of terminal alkynes as primary building blocks. While chemically feasible, terminal alkynes present substantial logistical and safety challenges in an industrial setting due to their inherent instability and high reactivity. These compounds often require stringent storage conditions and specialized handling procedures to prevent decomposition or hazardous side reactions. Furthermore, classical synthetic routes typically necessitate the use of strong bases and expensive transition metal catalysts to drive the cyclization forward. The reliance on heavy metals introduces a significant downstream burden, as rigorous purification steps are mandatory to reduce metal residues to parts-per-million levels acceptable for human therapeutic applications. This additional processing not only inflates the cost reduction in API manufacturing but also extends the overall production timeline, creating bottlenecks for supply chain managers who require consistent and rapid turnover of high-value intermediates.
The Novel Approach
In a significant departure from these legacy methods, the technology described in CN110590631B leverages arylpropiolic acids, which offer superior stability and ease of handling compared to their terminal alkyne counterparts. This shift in feedstock fundamentally alters the risk profile of the synthesis, allowing for safer storage and simpler logistics without compromising reactivity. The core innovation lies in the use of tert-butyl nitrite as a nitrogen source combined with an organic oxidant system, facilitating a cascade reaction that efficiently assembles the aziridine core. 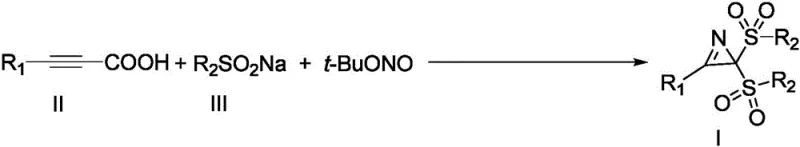 As illustrated in the general reaction scheme, this one-pot transformation bypasses the need for pre-functionalized substrates or multi-step sequences. The ability to forge multiple chemical bonds simultaneously while avoiding toxic metals represents a paradigm shift for cost reduction in pharmaceutical intermediate manufacturing, offering a cleaner, more direct route to these valuable heterocyclic structures that aligns perfectly with modern green chemistry principles.
As illustrated in the general reaction scheme, this one-pot transformation bypasses the need for pre-functionalized substrates or multi-step sequences. The ability to forge multiple chemical bonds simultaneously while avoiding toxic metals represents a paradigm shift for cost reduction in pharmaceutical intermediate manufacturing, offering a cleaner, more direct route to these valuable heterocyclic structures that aligns perfectly with modern green chemistry principles.
Mechanistic Insights into Metal-Free Oxidative Decarboxylation Cyclization
The mechanistic elegance of this transformation is rooted in the synergistic interaction between the organic oxidant and the fluoride promoter. The reaction initiates with the activation of the arylpropiolic acid, where the carboxylic acid group serves as a traceless directing group that is eventually expelled as carbon dioxide. This decarboxylation event is crucial as it generates a reactive vinyl radical or cationic species in situ, which is immediately trapped by the sulfonyl radical derived from the sodium sulfinate. The use of tert-butyl nitrite is particularly ingenious, as it acts as both an oxidant and the sole source of nitrogen for the aziridine ring, eliminating the need for external ammonia or amine sources that could lead to over-alkylation impurities. 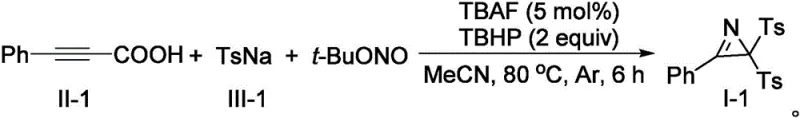 The presence of tetrabutylammonium fluoride (TBAF) plays a pivotal role in accelerating the reaction kinetics, likely by enhancing the nucleophilicity of the sulfinate or stabilizing transient intermediates. This carefully balanced catalytic cycle ensures that the reaction proceeds with high chemo- and regioselectivity, minimizing the formation of polymeric byproducts or isomeric impurities that often plague aziridine syntheses. For R&D directors, understanding this mechanism highlights the robustness of the process, as the mild conditions (80°C) and neutral pH environment preserve sensitive functional groups on the aromatic rings, thereby expanding the scope of accessible chemical space for drug design.
The presence of tetrabutylammonium fluoride (TBAF) plays a pivotal role in accelerating the reaction kinetics, likely by enhancing the nucleophilicity of the sulfinate or stabilizing transient intermediates. This carefully balanced catalytic cycle ensures that the reaction proceeds with high chemo- and regioselectivity, minimizing the formation of polymeric byproducts or isomeric impurities that often plague aziridine syntheses. For R&D directors, understanding this mechanism highlights the robustness of the process, as the mild conditions (80°C) and neutral pH environment preserve sensitive functional groups on the aromatic rings, thereby expanding the scope of accessible chemical space for drug design.
Impurity control is inherently built into this metal-free architecture. In traditional metal-catalyzed couplings, trace metals can coordinate with product molecules, leading to difficult-to-remove complexes that degrade product stability over time. By completely excluding transition metals like copper, palladium, or iron, this process guarantees a cleaner crude reaction profile. The primary byproducts are benign organic species and gases (CO2), which simplifies the workup procedure significantly. The patent data indicates that simple extraction followed by silica gel chromatography is sufficient to achieve high-purity 2,2-bissulfonyl-2H-aziridines. This level of purity is essential for high-purity pharmaceutical intermediate batches intended for subsequent coupling reactions, where even minor impurities can poison downstream catalysts or alter biological activity profiles. The broad substrate tolerance demonstrated in the patent, accommodating various electron-donating and electron-withdrawing groups, further confirms the versatility of this mechanistic pathway for generating diverse libraries of bioactive scaffolds.
How to Synthesize 2,2-Bissulfonyl-2H-Aziridines Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize yield and reproducibility. The standard protocol involves charging a Schlenk tube with the arylpropiolic acid substrate, sodium sulfinate, tert-butyl nitrite, TBAF, and TBHP in acetonitrile. The detailed standardized synthesis steps are outlined below to ensure technical teams can replicate the high yields reported in the patent literature.
- Combine arylpropiolic acid, sodium sulfinate, tert-butyl nitrite (t-BuONO), tetrabutylammonium fluoride (TBAF), and TBHP oxidant in acetonitrile solvent within a Schlenk tube.
- Purge the reactor with argon gas to create an inert atmosphere and stir the mixture in an oil bath at 80°C for approximately 6 hours.
- Upon completion, extract the reaction mixture with ethyl acetate, dry over anhydrous sodium sulfate, and purify the residue via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metal-free methodology offers tangible strategic benefits beyond mere chemical curiosity. The shift from unstable terminal alkynes to stable arylpropiolic acids drastically simplifies raw material sourcing and inventory management. Suppliers can stock larger quantities of the acid precursors without the fear of rapid degradation, ensuring continuity of supply even during market fluctuations. Furthermore, the elimination of precious metal catalysts removes a major cost driver and a significant regulatory hurdle. The absence of heavy metals means that the extensive and costly purification steps typically required to meet ICH Q3D guidelines for elemental impurities are rendered unnecessary. This streamlined downstream processing translates directly into reduced manufacturing costs and shorter lead times for high-purity pharmaceutical intermediates, allowing companies to bring new drug candidates to market faster and more economically.
- Cost Reduction in Manufacturing: The economic impact of removing transition metal catalysts cannot be overstated. Precious metals such as palladium or rhodium represent a significant portion of the bill of materials in fine chemical synthesis, and their prices are subject to volatile global markets. By utilizing an organic catalytic system based on inexpensive reagents like TBHP and TBAF, the process achieves substantial cost savings on raw materials. Additionally, the simplified workup procedure reduces the consumption of solvents and stationary phases required for metal scavenging, further lowering the operational expenditure. This efficiency makes the commercial scale-up of complex pharmaceutical intermediates much more financially viable, providing a competitive edge in pricing for generic drug manufacturers and CDMOs alike.
- Enhanced Supply Chain Reliability: Supply chain resilience is heavily dependent on the stability and availability of starting materials. Terminal alkynes, often used in older methods, can be hazardous to transport and have limited shelf lives, creating fragility in the supply network. In contrast, arylpropiolic acids are robust, commercially available solids that are easy to ship and store. This stability reduces the risk of production delays caused by spoiled raw materials. Moreover, the reaction uses common, commodity chemicals like acetonitrile and tert-butyl nitrite, which are widely produced and less susceptible to supply shortages compared to specialized ligands or organometallic complexes. This reliability ensures that production schedules can be maintained consistently, meeting the rigorous delivery timelines demanded by global pharmaceutical clients.
- Scalability and Environmental Compliance: As the industry moves towards more sustainable practices, the environmental footprint of chemical processes is under increasing scrutiny. This metal-free protocol generates minimal hazardous waste, primarily consisting of organic salts and CO2, which simplifies effluent treatment and disposal. The absence of toxic heavy metals in the waste stream significantly lowers the environmental compliance burden and associated disposal costs. The patent explicitly demonstrates scalability, with examples showing successful execution on larger scales without loss of efficiency. This scalability is crucial for transitioning from gram-scale R&D to kilogram or ton-scale commercial production, ensuring that the process remains robust and safe as volume increases, thereby supporting long-term growth and sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis method, derived directly from the experimental data and claims of the patent. These insights are intended to clarify the operational feasibility and strategic value of adopting this technology for your specific production needs.
Q: Why are arylpropiolic acids preferred over terminal alkynes for this synthesis?
A: Arylpropiolic acids are significantly more stable and easier to handle than terminal alkynes, which can be chemically volatile and difficult to store. This stability simplifies logistics and improves operational safety during large-scale manufacturing.
Q: Does this process require expensive transition metal catalysts?
A: No, the patented method operates under completely metal-free conditions using an organic catalytic system (TBAF/TBHP). This eliminates the risk of heavy metal contamination, a critical requirement for pharmaceutical intermediates.
Q: What represents the maximum yield achieved in the optimization studies?
A: Under optimized conditions using phenylpropiolic acid and sodium p-toluenesulfinate, the process achieved an isolated yield of 83%, demonstrating high efficiency for this complex multi-bond formation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,2-Bissulfonyl-2H-Aziridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free oxidative decarboxylation technology for the next generation of pharmaceutical intermediates. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, leveraging our rigorous QC labs to verify that every batch is free from residual metals and unwanted byproducts. Our state-of-the-art facilities are equipped to handle the specific safety and handling requirements of reagents like tert-butyl nitrite and organic peroxides, guaranteeing a safe and compliant manufacturing environment for our partners.
We invite you to collaborate with us to leverage this advanced synthetic route for your drug development programs. Whether you require custom synthesis of specific analogs or bulk supply of the core scaffold, our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your project's volume and timeline. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in metal-free catalysis can accelerate your path to market while optimizing your overall production costs.
