Advanced One-Pot Synthesis of 2-Azolyl-3-Alkylindoles for Pharmaceutical Manufacturing
Advanced One-Pot Synthesis of 2-Azolyl-3-Alkylindoles for Pharmaceutical Manufacturing
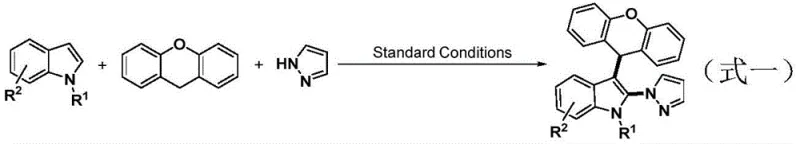
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for complex heterocyclic scaffolds, particularly those serving as critical intermediates for antiviral and anti-inflammatory agents. A significant breakthrough in this domain is detailed in patent CN115850252A, which discloses a highly efficient method for synthesizing 2-azolyl-3-alkylindole compounds. This technology leverages a novel iron-iodine co-catalytic oxidation system to achieve simultaneous C-H amination at the C2 position and alkylation at the C3 position of the indole core. By utilizing readily available indole, xanthene, and azole substrates, this one-pot protocol eliminates the necessity for pre-functionalized starting materials, thereby aligning perfectly with the principles of step economy and sustainable chemistry. For R&D directors and procurement specialists, this represents a paradigm shift from multi-step, low-yield sequences to a streamlined, high-atom-economy process that is both economically viable and environmentally responsible.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic strategies for constructing 2-substituted-3-alkylindoles often rely on classical Friedel-Crafts alkylations or transition-metal-catalyzed cross-coupling reactions that require pre-halogenated or pre-activated substrates. These conventional pathways are inherently inefficient, typically involving multiple discrete steps such as protection, functionalization, deprotection, and purification, each contributing to cumulative yield losses and increased waste generation. Furthermore, many established methods depend on expensive noble metal catalysts like palladium or rhodium, which not only escalate raw material costs but also introduce stringent requirements for residual metal removal to meet pharmaceutical purity standards. The reliance on stoichiometric oxidants in older protocols further exacerbates environmental concerns, generating substantial quantities of hazardous byproducts that complicate waste management and increase the overall carbon footprint of the manufacturing process.
The Novel Approach
In stark contrast, the methodology outlined in patent CN115850252A introduces a direct C-H functionalization strategy that bypasses the need for substrate pre-activation. By employing a synergistic iron-iodine catalytic system under an oxygen atmosphere, the process achieves dual functionalization in a single reaction vessel. This approach utilizes molecular oxygen as the terminal oxidant, which is not only cost-effective but also produces water as the primary byproduct, significantly enhancing the green chemistry profile of the synthesis. The reaction conditions are remarkably mild, typically operating at temperatures between 70°C and 80°C, which minimizes thermal degradation of sensitive functional groups. This direct assembly of the indole scaffold from simple building blocks like xanthene and azoles drastically simplifies the supply chain by reducing the number of required raw materials and intermediate storage requirements.
Mechanistic Insights into Iron-Iodine Co-Catalyzed Oxidation
The core of this technological advancement lies in the intricate interplay between the iron catalyst and the iodine co-catalyst within an oxidative environment. The proposed mechanism suggests that molecular oxygen plays a dual role: it participates in the regeneration of the active iodine species from iodide ions and simultaneously initiates the radical formation from the xanthene substrate. The iron catalyst, likely cycling between Fe(II) and Fe(III) oxidation states, facilitates the activation of the indole C-H bond, rendering it susceptible to nucleophilic attack or radical coupling. This cooperative catalysis ensures high chemical selectivity, favoring the formation of the desired C2-N and C3-C bonds while suppressing potential side reactions such as over-oxidation or polymerization. The presence of DDQ (2,3-dichloro-5,6-dicyanobenzoquinone) as a co-oxidant further stabilizes the reaction pathway, ensuring consistent conversion rates even with sterically hindered substrates.
From an impurity control perspective, this mechanism offers distinct advantages for process chemists. The high selectivity of the iron-iodine system means that fewer regioisomers and byproducts are generated compared to non-selective radical processes. The use of oxygen as a clean oxidant avoids the introduction of heavy metal contaminants often associated with stoichiometric oxidants like chromium or manganese salts. Consequently, the downstream purification process is simplified, often requiring only standard column chromatography to achieve high-purity specifications suitable for clinical applications. The compatibility of this catalytic system with a wide range of functional groups, including esters, halogens, and ethers, further underscores its robustness, allowing for the synthesis of diverse analogues without the need for extensive protecting group strategies.
How to Synthesize 2-Azolyl-3-Alkylindole Efficiently
The practical implementation of this synthesis is straightforward and amenable to standard laboratory and pilot-plant equipment. The process begins with the charging of indole derivatives, xanthene, and azole compounds into a reaction vessel, followed by the addition of the iron catalyst, iodine, and DDQ in a suitable organic solvent such as 1,4-dioxane or 1,2-dichloroethane. The detailed standardized synthesis steps are provided in the guide below, outlining the precise molar ratios and reaction parameters optimized in the patent examples to ensure maximum yield and reproducibility.
- Combine indole compounds, xanthene, and azole compounds in a reaction vessel with a metal catalyst (e.g., FeCl2), iodine, and DDQ oxidant in an organic solvent.
- Introduce oxygen gas and stir the mixture at a maintained temperature of 70-80°C for 8-12 hours to facilitate C-H activation and alkylation.
- Upon completion, cool the reaction to room temperature and purify the crude product using column chromatography with a petroleum ether and ethyl acetate gradient.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers compelling economic and logistical benefits that extend beyond mere chemical efficiency. The elimination of pre-functionalized substrates significantly reduces the complexity of the raw material portfolio, allowing for the sourcing of cheaper, commodity-grade chemicals like unsubstituted indoles and xanthenes. This simplification directly translates to lower inventory carrying costs and reduced risk of supply disruptions associated with specialized intermediates. Furthermore, the one-pot nature of the reaction consolidates multiple processing stages into a single unit operation, drastically cutting down on solvent usage, energy consumption, and labor hours required for intermediate handling and isolation.
- Cost Reduction in Manufacturing: The replacement of precious metal catalysts with abundant iron and iodine salts results in a substantial decrease in catalyst costs, which is a major driver of overall production expenses in fine chemical synthesis. Additionally, the use of molecular oxygen as the primary oxidant eliminates the need for purchasing expensive and hazardous stoichiometric oxidants, further driving down variable costs. The high atom economy of the reaction ensures that a greater proportion of the raw material mass is incorporated into the final product, minimizing waste disposal fees and maximizing material utilization efficiency.
- Enhanced Supply Chain Reliability: By relying on widely available and stable starting materials, manufacturers can mitigate the risks associated with the supply of exotic or custom-synthesized reagents. The robustness of the reaction conditions, which tolerate a broad spectrum of functional groups, allows for flexibility in sourcing substrates from multiple vendors without compromising product quality. This resilience is critical for maintaining continuous production schedules and meeting the demanding delivery timelines of downstream pharmaceutical clients.
- Scalability and Environmental Compliance: The simplicity of the workup procedure, involving basic cooling and chromatographic purification, facilitates easy scale-up from gram-scale laboratory synthesis to multi-ton commercial production. The reduced generation of hazardous waste and the avoidance of toxic heavy metals align with increasingly stringent global environmental regulations, ensuring long-term operational compliance and minimizing the regulatory burden on the manufacturing facility.

Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method, based on the data and examples provided in the patent documentation. These insights are designed to clarify the operational parameters and potential applications for stakeholders evaluating this technology for their own manufacturing pipelines.
Q: What are the key advantages of the iron-iodine co-catalytic system?
A: The system utilizes inexpensive and abundant iron and iodine catalysts, eliminating the need for precious metals. It operates under mild oxidative conditions using molecular oxygen, ensuring high atom economy and reduced environmental impact compared to traditional stoichiometric oxidants.
Q: Does this method support diverse substrate modifications?
A: Yes, the method demonstrates excellent functional group tolerance. It accommodates various substituents on the indole nitrogen (alkyl, allyl), the indole ring (electron-donating or withdrawing groups), and the xanthene core, allowing for the synthesis of a wide library of derivatives for drug discovery.
Q: How does this one-pot approach improve process efficiency?
A: By achieving simultaneous C2 amination and C3 alkylation in a single reaction vessel, the method removes the need for pre-functionalized substrates and intermediate isolation steps. This drastically reduces solvent consumption, waste generation, and overall processing time.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Azolyl-3-Alkylindole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the iron-iodine co-catalytic oxidation technology for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a GMP-compliant manufacturing environment. Our rigorous QC labs and stringent purity specifications guarantee that every batch of 2-azolyl-3-alkylindole meets the exacting standards required for drug substance manufacturing, providing our clients with the confidence needed to advance their development programs.
We invite you to collaborate with us to leverage this advanced synthetic route for your specific project needs. Our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this methodology can optimize your budget. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a reliable, cost-effective supply of these critical intermediates.
