Optimizing Fenofibrate Production: A Technical Analysis of In-Situ Chlorination for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust synthetic pathways that balance high purity with economic efficiency, particularly for established cardiovascular therapeutics like fenofibrate. The patent CN101730675A introduces a transformative methodology that addresses longstanding inefficiencies in the esterification of fenofibric acid, offering a streamlined approach that bypasses the isolation of unstable intermediates. This technical advancement is critical for manufacturers aiming to secure a reliable fenofibrate supplier status in a competitive global market where cost reduction in pharmaceutical intermediates manufacturing is paramount. By leveraging in-situ generation of the acid chloride, the process minimizes unit operations and solvent usage, directly impacting the bottom line for procurement teams. The following analysis dissects the chemical ingenuity behind this patent and its implications for commercial scalability and supply chain resilience.
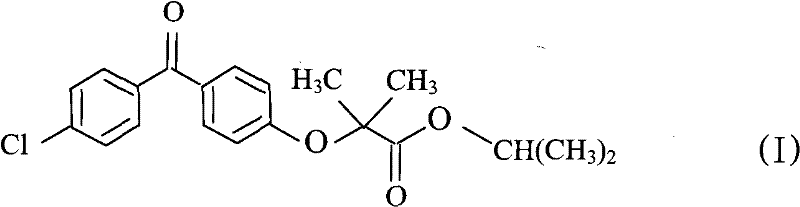
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of fenofibrate has relied on routes that involve the preparation and isolation of reactive halogenated intermediates, which introduce significant operational complexity and safety hazards. Prior art, such as that described in British patent application GB1539897, utilizes brominated derivatives that require stringent handling conditions and often result in lower overall yields due to intermediate degradation. The necessity to isolate and purify these intermediates before the final esterification step increases the consumption of auxiliary solvents and extends the production cycle time substantially. Furthermore, traditional methods often employ harsh conditions or expensive catalysts that complicate waste treatment and increase the environmental footprint of the manufacturing process. These inefficiencies create bottlenecks in the supply chain, making it difficult to respond rapidly to market demand fluctuations for high-purity pharmaceutical intermediates.
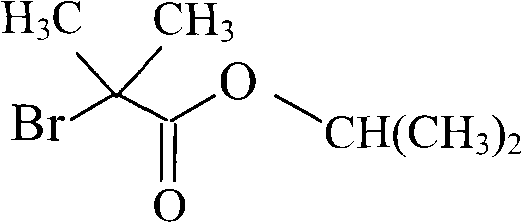
The Novel Approach
In stark contrast, the novel approach detailed in the provided patent data utilizes a direct in-situ chlorination strategy that fundamentally simplifies the reaction sequence. By reacting fenofibric acid directly with a chlorinating agent like thionyl chloride in the presence of isopropanol, the process eliminates the discrete isolation step of the acid chloride. This telescoping of reactions not only reduces the physical footprint required for production but also mitigates the risk of exposure to moisture-sensitive intermediates. The use of isopropanol as both the reaction solvent and the nucleophile further streamlines the material balance, removing the need for solvent exchange operations that typically incur high energy costs. Consequently, this methodology offers a pathway to cost reduction in pharmaceutical intermediates manufacturing by drastically cutting down on processing time and raw material overheads without compromising on the quality of the final active ingredient.
Mechanistic Insights into In-Situ Acid Chloride Formation
The core chemical innovation lies in the controlled generation of fenofibric acid chloride within the reaction medium, which immediately undergoes nucleophilic attack by isopropanol to form the ester bond. This mechanism relies on the precise stoichiometric control of the chlorinating agent, typically thionyl chloride, added in a slight excess ranging from 1.1 to 1.5 equivalents relative to the acid. The reaction proceeds efficiently at moderate temperatures between 60-90°C, avoiding the extreme thermal conditions that often lead to decomposition in sensitive molecular scaffolds. The presence of a base, such as potassium carbonate or triethylamine, serves to neutralize the hydrochloric acid byproduct generated during the chlorination, driving the equilibrium towards product formation. This careful management of reaction kinetics ensures that the acid chloride concentration remains low enough to prevent side reactions while being sufficient to maintain a rapid conversion rate to the desired ester.
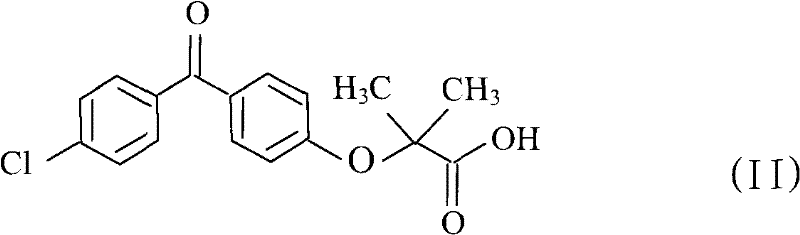
Impurity control is inherently enhanced in this system due to the absence of intermediate work-up steps where contamination typically occurs. In conventional multi-step syntheses, each isolation event presents an opportunity for the introduction of particulate matter or residual solvents, but this one-pot approach minimizes such risks. The patent data indicates that the final product achieves an HPLC purity of 99.98%, with residual fenofibric acid limited to merely 0.02%, demonstrating exceptional selectivity. This high level of purity is achieved without the need for recrystallization of the intermediate, which is a common source of yield loss in traditional protocols. For R&D directors focused on the commercial scale-up of complex pharmaceutical intermediates, this mechanism offers a reproducible and robust framework that aligns with stringent regulatory requirements for impurity profiles.
How to Synthesize Fenofibrate Efficiently
Implementing this synthesis route requires careful attention to the addition rate of the chlorinating agent and the maintenance of reflux conditions to ensure complete conversion. The process begins by suspending the fenofibric acid in isopropanol, followed by the gradual addition of thionyl chloride over a period of approximately 3 hours to manage exothermicity. Once the acid chloride is formed in situ, the mixture is maintained at reflux for several hours to allow the esterification to reach completion, monitored via HPLC to confirm the disappearance of starting materials. The detailed standardized synthetic steps see the guide below for specific operational parameters regarding quenching and crystallization.
- Suspend fenofibric acid in isopropanol and heat the mixture to reflux conditions to ensure proper solvation and reaction initiation.
- Add thionyl chloride gradually over a controlled period to generate the acid chloride intermediate in situ while maintaining temperature between 60-90°C.
- Quench the reaction mixture into an aqueous base solution to neutralize acid byproducts and induce crystallization of the high-purity fenofibrate product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented process delivers substantial value by addressing key pain points related to manufacturing costs and supply continuity. The elimination of intermediate isolation steps translates directly into reduced labor costs and lower utility consumption, as fewer distillation and filtration cycles are required. Additionally, the ability to use isopropanol as a dual-purpose reagent simplifies inventory management and reduces the volume of hazardous waste generated, leading to significant cost savings in waste disposal. These efficiencies make the production of high-purity pharmaceutical intermediates more economically viable, allowing suppliers to offer competitive pricing without sacrificing margin. For supply chain heads, the robustness of this method ensures reducing lead time for high-purity pharmaceutical intermediates by minimizing the potential for batch failures or reprocessing.
- Cost Reduction in Manufacturing: The process achieves cost optimization primarily through the removal of expensive purification stages associated with intermediate isolation. By avoiding the recrystallization of the acid chloride, manufacturers save on solvent volumes and energy required for drying and heating, which cumulatively results in drastic simplification of the cost structure. Furthermore, the high yield reported in the patent data implies less raw material waste per kilogram of finished product, enhancing overall material efficiency. This logical deduction of cost benefits suggests a highly competitive pricing model for bulk procurement without the need for speculative financial projections.
- Enhanced Supply Chain Reliability: The simplified operational workflow reduces the number of potential failure points in the manufacturing chain, thereby increasing batch success rates. Since the reaction conditions are moderate and do not require specialized cryogenic equipment or high-pressure vessels, the barrier to entry for scaling production is lowered. This accessibility ensures that supply can be ramped up quickly to meet sudden surges in demand, providing a stable source of reliable fenofibrate supplier capacity. The use of common reagents like thionyl chloride and isopropanol also mitigates the risk of raw material shortages that might plague more exotic synthetic routes.
- Scalability and Environmental Compliance: Scaling this process from laboratory to industrial quantities is straightforward due to the homogeneous nature of the reaction mixture once the acid chloride forms. The reduced solvent load and the absence of heavy metal catalysts simplify the effluent treatment process, ensuring compliance with increasingly strict environmental regulations. This environmental advantage is crucial for long-term sustainability and avoids potential shutdowns due to non-compliance issues. Consequently, the commercial scale-up of complex pharmaceutical intermediates becomes a lower-risk investment for manufacturing partners looking to expand their portfolio.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation to ensure accuracy and relevance. Understanding these details is essential for stakeholders evaluating the feasibility of adopting this method for their own production lines or sourcing strategies.
Q: What are the primary advantages of the in-situ chlorination method for fenofibrate?
A: The in-situ method eliminates the need to isolate and recrystallize the unstable acid chloride intermediate, significantly reducing processing time and solvent consumption while achieving yields exceeding 96%.
Q: How does this process improve impurity control compared to conventional routes?
A: By avoiding the isolation of reactive intermediates and utilizing a direct esterification pathway, the formation of side products is minimized, resulting in assay purity levels reaching 99.98% without complex purification steps.
Q: Is this synthesis route suitable for large-scale commercial manufacturing?
A: Yes, the process operates at moderate temperatures (60-90°C) and uses isopropanol as both solvent and reactant, simplifying equipment requirements and enhancing safety profiles for metric-ton scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fenofibrate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthesis routes in maintaining a competitive edge in the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the in-situ chlorination method are translated into tangible supply chain assets. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of fenofibrate meets the highest international standards. Our commitment to technical excellence allows us to navigate the complexities of fine chemical manufacturing with precision and reliability.
We invite you to collaborate with us to leverage these advanced manufacturing capabilities for your specific project needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized processes can enhance your product lifecycle and profitability.
