Advanced Visible Light Catalysis for High-Purity Chiral Azaarene Intermediates
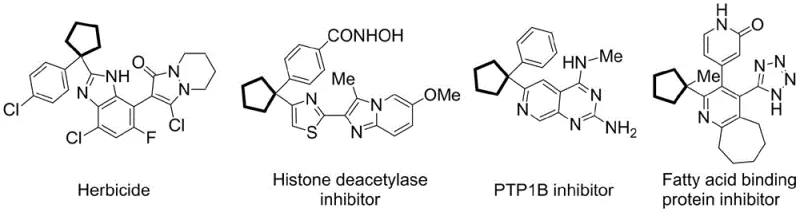
The pharmaceutical and agrochemical industries are constantly seeking more efficient and environmentally benign pathways to construct complex molecular architectures, particularly those containing chiral quaternary carbon centers. Patent CN113248422A introduces a groundbreaking visible-light asymmetric catalytic synthesis method for chiral alpha-azaarene quaternary carbon center compounds, addressing critical challenges in modern organic synthesis. This technology leverages a dual-catalyst system comprising DPZ as a visible light redox catalyst and BINOL-derived biaxial chiral phosphinimide acid (BINOL-IDPA) to drive the reaction under mild conditions. The significance of this innovation lies in its ability to construct a chiral pentacyclopentane structure at the alpha-position of azaarenes, a skeleton widely found in bioactive molecules such as herbicides, histone deacetylase inhibitors, and PTP1B inhibitors. By utilizing a metal-free photocatalytic approach, this method offers a sustainable alternative to traditional transition-metal catalyzed processes, ensuring high purity and safety profiles essential for reliable pharmaceutical intermediate supplier standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral alpha-azaarene derivatives has relied heavily on transition metal catalysis or harsh thermal conditions, which present significant drawbacks for large-scale manufacturing. Traditional methods often require expensive noble metals like palladium or rhodium, leading to substantial cost increases and complex downstream processing to remove trace metal residues that are strictly regulated in drug substances. Furthermore, these conventional routes frequently suffer from limited substrate scope and poor stereoselectivity, necessitating tedious resolution steps that drastically reduce overall yield. The reliance on high temperatures and strong bases in older methodologies also poses safety risks and limits the compatibility with sensitive functional groups, thereby restricting the diversity of accessible chemical space for drug discovery teams.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a visible-light mediated radical addition cyclization strategy that operates at cryogenic temperatures ranging from -50 to -70°C. This method employs N-aryl substituted cyclopropylamine and alpha-aryl-alpha-(2-azaaryl) substituted terminal olefin as readily available starting materials, reacting them in a mixed solvent system of diethyl ether, cyclopentyl methyl ether, and benzene. The use of DPZ as an organic photocatalyst eliminates the need for heavy metals entirely, while the chiral BINOL-IDPA catalyst ensures exceptional stereocontrol. This green chemistry protocol not only simplifies the reaction setup but also significantly enhances the environmental profile of the synthesis, aligning perfectly with the industry's shift towards cost reduction in API manufacturing through process intensification and waste minimization.
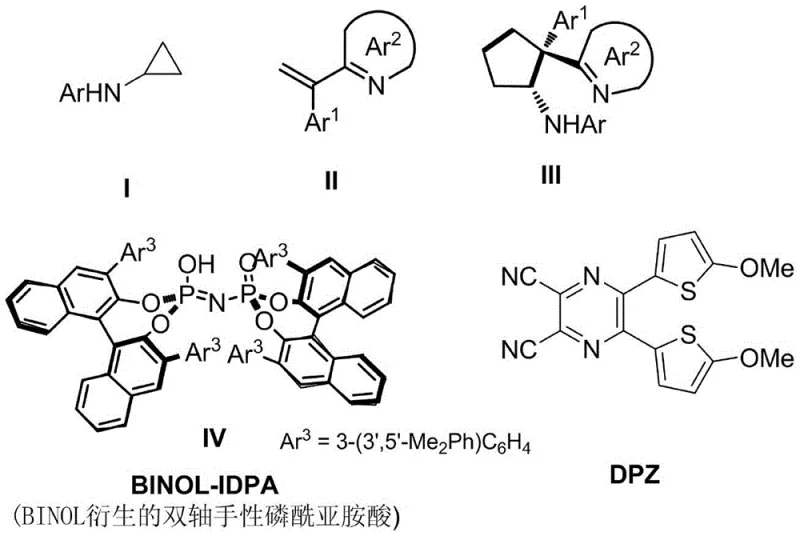
Mechanistic Insights into Visible Light Photocatalytic Cyclization
The core of this technological breakthrough lies in the intricate interplay between the excited state of the DPZ photocatalyst and the chiral environment provided by the BINOL-IDPA. Upon irradiation with blue light (450-455nm), the DPZ catalyst enters an excited state capable of engaging in single-electron transfer (SET) processes with the electron-deficient azaarene moiety. This initiates a radical cascade where the cyclopropylamine ring opens to generate a distal radical species, which subsequently undergoes intramolecular addition to the azaarene double bond. The chiral phosphinimide acid activates the imine group through hydrogen bonding, creating a rigid chiral pocket that dictates the facial selectivity of the radical attack. This precise mechanistic control is what allows the reaction to achieve diastereoselectivity ratios greater than 19:1 and enantiomeric excess values up to 96%, ensuring the production of high-purity chiral intermediates with minimal isomeric impurities.
From an impurity control perspective, the mild reaction conditions play a pivotal role in maintaining product integrity. Unlike thermal reactions that can promote side reactions such as polymerization or decomposition of sensitive heterocycles, the photochemical pathway proceeds with high chemoselectivity. The absence of strong acids or bases prevents the racemization of the newly formed chiral center, a common issue in traditional acid-catalyzed cyclizations. Furthermore, the specific solvent mixture chosen optimizes the solubility of both the organic photocatalyst and the chiral acid, ensuring a homogeneous reaction medium that maximizes catalyst turnover and minimizes the formation of byproducts. This level of control is critical for meeting the stringent quality specifications required by global regulatory bodies for active pharmaceutical ingredients.
How to Synthesize Chiral Alpha-Azaarene Compounds Efficiently
To implement this synthesis in a laboratory or pilot plant setting, operators must adhere to strict protocols regarding light intensity and temperature control to maximize yield and selectivity. The process begins with the precise weighing of substrates and catalysts, followed by the preparation of the ternary solvent system which is crucial for stabilizing the radical intermediates. The reaction vessel must be thoroughly degassed to prevent oxygen quenching of the excited photocatalyst, typically achieved through multiple freeze-pump-thaw cycles. Once the reaction is initiated under blue LED irradiation, it requires a prolonged stirring period of approximately 60 hours to reach full conversion, after which standard workup procedures involving solvent evaporation and silica gel chromatography are employed to isolate the final product.
- Prepare the reaction mixture by dissolving N-aryl substituted cyclopropylamine, alpha-aryl-alpha-(2-azaaryl) substituted terminal olefin, DPZ photocatalyst, and BINOL-IDPA chiral catalyst in a mixed solvent of diethyl ether, cyclopentyl methyl ether, and benzene.
- Subject the mixture to freeze-pump-thaw cycles to ensure an inert atmosphere, then maintain the reaction at -50 to -70°C under irradiation with a 3W blue LED lamp (450-455nm).
- After 60 hours, remove the solvent via rotary evaporation and purify the crude product using column chromatography with a hexane/ethyl acetate gradient to isolate the target chiral compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this visible-light catalytic method presents a compelling value proposition driven by operational efficiency and raw material accessibility. The elimination of precious metal catalysts removes a significant cost driver associated with both the initial purchase of reagents and the subsequent validation of metal clearance, which often requires specialized resin treatments. Additionally, the use of commodity solvents like diethyl ether and benzene, rather than exotic fluorinated solvents, ensures a stable and predictable supply chain that is less susceptible to market volatility. The robustness of the reaction conditions also implies a lower risk of batch failure, enhancing overall supply reliability for critical drug development programs.
- Cost Reduction in Manufacturing: The transition to a metal-free photocatalytic system fundamentally alters the cost structure of producing these complex intermediates. By removing the necessity for expensive transition metals and the associated scavenging agents, manufacturers can achieve substantial cost savings in raw material expenditure. Furthermore, the mild reaction temperatures reduce the energy load required for heating, shifting the utility demand to lighting which is generally more efficient and easier to control in a reactor setting. The high yields and selectivity observed in the patent examples mean that less starting material is wasted, directly improving the atom economy and reducing the cost per kilogram of the final active ingredient.
- Enhanced Supply Chain Reliability: The substrates required for this synthesis, specifically N-aryl cyclopropylamines and vinyl-azaarenes, are structurally simple and can be sourced from multiple global suppliers, mitigating the risk of single-source dependency. The stability of the DPZ and BINOL-IDPA catalysts allows for long-term storage without significant degradation, facilitating better inventory management and reducing the frequency of urgent reordering. This resilience in the supply chain is crucial for maintaining continuous production schedules, especially when scaling up from clinical trial materials to commercial launch volumes where consistency is paramount.
- Scalability and Environmental Compliance: Scaling photochemical reactions has historically been challenging due to light penetration issues, but advancements in flow chemistry and LED array technology have made this increasingly viable for commercial scale-up of complex heterocycles. The method's inherent green chemistry credentials, characterized by the absence of toxic heavy metals and the use of ambient pressure, simplify the environmental permitting process and reduce the burden of hazardous waste disposal. This alignment with sustainability goals not only lowers compliance costs but also enhances the corporate social responsibility profile of the manufacturing entity, a factor increasingly weighted in vendor selection processes by major pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this visible-light catalytic technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear understanding of the method's capabilities and limitations for potential partners and licensees.
Q: What are the primary advantages of using DPZ photocatalyst over traditional transition metal catalysts?
A: The use of DPZ, a metal-free organic photocatalyst, eliminates the risk of heavy metal contamination in the final product, which is critical for pharmaceutical compliance. It also operates under milder conditions compared to traditional thermal methods, reducing energy consumption and simplifying the purification process by removing the need for expensive metal scavengers.
Q: How does the BINOL-IDPA catalyst influence the stereoselectivity of the reaction?
A: BINOL-IDPA acts as a chiral Brønsted acid catalyst that creates a highly organized chiral environment during the radical addition cyclization. This precise spatial control ensures high enantioselectivity (up to 96% ee) and excellent diastereoselectivity (dr > 19:1), which is essential for producing single-isomer drug candidates.
Q: Is this synthesis method scalable for industrial production of API intermediates?
A: Yes, the method is highly scalable due to its simple operational requirements and the stability of the catalysts. The use of common solvents and the absence of sensitive transition metals make it suitable for commercial scale-up, offering a robust pathway for manufacturing complex heterocyclic intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Alpha-Azaarene Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the visible-light catalytic synthesis described in CN113248422A for the next generation of therapeutic agents. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to market supply is seamless and efficient. Our state-of-the-art facilities are equipped with advanced photochemical reactors and rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to guarantee the high enantiomeric excess required for your drug candidates. We are committed to delivering high-purity chiral intermediates that meet the highest global regulatory standards.
We invite you to collaborate with our technical team to explore how this innovative synthesis route can optimize your specific project requirements. By leveraging our expertise in process development, we can provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating exactly how this metal-free approach can improve your bottom line. Please contact our technical procurement team today to request specific COA data for similar scaffolds and discuss route feasibility assessments for your target molecules, ensuring a secure and competitive supply chain for your future products.
