Advanced Catalytic Synthesis of Desoxytazobactam Diphenylmethyl Ester for Commercial API Production
Advanced Catalytic Synthesis of Desoxytazobactam Diphenylmethyl Ester for Commercial API Production
The pharmaceutical industry continuously seeks robust and scalable methodologies for the production of critical beta-lactamase inhibitor intermediates, specifically focusing on the synthesis of desoxytazobactam diphenylmethyl ester, a pivotal precursor for the widely prescribed antibiotic combination tazobactam/piperacillin. Patent CN113861222A introduces a groundbreaking advancement in this domain by disclosing a method that utilizes a novel weakly alkaline supported bimetal porous coordination polymer catalyst. This technological leap addresses longstanding challenges in the field, offering a pathway that not only enhances reaction kinetics but also significantly improves the safety profile and economic feasibility of large-scale manufacturing. By leveraging a unique bimetallic system involving copper and tin stabilized within a silica matrix, the invention achieves yields ranging from 82% to 86% under mild conditions, representing a substantial improvement over legacy processes that often suffer from low efficiency and hazardous reagent requirements.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of tazobactam intermediates has been plagued by significant safety and efficiency hurdles, primarily stemming from the reliance on perilous reagents and unstable reaction intermediates. One of the most prevalent traditional routes involves the use of sodium azide and acetylene, chemicals that are notoriously flammable, explosive, and highly toxic, thereby imposing severe safety constraints and requiring specialized, costly infrastructure for handling and waste disposal. Furthermore, alternative direct condensation methods, while avoiding azides, often encounter kinetic bottlenecks where the reaction time for triazole incorporation is excessively long, leading to reduced throughput in commercial reactors. A critical chemical deficiency in these older pathways is the propensity for carbocation rearrangement; under alkaline conditions, the chloride ions in the starting material are easily displaced, leading to the formation of undesirable six-membered ring byproducts that drastically lower the overall yield and complicate downstream purification efforts.
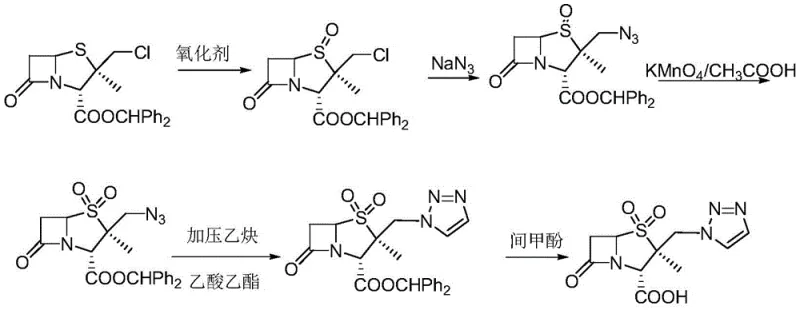
The Novel Approach
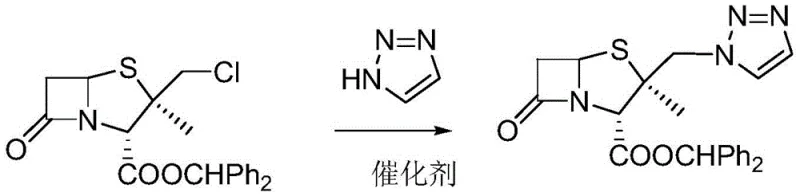
In stark contrast to these legacy methodologies, the innovative process detailed in the patent employs a sophisticated catalytic system that fundamentally alters the reaction landscape to favor the desired product with high selectivity. The core of this advancement lies in the deployment of a weakly alkaline load-type bimetal porous coordination polymer catalyst, which is engineered to possess superior thermal stability and a high density of active sites. This catalyst facilitates the nucleophilic substitution of the chloromethyl group with the triazole moiety in a remarkably short timeframe, effectively suppressing the side reactions that typically degrade product quality in conventional syntheses. By operating within a benign two-phase system of acetone and water, the new method not only accelerates the reaction rate but also creates a homogeneous environment that further mitigates the risk of byproduct formation, ensuring a cleaner reaction profile and a more streamlined isolation process for the final ester.
Mechanistic Insights into Bimetallic Coordination Polymer Catalysis
The efficacy of this novel synthesis route is deeply rooted in the intricate structural design of the catalyst, which functions as a heterogeneous yet highly active promoter for the triazole coupling reaction. The catalyst is constructed through a solvothermal synthesis of a copper-tin coordination polymer, which is subsequently loaded into the pore channels of a silica gel carrier to prevent aggregation and leaching of the active metal species. A crucial step in the catalyst preparation involves the reduction of the metal centers using sodium borohydride, which generates zero-valent metal species encapsulated within the carrier's pores; this stabilization mechanism prevents the deactivation of active sites that commonly occurs during repetitive catalytic cycles. The resulting material exhibits weak alkalinity, which is perfectly tuned to activate the triazole nucleophile without promoting the deleterious elimination reactions that lead to carbocation rearrangements, thereby maintaining the integrity of the beta-lactam scaffold throughout the transformation.
From an impurity control perspective, the mechanistic advantage of this system is its ability to kinetically outcompete the formation of rearrangement byproducts. In traditional uncatalyzed or poorly catalyzed systems, the lone pair electrons on the sulfur atom can facilitate the departure of the chloride leaving group, generating a reactive carbocation that readily undergoes skeletal rearrangement to form thermodynamically stable but unwanted six-membered rings. The novel bimetallic catalyst, however, provides a coordinated environment that likely activates the triazole ring towards nucleophilic attack while simultaneously stabilizing the transition state of the substitution reaction. This dual action ensures that the substitution occurs rapidly and selectively at the intended chloromethyl position, effectively shutting down the pathway to rearrangement impurities and resulting in a crude product with significantly higher purity, which reduces the burden on subsequent crystallization and purification steps.
How to Synthesize Desoxytazobactam Diphenylmethyl Ester Efficiently
The practical implementation of this synthesis involves a straightforward sequence of operations that begins with the preparation of the specialized catalyst followed by the main coupling reaction under controlled thermal conditions. The process is designed to be operationally simple, utilizing common solvents like acetone and water, and requires only mild heating or cooling to maintain the optimal reaction window of 10-20°C. Detailed standardized synthesis steps, including precise stoichiometric ratios and workup procedures, are outlined below to ensure reproducibility and adherence to the high-quality standards expected in pharmaceutical intermediate manufacturing.
- Preparation of the bimetal coordination polymer via solvothermal method using copper chloride, stannic chloride, pyridine, and imidazole.
- Loading the polymer onto silica gel and stabilizing via metal reduction with sodium borohydride to create the active catalyst.
- Reacting 2-beta-chloromethyl penicillanic acid diphenylmethyl ester with triazole in an acetone-water system using the novel catalyst at 10-20°C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this catalytic technology translates into tangible strategic benefits that extend far beyond simple chemical yield improvements. The elimination of hazardous reagents like sodium azide removes a significant logistical burden, as the sourcing, storage, and disposal of such materials often incur high regulatory costs and insurance premiums. Furthermore, the enhanced stability and recyclability of the bimetallic catalyst mean that the consumption of expensive catalytic materials is drastically reduced over time, leading to a more predictable and lower cost of goods sold (COGS) for the final intermediate. The shortened reaction time directly correlates to increased reactor turnover rates, allowing manufacturing facilities to produce larger volumes of high-purity pharmaceutical intermediates within the same operational timeframe, thereby enhancing overall supply chain responsiveness.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the removal of expensive and dangerous reagents and the implementation of a reusable catalyst system. By avoiding the need for sodium azide and acetylene, manufacturers eliminate the costs associated with specialized safety equipment, hazardous waste treatment, and the premium pricing of these controlled substances. Additionally, the catalyst's ability to be recovered and reused for multiple batches without significant loss of activity means that the effective cost per kilogram of catalyst consumed is negligible, contributing to substantial long-term savings in raw material expenditures.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the use of commodity chemicals such as acetone, water, and silica gel, which are readily available in global markets and less susceptible to the supply disruptions that often affect specialty or hazardous reagents. The robustness of the catalyst also ensures consistent batch-to-batch quality, reducing the risk of production delays caused by failed batches or the need for reprocessing. This reliability allows for more accurate demand forecasting and inventory management, ensuring a steady flow of critical intermediates to downstream API production lines.
- Scalability and Environmental Compliance: The process is inherently scalable due to its reliance on a heterogeneous catalyst that can be easily separated via filtration, a unit operation that scales linearly from pilot plant to commercial production without complex engineering changes. From an environmental standpoint, the use of a green solvent system (acetone/water) and the absence of heavy metal leaching or toxic byproducts simplify wastewater treatment and align with increasingly stringent global environmental regulations. This compliance reduces the risk of regulatory shutdowns and enhances the corporate sustainability profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel catalytic synthesis method. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the technology's capabilities and limitations for potential adopters.
Q: What are the safety advantages of this new catalytic route compared to traditional azide methods?
A: The novel catalytic route eliminates the need for highly toxic and explosive reagents such as sodium azide and acetylene, which are required in conventional synthesis pathways. By utilizing a stable bimetallic coordination polymer catalyst in a mild acetone-water system, the process significantly reduces operational hazards and simplifies regulatory compliance for manufacturing facilities.
Q: How does the new catalyst improve product yield and purity?
A: The weakly alkaline supported bimetal porous coordination polymer catalyst features multiple active sites that accelerate the triazole reaction while suppressing side reactions like carbocation rearrangement. This results in a substantial increase in product yield, reaching 82-86%, and ensures high purity levels suitable for downstream API synthesis without extensive purification steps.
Q: Is the catalyst reusable for large-scale production?
A: Yes, the catalyst is designed for high stability and recyclability. The bimetallic polymer is loaded into silica gel pore channels and stabilized through metal reduction, preventing the loss of active sites during the reaction. This allows the catalyst to be recovered via simple filtration and reused for multiple batches, enhancing the economic viability of the process.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Desoxytazobactam Diphenylmethyl Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and safe synthesis routes for high-value pharmaceutical intermediates like desoxytazobactam diphenylmethyl ester. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of advanced catalytic technologies are fully realized in a GMP-compliant manufacturing environment. Our rigorous QC labs and commitment to stringent purity specifications guarantee that every batch of intermediate we supply meets the exacting standards required for the synthesis of life-saving beta-lactamase inhibitors.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this cutting-edge synthesis technology for their supply chains. By partnering with our technical procurement team, you can obtain a Customized Cost-Saving Analysis tailored to your specific volume requirements, along with specific COA data and route feasibility assessments. Contact us today to discuss how we can support your production goals with reliable, high-quality intermediates produced via this innovative and sustainable catalytic process.
