Scalable Synthesis of Novel Monohydroxy-2-acylphenylacetates for Advanced Pharmaceutical Intermediates
Scalable Synthesis of Novel Monohydroxy-2-acylphenylacetates for Advanced Pharmaceutical Intermediates
The pharmaceutical industry continuously seeks novel scaffolds that can serve as potent leads for antitumor therapy, particularly those that offer distinct mechanisms of action compared to existing cytotoxic agents. Patent CN101407459B introduces a significant advancement in this domain by disclosing a class of monohydroxy-2-acylphenylacetates, designated as compounds CsM, which exhibit promising bioactivity against human gastric cancer cell lines. Unlike their naturally occurring polyhydroxy counterparts which often suffer from complex isolation procedures and structural heterogeneity, these synthetic analogs provide a defined, modular architecture that facilitates rigorous structure-activity relationship (SAR) studies. The disclosed methodology leverages a robust seven-step synthetic sequence starting from readily available hydroxybenzaldehydes, ensuring that the production of these high-purity pharmaceutical intermediates can be achieved with consistent quality and reproducibility essential for drug development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the acquisition of bioactive phenylacetate derivatives has relied heavily on the extraction of secondary metabolites from fungal sources, a process fraught with significant supply chain vulnerabilities and technical inefficiencies. Natural fermentation processes typically yield complex mixtures of polyhydroxy analogs, such as Cytosporone B, where separating individual congeners requires extensive and costly chromatographic purification, often resulting in poor overall recovery rates. Furthermore, the structural rigidity of natural products limits the ability of medicinal chemists to modify specific functional groups to enhance potency or improve pharmacokinetic properties without resorting to semi-synthetic modifications that are often low-yielding. The reliance on biological sources also introduces batch-to-batch variability due to strain mutations or fermentation condition fluctuations, making it nearly impossible to guarantee the consistent supply of high-purity material required for preclinical and clinical trials.
The Novel Approach
The synthetic strategy outlined in the patent represents a paradigm shift by establishing a fully chemical route that bypasses the unpredictability of biological sourcing entirely. By utilizing a convergent synthesis approach centered on a Friedel-Crafts intramolecular acylation, the process constructs the core indanone scaffold with high regioselectivity, allowing for the precise placement of the hydroxyl group at the C3, C4, or C6 positions. This total synthesis method enables the systematic variation of both the acyl side chain (R2) and the ester moiety (R1), granting researchers unprecedented control over the physicochemical properties of the final molecule. The use of standard organic transformations such as Grignard additions and oxidative cleavages ensures that the process is not only chemically elegant but also practically viable for industrial implementation, effectively transforming a niche natural product class into a commercially accessible library of drug candidates.
Mechanistic Insights into AlCl3-Catalyzed Cyclization and Oxidative Cleavage
The cornerstone of this synthetic pathway is the aluminum chloride-mediated intramolecular acylation which converts the linear hydroxyphenylpropionic acid into the cyclic indanone structure. This Friedel-Crafts reaction proceeds through the formation of a highly reactive acylium ion intermediate, which subsequently attacks the electron-rich aromatic ring ortho to the activating hydroxyl group. The choice of reaction conditions, specifically the use of molten salt conditions or high-temperature stirring with anhydrous AlCl3, is critical for driving the equilibrium towards cyclization while minimizing polymerization side reactions. Understanding the electronic effects of the substituents on the benzene ring is paramount for R&D teams, as the position of the hydroxyl group dictates the orientation of the incoming acyl group, thereby determining the final substitution pattern of the pharmacophore.
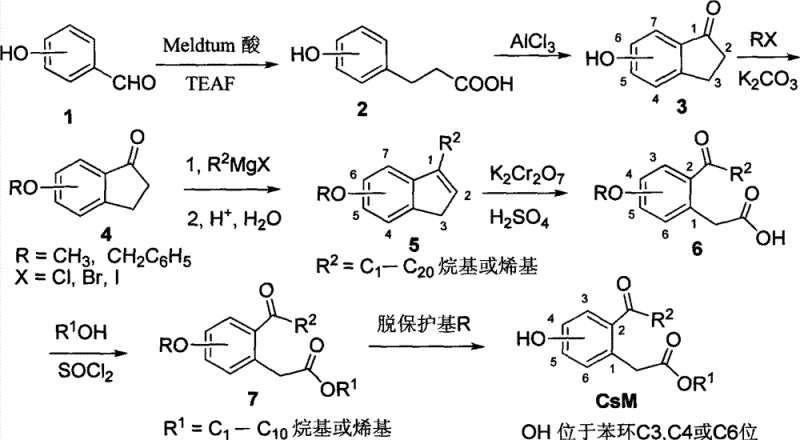
Following the construction of the carbon skeleton, the oxidative cleavage of the indene double bond serves as the pivotal step for generating the acetic acid side chain functionality. The patent specifies the use of potassium dichromate in acidic media, a potent oxidizing system capable of breaking the carbon-carbon double bond to yield the corresponding ketone and carboxylic acid moieties simultaneously. From a process chemistry perspective, controlling the stoichiometry of the oxidant is essential to prevent over-oxidation of the sensitive benzylic positions or the aromatic ring itself. The subsequent esterification using thionyl chloride activates the carboxylic acid for nucleophilic attack by various alcohols, allowing for the rapid generation of diverse ester derivatives. Finally, the removal of the protecting group, whether via catalytic hydrogenolysis for benzyl ethers or Lewis acid mediated cleavage for methyl ethers, reveals the active phenolic hydroxyl group necessary for biological interaction, completing the transformation from simple building blocks to complex bioactive esters.
How to Synthesize Monohydroxy-2-acylphenylacetate Efficiently
Executing this synthesis requires careful attention to moisture control during the Grignard addition and the Friedel-Crafts cyclization steps, as water can quench reactive intermediates and drastically reduce yields. The process begins with the condensation of the aldehyde and Meldrum's acid, followed by the critical ring-closing step which establishes the core geometry. Subsequent functionalization involves protecting the phenol to prevent interference during the organometallic addition, followed by the oxidative ring opening which installs the key acetyl-acetate motif. For detailed operational parameters including specific molar ratios, temperature profiles, and workup procedures, please refer to the standardized guide below.
- Condense hydroxybenzaldehyde with Meldrum acid using TEAF to form hydroxyphenylpropionic acid.
- Perform intramolecular acylation with AlCl3 to generate the indanone core structure.
- Protect the hydroxyl group with alkyl halides, followed by Grignard addition to introduce the acyl side chain precursor.
- Oxidatively cleave the indene ring using potassium dichromate to yield the 2-acyl-arylacetic acid.
- Esterify the acid with thionyl chloride and alcohol, then remove the protecting group to obtain the final CsM compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from extraction-based sourcing to this fully synthetic route offers profound strategic benefits regarding cost stability and supply security. The reliance on commodity chemicals such as hydroxybenzaldehydes, Meldrum's acid, and simple alkyl halides means that the raw material basket is insulated from the volatility often associated with specialized fermentation substrates or rare natural extracts. This shift fundamentally alters the cost structure of the intermediate, moving it from a high-margin, low-volume specialty category to a more predictable manufacturing cost model driven by bulk chemical pricing. Furthermore, the synthetic route eliminates the lengthy lead times associated with biological cultivation and extraction, enabling a much more responsive production schedule that can adapt quickly to fluctuating demand from downstream drug development partners.
- Cost Reduction in Manufacturing: The elimination of expensive chromatographic separations required for natural product isolation significantly lowers the operational expenditure per kilogram of produced material. By replacing complex purification trains with standard crystallization and extraction techniques, the process reduces solvent consumption and waste disposal costs, leading to substantial overall cost savings in pharmaceutical intermediate manufacturing. Additionally, the use of non-precious metal catalysts like aluminum chloride instead of palladium or rhodium in the key bond-forming steps further optimizes the bill of materials, ensuring that the final price point remains competitive for large-scale API production.
- Enhanced Supply Chain Reliability: Synthetic manufacturing decouples production from seasonal or biological variables, guaranteeing a consistent and uninterrupted supply of critical intermediates. The ability to source starting materials from multiple global suppliers mitigates the risk of single-source bottlenecks, while the robustness of the chemical steps ensures high batch success rates. This reliability is crucial for maintaining the continuity of clinical trial material supply, preventing costly delays in drug development timelines that can arise from material shortages.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing unit operations that are standard in modern multipurpose chemical plants, facilitating a smooth transition from gram-scale laboratory synthesis to multi-ton commercial production. The waste streams generated, primarily consisting of inorganic salts and aqueous acidic washes, are well-characterized and can be treated using conventional effluent treatment protocols, ensuring compliance with stringent environmental regulations. This ease of scale-up combined with manageable environmental impact makes the technology an attractive option for contract development and manufacturing organizations (CDMOs) looking to expand their oncology portfolio.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these novel compounds, derived directly from the patent specifications and experimental data. These insights are intended to clarify the feasibility of the synthesis and the potential value proposition for pharmaceutical developers seeking new antitumor leads. Understanding these details is essential for making informed decisions about integrating this technology into your R&D pipeline.
Q: What distinguishes monohydroxy-2-acylphenylacetates from existing polyhydroxy analogs?
A: Unlike naturally occurring polyhydroxy derivatives which are difficult to isolate in pure forms, these monohydroxy variants offer a simplified structural scaffold that allows for precise tuning of lipophilicity and metabolic stability while retaining potent antitumor activity as demonstrated in MTT assays.
Q: Is the synthetic route suitable for large-scale manufacturing?
A: Yes, the process utilizes robust, commodity chemicals such as Meldrum's acid, aluminum chloride, and standard Grignard reagents. The avoidance of exotic catalysts and the use of standard workup procedures like column chromatography and crystallization make it highly amenable to commercial scale-up.
Q: How does the R-group variability impact the biological application?
A: The patent demonstrates that varying the R1 (ester) and R2 (acyl chain) groups significantly alters the dissociation constants and cell inhibition rates. This modularity allows medicinal chemists to optimize the structure-activity relationship (SAR) for specific cancer cell lines without redesigning the entire synthetic pathway.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Monohydroxy-2-acylphenylacetate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of having a dependable partner who can translate complex patent methodologies into reliable commercial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can grow seamlessly from early discovery to late-stage clinical supply. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of Monohydroxy-2-acylphenylacetate meets the exacting standards required for oncology drug development. Our commitment to quality assurance means that you can trust our materials to perform consistently in your biological assays, accelerating your path to identifying a viable drug candidate.
We invite you to engage with our technical procurement team to discuss how we can tailor this synthesis to your specific needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of how our optimized manufacturing processes can reduce your overall project costs. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our capability to be your long-term strategic partner in the development of next-generation antitumor therapies.
