Advanced Synthesis of Fall Webworm Sex Pheromone for Scalable Agrochemical Production
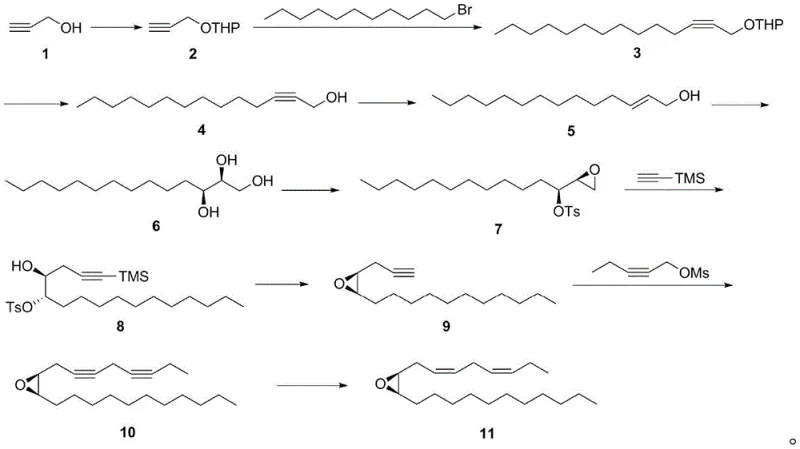
The global agricultural sector faces persistent challenges from invasive pests such as the fall webworm (Hyphantria cunea), which causes devastating damage to forestry and fruit crops across North America, Europe, and Asia. Traditional chemical control methods, while effective in the short term, often lead to environmental pollution and the development of pesticide resistance, necessitating the adoption of greener alternatives like sex pheromone trapping. Patent CN112661725B discloses a breakthrough synthetic method for producing (3Z, 6Z,9S, 10R)-9, 10-epoxy-3, 6-heneicosadiene, the primary sex pheromone component of the fall webworm. This innovative approach utilizes propiolic alcohol as a starting material and leverages Sharpless asymmetric dihydroxylation as a key stereocontrolling step, offering a robust alternative to previous methods that relied on unstable intermediates and hazardous reagents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this complex epoxy-diene pheromone has been plagued by significant chemical inefficiencies and safety concerns associated with prior art methodologies. Earlier routes, such as those described in Chinese patent CN 101798293A, relied on glycerol as a starting material but required a critical sulfoesterification step using trifluoromethyl sulfonic anhydride on a key epoxy-alcohol intermediate. This specific reagent is highly reactive and possesses strong oxidizing properties, which frequently triggers uncontrollable side reactions including elimination and nucleophilic substitution during the process. Consequently, manufacturers often struggle to isolate the desired trifluoromethyl sulfonate intermediate in high purity, leading to inconsistent batch quality and reduced overall yields that hinder commercial viability.
The Novel Approach
In stark contrast, the methodology outlined in CN112661725B circumvents these pitfalls by adopting a linear synthesis strategy that builds the carbon backbone before establishing the critical chiral centers. By initiating the synthesis with propiolic alcohol and employing a coupling reaction with 1-bromoundecane, the process constructs the necessary 21-carbon chain in a stable manner before introducing oxygen functionality. This strategic reversal of steps eliminates the need for the problematic triflic anhydride activation entirely, replacing it with a sequence of mild, high-yielding transformations that are far easier to manage in a production environment. The result is a streamlined 10-step protocol that achieves a total yield of approximately 10 percent with superior enantioselectivity, demonstrating a clear evolution in process chemistry design.
Mechanistic Insights into Sharpless Asymmetric Dihydroxylation and Epoxide Formation
The cornerstone of this synthetic achievement is the application of the Sharpless asymmetric dihydroxylation reaction in step 5, which serves as the primary engine for stereochemical induction. In this transformation, the internal alkene of the precursor compound 5 is treated with AD-mix-alpha at a controlled temperature of 0°C, facilitating the syn-addition of two hydroxyl groups across the double bond. The chiral ligand within the AD-mix-alpha complex directs the approach of the osmium tetroxide species to specifically generate the (9S, 10R) diol configuration, which is absolutely critical for the biological attraction activity of the final pheromone molecule. Following this, the diol is selectively monotosylated and subjected to intramolecular cyclization under basic conditions to form the 9,10-epoxide ring with inversion of configuration, preserving the high optical purity established in the previous step.
Furthermore, the final stages of the synthesis demonstrate precise control over alkene geometry through selective hydrogenation techniques. After coupling the epoxy-alcohol fragment with a protected alkyne unit to form the diyne precursor (compound 10), the molecule undergoes catalytic hydrogenation using a P-2 nickel catalyst or a Lindlar catalyst. These specific catalysts are chosen for their ability to reduce triple bonds to cis-double bonds (Z-alkenes) without over-reducing the molecule to the saturated alkane or causing isomerization to the thermodynamically more stable trans-form. This meticulous attention to geometric fidelity ensures that the final product matches the natural pheromone's (3Z, 6Z) configuration, maximizing its efficacy in field applications for pest monitoring and control.
How to Synthesize (3Z, 6Z,9S, 10R)-9, 10-epoxy-3, 6-heneicosadiene Efficiently
The execution of this synthesis requires careful attention to reaction conditions, particularly regarding temperature control and the exclusion of moisture during organometallic steps. The process begins with the protection of propiolic alcohol as a THP ether, followed by lithiation and coupling with 1-bromoundecane to extend the carbon chain, setting the stage for the subsequent stereochemical operations. Operators must ensure strict adherence to the specified molar ratios, such as the 1:1 ratio of 1-bromoundecane to the protected alkyne, to minimize homocoupling byproducts and maximize the efficiency of the carbon-carbon bond formation. Detailed standardized synthesis steps see the guide below.
- Protection of propiolic alcohol followed by coupling with 1-bromoundecane and subsequent deprotection to form the carbon skeleton.
- Execution of Sharpless asymmetric dihydroxylation using AD-mix-alpha to establish the (9S, 10R) chiral centers, followed by epoxide formation.
- Coupling with trimethylsilyl acetylene and 2-pentyne-1-ol mesylate, concluding with stereoselective hydrogenation to yield the final Z,Z-diene pheromone.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented methodology offers substantial advantages by fundamentally simplifying the reagent profile and reducing the reliance on hazardous specialty chemicals. The elimination of trifluoromethyl sulfonic anhydride not only lowers the direct cost of raw materials but also significantly reduces the logistical complexity and safety costs associated with handling and disposing of highly corrosive fluorinated waste streams. By shifting to a route based on commodity chemicals like propiolic alcohol and standard catalysts, manufacturers can secure a more stable and resilient supply chain that is less vulnerable to the price volatility often seen with exotic fluorinating agents. This stability translates into more predictable production schedules and lower inventory holding costs for critical reagents.
- Cost Reduction in Manufacturing: The removal of the sulfoesterification step using triflic anhydride drastically simplifies the purification workflow, as there is no longer a need to separate complex mixtures of elimination and substitution byproducts that typically plague the older method. This simplification leads to a significant reduction in solvent consumption and chromatography media usage, which are major cost drivers in fine chemical manufacturing. Additionally, the higher selectivity of the Sharpless dihydroxylation step minimizes the loss of valuable chiral intermediates, thereby improving the overall mass balance and reducing the cost per kilogram of the active pheromone ingredient.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials such as propiolic alcohol and 1-bromoundecane ensures that production is not bottlenecked by the scarcity of specialized precursors. Unlike methods requiring divinyl methanol or unstable polyacetylene intermediates which may have limited commercial suppliers, the reagents for this route are standard industrial chemicals with robust global supply networks. This accessibility allows for rapid scaling of production capacity to meet seasonal demand spikes for agrochemical products without the risk of raw material shortages disrupting the manufacturing timeline.
- Scalability and Environmental Compliance: The reaction conditions described in the patent are notably mild, with key steps proceeding at 0°C or room temperature, which reduces the energy load required for heating or deep cooling compared to cryogenic processes. The avoidance of heavy metal contaminants associated with certain alternative coupling methods, combined with the use of recyclable solvents like ethyl acetate and methanol, facilitates easier compliance with increasingly stringent environmental regulations. This green chemistry profile not only lowers waste treatment costs but also enhances the marketability of the final product to eco-conscious agricultural stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic route for fall webworm pheromone production. These answers are derived directly from the experimental data and beneficial effects reported in patent CN112661725B, providing clarity on the process's robustness and advantages over legacy technologies. Understanding these details is essential for technical teams evaluating the feasibility of adopting this method for commercial manufacturing.
Q: Why does this new method avoid trifluoromethyl sulfonic anhydride?
A: The conventional method using trifluoromethyl sulfonic anhydride for sulfoesterification of the epoxy-alcohol intermediate is prone to severe side reactions such as elimination and substitution, making the process difficult to control and lowering yields. The new method bypasses this hazardous step entirely by constructing the carbon chain first and introducing chirality via Sharpless dihydroxylation, resulting in a milder and more controllable reaction profile.
Q: What is the key step for ensuring optical purity in this synthesis?
A: The critical step for stereocontrol is the Sharpless asymmetric dihydroxylation performed in step 5 using AD-mix-alpha. This reaction selectively hydroxylates the alkene to generate the (9S, 10R) diol configuration with high enantioselectivity (95% e.e.), which is subsequently converted into the epoxide ring without racemization, ensuring the biological efficacy of the final pheromone.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the route is designed for scalability by utilizing readily available starting materials like propiolic alcohol and avoiding unstable polyacetylene intermediates found in older methods. The reaction conditions are generally mild, with key steps occurring at 0°C or room temperature, and the final hydrogenation uses standard P-2 nickel or Lindlar catalysts, facilitating safe and efficient commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (3Z, 6Z,9S, 10R)-9, 10-epoxy-3, 6-heneicosadiene Supplier
As the demand for sustainable pest management solutions grows, the ability to produce high-purity pheromone intermediates efficiently becomes a critical competitive advantage for agrochemical companies. NINGBO INNO PHARMCHEM stands ready to support your development goals with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the sensitive organometallic and asymmetric catalysis steps required by this patent, ensuring that every batch meets stringent purity specifications through our rigorous QC labs and advanced analytical capabilities.
We invite you to collaborate with us to leverage this advanced synthetic technology for your product pipeline. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing expertise can optimize your supply chain for this vital agrochemical intermediate.
