Advanced Synthesis of Chloro-benzothiepin Intermediates for Scalable Pharmaceutical Manufacturing
The pharmaceutical industry continuously seeks robust synthetic routes for complex heterocyclic intermediates that balance high purity with manufacturing efficiency. Patent CN111620838A introduces a groundbreaking preparation method for chloro-benzothiepin compounds, specifically targeting the synthesis of 6-chloro-2,3,4,5-tetrahydro-1,4-benzothiazepine-1,1-dioxide. This specific molecular architecture is critical for developing next-generation antiviral agents, particularly those targeting Respiratory Syncytial Virus (RSV). The innovation lies in the strategic introduction of the chlorine element at the early stages of synthesis using 2-chloro-6-fluoro-benzaldehyde as a starting material. This approach not only streamlines the reaction pathway but also ensures that the halogen functionality is preserved throughout the rigorous chemical transformations required to build the benzothiazepine core. For R&D directors and process chemists, this represents a significant leap forward in accessing high-value scaffolds that were previously difficult to functionalize with high regioselectivity.
Furthermore, the technical implications of this patent extend beyond mere molecular construction; it addresses the fundamental challenges of process chemistry in a commercial setting. The methodology employs a series of well-controlled steps including sulfo-group introduction, aldehyde-amine condensation, and selective oxidation, all optimized to minimize byproduct formation. By leveraging cesium carbonate as a base and m-chloroperoxybenzoic acid as a selective oxidant, the process achieves a level of chemical precision that translates directly into improved impurity profiles. This is paramount for pharmaceutical intermediates where downstream purification costs can erode profit margins. The ability to produce such complex structures with consistent quality positions this technology as a cornerstone for reliable pharmaceutical intermediate supplier networks aiming to support global drug development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for benzothiazepine derivatives often suffer from severe limitations regarding reaction conditions and functional group tolerance. Many conventional methods rely on harsh thermal conditions or aggressive reagents that can compromise the integrity of sensitive halogen substituents on the aromatic ring. When chlorine atoms are introduced late in the synthesis, there is a heightened risk of dehalogenation or unwanted side reactions, leading to complex impurity spectra that are costly to remove. Additionally, older protocols frequently utilize strong bases or acids that require specialized equipment and stringent safety measures, increasing the overall capital expenditure for manufacturing facilities. The lack of mild, scalable options has historically constrained the availability of high-purity chloro-benzothiepin derivatives, creating bottlenecks in the supply chain for antiviral drug manufacturers who require consistent material for clinical and commercial batches.
The Novel Approach
In stark contrast, the novel approach detailed in the patent data utilizes a convergent strategy that introduces the chlorine atom at the very beginning using 2-chloro-6-fluoro-benzaldehyde. This strategic decision ensures that the halogen is securely integrated into the molecular framework before the more vigorous cyclization and oxidation steps occur. The process operates under remarkably mild conditions, with key steps such as reduction and oxidation proceeding efficiently at room temperature. This eliminates the need for energy-intensive heating or cooling systems, drastically simplifying the engineering requirements for scale-up. Moreover, the use of specific protecting groups like the tert-butyloxycarbonyl (Boc) group allows for precise control over reactivity, preventing premature side reactions and ensuring that the final deprotection step yields the target compound with exceptional cleanliness. This modern methodology effectively resolves the historical pain points of yield loss and impurity generation associated with legacy synthesis routes.
Mechanistic Insights into Cs2CO3-Mediated Cyclization and Oxidation
The core of this synthetic success lies in the meticulous optimization of the catalytic and stoichiometric reagents used throughout the six-step sequence. The initial thioetherification step leverages cesium carbonate in dimethylformamide (DMF) at 70°C to facilitate the nucleophilic attack of the thiol on the fluorinated aldehyde. Cesium carbonate is chosen for its moderate alkalinity and solubility profile, which promotes the reaction without causing excessive decomposition of the starting materials. Following this, the aldehyde-amine condensation is triggered by an acidic reagent, specifically a 4 M HCl/dioxane solution, which catalyzes the ring closure to form the dihydro-benzothiazepine core. The subsequent reduction using sodium borohydride is carefully controlled to reduce the imine bond without affecting other sensitive functionalities. Each reagent is selected not just for reactivity but for its compatibility with the overall process flow, ensuring a seamless transition between intermediates.
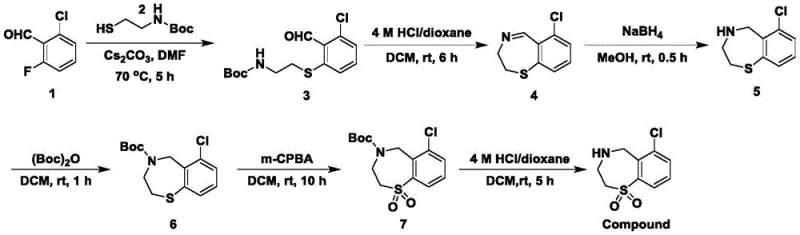
The oxidation step represents a critical juncture in the mechanism, where the sulfur atom within the heterocyclic ring is converted to a sulfone. This transformation is achieved using m-chloroperoxybenzoic acid (m-CPBA) at room temperature, a condition that prevents thermal degradation of the organic scaffold. The mechanism involves the transfer of oxygen atoms to the sulfur center, a process that must be strictly monitored to avoid over-oxidation or damage to the chlorine substituent. The final deprotection step utilizes acidolysis to remove the Boc group, regenerating the free amine necessary for biological activity. Throughout this cascade, the impurity control mechanism is inherent in the choice of reagents; for instance, the use of sodium bicarbonate during workup neutralizes residual acids without introducing volatile ammonia, thereby maintaining a cleaner reaction environment. This attention to mechanistic detail ensures that the final product meets the rigorous purity specifications demanded by regulatory bodies for pharmaceutical applications.
How to Synthesize 6-chloro-2,3,4,5-tetrahydro-1,4-benzothiazepine-1,1-dioxide Efficiently
To implement this synthesis effectively, process engineers must adhere to the specific molar ratios and temperature controls outlined in the patent examples. The procedure begins with the precise weighing of 2-chloro-6-fluoro-benzaldehyde and 2-tert-butyloxycarbonylaminoethanethiol, followed by reaction in DMF with cesium carbonate. Detailed standardized synthesis steps are provided below to ensure reproducibility and safety during scale-up operations. Adhering to these parameters is essential for maintaining the high yields and purity levels observed in the experimental data, particularly when transitioning from laboratory glassware to industrial reactors.
- Perform thioetherification of 2-chloro-6-fluoro-benzaldehyde with 2-tert-butyloxycarbonylaminoethanethiol using cesium carbonate in DMF at 70°C.
- Execute aldehyde-amine condensation under acidic conditions to form the dihydro-benzothiazepine ring structure.
- Reduce the imine bond using sodium borohydride, protect the amine, oxidize the sulfur to sulfone using m-CPBA, and finally deprotect to yield the final product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this synthesis method offers transformative benefits that directly impact the bottom line and operational resilience. The shift towards room-temperature reactions for the majority of the synthetic steps significantly lowers energy consumption, which is a major cost driver in fine chemical manufacturing. By eliminating the need for extreme heating or cryogenic cooling, facilities can reduce their utility overheads and minimize the wear and tear on processing equipment. Furthermore, the use of commercially available raw materials such as 2-chloro-6-fluoro-benzaldehyde ensures a stable supply chain, reducing the risk of production delays caused by scarce reagents. The streamlined workflow also shortens the overall cycle time, allowing for faster turnover of batches and improved responsiveness to market demand fluctuations without compromising on quality standards.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the reliance on common organic reagents like sodium borohydride and m-CPBA leads to substantial cost savings in raw material procurement. Additionally, the mild reaction conditions reduce the need for specialized high-pressure or high-temperature vessels, lowering capital investment requirements for new production lines. The efficient workup procedures, which avoid the use of hazardous ammonia water, also reduce waste disposal costs and simplify environmental compliance reporting. These factors combine to create a leaner, more cost-effective manufacturing process that enhances the competitiveness of the final pharmaceutical intermediate in the global market.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures consistent output even when scaling from pilot plants to full commercial production. The use of stable intermediates and straightforward purification techniques, such as silica gel column chromatography and crystallization, minimizes the risk of batch failures. This reliability is crucial for supply chain heads who must guarantee continuous delivery to downstream drug manufacturers. By adopting this method, suppliers can offer more predictable lead times and maintain higher inventory levels of critical intermediates, thereby strengthening the overall resilience of the pharmaceutical supply network against external disruptions.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, with each step demonstrating high conversion rates and minimal byproduct formation. The replacement of volatile and toxic reagents with safer alternatives aligns with modern green chemistry principles, making it easier to obtain environmental permits and maintain sustainable operations. The reduced generation of hazardous waste lowers the environmental footprint of the manufacturing site, which is increasingly important for corporate social responsibility goals. This alignment with environmental standards not only mitigates regulatory risks but also enhances the brand reputation of the manufacturer as a responsible partner in the global healthcare ecosystem.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this chloro-benzothiepin compound. These answers are derived directly from the patent specifications and experimental data to provide accurate guidance for potential partners. Understanding these details is essential for evaluating the feasibility of integrating this intermediate into your specific drug development pipeline.
Q: What are the key advantages of this chloro-benzothiepin synthesis method?
A: The method utilizes mild reaction conditions, primarily at room temperature, which significantly reduces energy consumption and operational complexity compared to traditional high-temperature processes.
Q: How does this process improve environmental compliance?
A: By replacing ammonia water with weak alkaline sodium bicarbonate for neutralization, the process eliminates pungent odors and hazardous volatile emissions, enhancing workplace safety and environmental standards.
Q: Is this intermediate suitable for large-scale antiviral drug production?
A: Yes, the robust yield and scalable room-temperature steps make it highly suitable for commercial scale-up in the manufacturing of active pharmaceutical ingredients for RSV treatment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6-chloro-2,3,4,5-tetrahydro-1,4-benzothiazepine-1,1-dioxide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of life-saving medications. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and efficiency. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which employ advanced analytical techniques to verify every batch. Our infrastructure is designed to handle complex chemistries safely, providing you with a secure and dependable source for your pharmaceutical raw materials.
We invite you to collaborate with us to leverage this advanced synthesis technology for your upcoming projects. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can accelerate your time to market. Let us be your partner in turning innovative chemical designs into commercial realities.
