Revolutionizing Vorinostat Production: A Deep Dive into One-Pot Mesoporous Catalysis
Revolutionizing Vorinostat Production: A Deep Dive into One-Pot Mesoporous Catalysis
The pharmaceutical industry continuously seeks robust synthetic pathways that balance high purity with operational efficiency, particularly for oncology therapeutics like Vorinostat (Suberoylanilide hydroxamic acid). A pivotal advancement in this domain is detailed in patent CN109096148B, which discloses a sophisticated one-pot preparation method utilizing a modified mesoporous silica coupling and catalysis system. This technology represents a significant departure from conventional multi-step syntheses, addressing critical pain points regarding yield, purity, and process complexity. By employing a specially engineered hydrophilic modified mesoporous silica, the process achieves selective activation of suberic acid without the need for isolating unstable intermediates. This innovation not only streamlines the workflow but also ensures the final product meets stringent pharmaceutical standards, making it a highly attractive route for reliable vorinostat suppliers aiming to optimize their manufacturing portfolios.
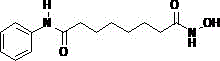
The structural integrity and functional group compatibility of Vorinostat require precise control during synthesis to avoid degradation or side reactions. The approach outlined in CN109096148B leverages the unique surface properties of mesoporous materials to create a microenvironment that favors the desired amidation and hydroxamation reactions. This method effectively mitigates the challenges associated with the dual carboxyl groups of suberic acid, which often lead to non-selective polymerization or anhydride formation in traditional protocols. For R&D directors and process chemists, understanding this mechanistic nuance is crucial for evaluating the scalability and reproducibility of the technology in a commercial setting.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Vorinostat has been plagued by inefficient multi-step sequences that suffer from low overall yields and difficult purification requirements. For instance, prior art such as WO 2006/127319 describes a route involving the amidation of suberic acid followed by esterification and subsequent reaction with hydroxylamine, a process that typically results in a total yield of less than 25%. Furthermore, alternative methods reported in literature often necessitate harsh reaction conditions, such as temperatures reaching 190°C, which not only increase energy costs but also promote the formation of thermal degradation impurities. The reliance on soluble coupling agents like DCC or CDI in traditional approaches generates substantial amounts of urea by-products that are notoriously difficult to remove completely, thereby compromising the purity profile of the final API. These inefficiencies create significant bottlenecks for cost reduction in HDAC inhibitor manufacturing, as extensive chromatographic purification or recrystallization steps are invariably required to meet regulatory specifications.
The Novel Approach
In stark contrast, the novel one-pot method disclosed in the patent utilizes a modified mesoporous silica system that functions simultaneously as a coupling agent and a heterogeneous catalyst. This innovative strategy allows for the direct conversion of suberic acid to Vorinostat in a single reactor vessel, eliminating the need to isolate the intermediate N-hydroxy-7-carboxyl-heptanamide. The process operates under mild conditions, typically between 25°C and 30°C, which drastically reduces energy consumption and thermal stress on the reactants. By integrating the activation and coupling steps, the method simplifies the post-treatment workflow; the solid catalyst can be removed via simple filtration, leaving a filtrate that requires minimal downstream processing. This streamlined approach not only enhances the molar yield to over 80% but also ensures a purity profile exceeding 99.7%, demonstrating a clear superiority over legacy techniques that struggle with selectivity and impurity control.
Mechanistic Insights into Modified Mesoporous Silica Catalysis
The core of this technological breakthrough lies in the precise engineering of the catalyst surface. The mesoporous silica is modified using suberic anhydride and a coupling agent such as EDC (1-ethyl-3-(3-dimethylaminopropyl) carbodiimide) in the presence of KH-550. This modification creates a hydrophilic surface enriched with active sites that mimic the function of soluble coupling reagents but remain anchored to the solid support. When suberic acid is introduced into the THF/DMF mixed solvent system, it interacts with these activated sites to form a reactive intermediate that is spatially constrained within the mesopores. This confinement effect enhances the selectivity of the reaction, preventing the intermolecular condensation that leads to polymeric anhydrides. The addition of a reaction promoter, specifically B-halogenated o-phenylenedioxyborane, further activates the hydroxylamine species, facilitating the nucleophilic attack on the activated carboxyl group. This synergistic interaction between the solid support and the solution-phase reagents is key to achieving high conversion rates without the accumulation of toxic or difficult-to-remove by-products.
From an impurity control perspective, the heterogeneous nature of the catalyst provides a distinct advantage. In homogeneous catalysis, residual metal ions or organic coupling agents often persist in the final product, requiring rigorous scavenging steps. However, with the modified mesoporous silica, the catalyst is physically separated from the product mixture upon completion of the reaction. The patent data indicates that this physical separation, combined with the mild pH adjustments (pH 10-12) used to drive the final aniline coupling step, results in a crude product with exceptionally high purity. The absence of competing side reactions, such as the formation of octanedioic anhydride, is attributed to the steric and electronic environment provided by the modified silica surface. For quality assurance teams, this implies a more consistent impurity profile batch-to-batch, reducing the risk of out-of-specification results during commercial production.
How to Synthesize Vorinostat Efficiently
The implementation of this one-pot synthesis route requires careful attention to the preparation of the catalyst and the sequential addition of reagents to maintain reaction kinetics. The process begins with the pre-treatment of suberic acid using the hydrophilic modified mesoporous silica in a specific solvent ratio of THF to DMF, which ensures optimal solubility and interaction with the catalyst surface. Following this activation phase, hydroxylamine hydrochloride and the boron-based promoter are introduced to generate the hydroxamic acid moiety. The final step involves a pH adjustment to facilitate the coupling with aniline, completing the molecular assembly of Vorinostat. Detailed standardized operating procedures for scaling this chemistry from laboratory to pilot plant are critical for ensuring reproducibility and safety.
- Prepare the hydrophilic modified mesoporous silica catalyst by reacting suberic anhydride and EDC with KH-550, followed by dispersion and modification of silica particles.
- Pre-treat suberic acid with the modified catalyst in a THF/DMF mixed solvent system to activate the carboxyl groups selectively.
- Add hydroxylamine hydrochloride and a boron-based promoter, stir, then adjust pH to 10-12 and add aniline to complete the one-pot conversion to Vorinostat.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this one-pot mesoporous catalysis method offers transformative benefits regarding cost structure and operational reliability. The elimination of intermediate isolation steps translates directly into reduced solvent usage, lower waste disposal costs, and decreased equipment occupancy time. Traditional multi-step syntheses often require dedicated reactors for each transformation, along with extensive filtration and drying units for intermediate handling. By consolidating these operations, the new method significantly lowers the capital expenditure required for production capacity and reduces the variable costs associated with utilities and labor. Furthermore, the ability to recycle the mesoporous silica catalyst adds a layer of sustainability and long-term cost efficiency, as the expensive catalytic material does not need to be replenished for every batch. This aligns perfectly with the goals of cost reduction in HDAC inhibitor manufacturing, allowing for more competitive pricing strategies in the generic pharmaceutical market.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the removal of stoichiometric soluble coupling agents like DCC or CDI, which are expensive and generate significant solid waste (dicyclohexylurea). By replacing these with a reusable heterogeneous catalyst, the material cost per kilogram of Vorinostat is drastically reduced. Additionally, the simplified work-up procedure, which avoids complex chromatographic purifications, lowers the consumption of silica gel and elution solvents. The overall process efficiency means that fewer man-hours are spent on monitoring and handling, leading to substantial operational savings that enhance the margin profile for commercial scale-up of complex histone deacetylase inhibitors.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the availability of specialized reagents and the complexity of multi-step logistics. This one-pot method relies on readily available bulk chemicals such as suberic acid, aniline, and hydroxylamine, reducing dependency on niche suppliers. The robustness of the reaction conditions, which tolerate mild temperatures and standard pressure, minimizes the risk of batch failures due to equipment malfunction or thermal runaway. Consequently, lead times for high-purity Vorinostat batches can be shortened, ensuring a steady flow of material to downstream formulation partners. The predictability of the yield, consistently exceeding 80% in experimental data, allows for more accurate demand forecasting and inventory management.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges regarding heat transfer and mixing, but the heterogeneous nature of this catalysis system facilitates easier scale-up. The solid catalyst can be managed using standard filtration equipment available in most GMP facilities, avoiding the need for specialized high-pressure or cryogenic infrastructure. From an environmental standpoint, the reduction in solvent volume and the elimination of toxic urea by-products simplify wastewater treatment protocols. This compliance with green chemistry principles not only reduces regulatory burdens but also enhances the corporate sustainability profile, a factor increasingly weighted by global pharmaceutical buyers when selecting a reliable vorinostat supplier.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. Understanding these details is essential for stakeholders evaluating the feasibility of adopting this technology for large-scale production. The answers are derived directly from the experimental data and technical disclosures found in the patent literature, ensuring accuracy and relevance for decision-makers.
Q: How does the modified mesoporous silica improve yield compared to traditional coupling agents?
A: The modified mesoporous silica acts as both a coupling agent and a heterogeneous catalyst, eliminating the need for stoichiometric amounts of soluble coupling reagents like DCC or CDI. This dual functionality enhances selectivity for the mono-amide intermediate and prevents the formation of symmetrical anhydride by-products, boosting yields from typical lows of 25% to over 80%.
Q: What are the specific advantages of the one-pot method for supply chain stability?
A: By avoiding the isolation and purification of intermediate products, the one-pot method significantly reduces solvent consumption, processing time, and equipment occupancy. This consolidation of steps minimizes material loss during transfer and filtration, leading to a more robust and continuous manufacturing process suitable for large-scale supply.
Q: Is the catalyst reusable in this Vorinostat synthesis process?
A: Yes, the patent explicitly states that the mesoporous silica catalyst can be regenerated and recycled. After the reaction, the solid catalyst is removed by filtration, and adsorbates can be cleared to restore activity, which contributes to long-term cost efficiency and waste reduction.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Vorinostat Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the evolving demands of the global pharmaceutical market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the mesoporous silica-catalyzed one-pot synthesis can be seamlessly transferred from the lab to the plant. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of Vorinostat meets the highest international standards. Our infrastructure is designed to handle complex chemistries with precision, providing our partners with a secure and compliant source of high-quality oncology intermediates.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this efficient manufacturing process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project timelines. Let us collaborate to drive efficiency and quality in the production of life-saving medications.
