Advanced Saxagliptin Manufacturing: A 3-Step Route for High-Purity API Production
Advanced Saxagliptin Manufacturing: A 3-Step Route for High-Purity API Production
The pharmaceutical industry constantly seeks more efficient pathways for the production of critical antidiabetic agents, and the synthesis of Saxagliptin stands as a prime example of process innovation. Patent CN111205216A discloses a groundbreaking method that fundamentally restructures the synthetic landscape for this potent DPP-IV inhibitor. By strategically reordering reaction sequences and selecting superior reagents, this technology transforms a traditionally cumbersome four-step protocol into a streamlined three-step operation. This advancement is not merely an academic exercise but a robust industrial solution that addresses long-standing pain points in impurity control and operational complexity. For global supply chain leaders, this represents a tangible opportunity to enhance the reliability of their active pharmaceutical ingredient (API) sourcing while simultaneously driving down manufacturing overheads through simplified processing.
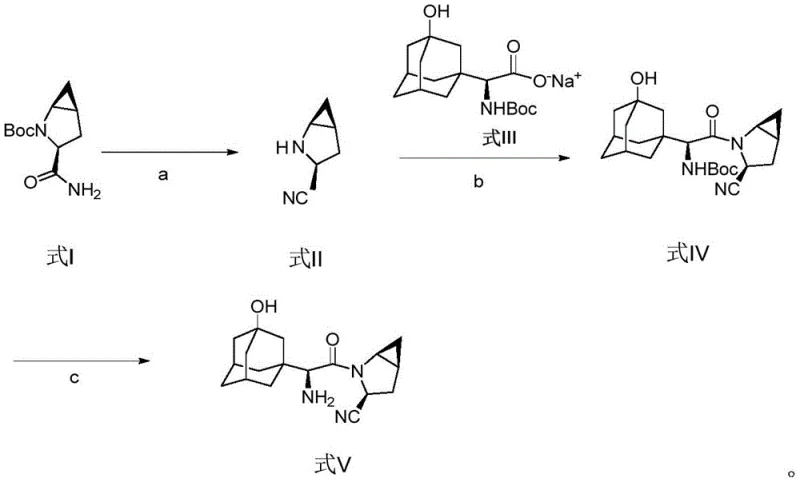
The core innovation lies in the clever integration of the cyanation and deprotection stages, which were historically distinct and resource-intensive operations. In the conventional background art, the synthesis often relied on methanesulfonic acid for salt formation followed by separate condensation and dehydration steps using aggressive reagents. The new methodology described in the patent leverages aryl sulfonyl chlorides to effectuate the conversion of the primary amide to a nitrile group while concurrently stripping the tert-butyloxycarbonyl (Boc) protecting group. This dual-functionality step significantly reduces the number of unit operations required, thereby minimizing material handling and potential yield losses associated with multiple isolation stages. Such process intensification is critical for maintaining high throughput in modern cGMP facilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of Saxagliptin has been plagued by inefficiencies inherent in the traditional four-step synthetic route. As detailed in prior art documents such as Org. Process Res. Dev. 2009, the legacy process necessitates an initial salification step using methanesulfonic acid to remove the Boc group, followed by a condensation reaction. Subsequently, a dehydration reaction is required to convert the amide to a nitrile, typically employing trifluoroacetic anhydride (TFAA) and ethyl nicotinate. This specific combination of reagents introduces severe downstream processing challenges. Ethyl nicotinate, while effective as a scavenger or activator, is notoriously difficult to remove completely from the reaction mixture, often requiring repeated extractions with dilute hydrochloric acid and water washes. Furthermore, the use of TFAA can lead to unwanted side reactions, such as the esterification of hydroxyl groups present on the adamantane moiety or premature cleavage of other protecting groups. These side reactions generate complex impurity profiles that are costly and time-consuming to purge, ultimately compromising the overall yield and purity of the final API.
The Novel Approach
In stark contrast, the novel approach outlined in patent CN111205216A offers a paradigm shift by collapsing the synthetic sequence into three highly efficient steps. The most significant breakthrough is the first step, where the starting material (Formula I) undergoes concurrent cyanation and Boc-deprotection to yield the key intermediate (Formula II). This is achieved using benzenesulfonyl chloride and pyridine at room temperature, followed by a straightforward acidification workup. This eliminates the need for the problematic ethyl nicotinate and TFAA entirely. The subsequent condensation step couples this intermediate with the adamantane derivative (Formula III) under mild conditions using standard peptide coupling agents. Finally, a single acidic deprotection yields the target Saxagliptin (Formula V). This route not only simplifies the workflow but also operates under much milder conditions, with key reactions proceeding effectively at ambient temperatures, thereby reducing the thermal load on the manufacturing plant.
Mechanistic Insights into Aryl Sulfonyl Chloride Mediated Dehydration
The mechanistic elegance of this new process centers on the activation of the primary amide using aryl sulfonyl chloride. In the presence of a nucleophilic organic base like pyridine, the sulfonyl chloride reacts with the amide nitrogen to form a reactive sulfonimidate or similar activated intermediate. This activation renders the carbonyl carbon highly electrophilic and facilitates the elimination of water to form the nitrile triple bond. Crucially, the reaction conditions are tuned such that the acid-labile Boc group on the azabicyclo ring is also cleaved, either during the reaction or immediately upon the addition of the hydrochloric acid workup solution. This tandem transformation is chemically sophisticated yet operationally simple, avoiding the need for cryogenic temperatures or hazardous dehydrating agents like phosphorus oxychloride. The use of benzenesulfonyl chloride ensures that the byproduct formed is a benign sulfonamide or sulfonic acid derivative, which is easily separated during the aqueous extraction phases, thus ensuring the high purity of the resulting aminonitrile intermediate.
Furthermore, the condensation step (Step b) utilizes robust peptide coupling chemistry, employing agents such as EDC.HCl-HOBt or HBTU. The patent specifies that dissolving the adamantane acid derivative (Formula III) first and activating it with the coupling agent before adding the aminonitrile (Formula II) is a critical procedural nuance. This order of addition minimizes the self-polymerization of the aminonitrile and ensures that the activated ester species is ready to react immediately upon contact with the nucleophile. This kinetic control is vital for suppressing the formation of dimeric or oligomeric impurities. The final deprotection step utilizes standard mineral acids like hydrochloric acid in an alcohol-water mixture. The moderate heating to 55-65°C ensures complete removal of the remaining Boc group on the adamantane side chain without degrading the sensitive nitrile functionality or the strained azabicyclo ring system, resulting in a final product with exceptional chemical integrity.
How to Synthesize Saxagliptin Efficiently
The implementation of this synthesis route requires precise control over stoichiometry and addition sequences to maximize the benefits of the novel chemistry. The process begins with the dissolution of the Boc-protected amide in a solvent such as ethyl acetate or tetrahydrofuran, followed by the controlled addition of pyridine and benzenesulfonyl chloride. Maintaining the reaction at room temperature for 3 to 5 hours allows for complete conversion before the acidic quench. The subsequent isolation of the intermediate involves a unique partition between hydrochloric acid and diethyl ether, which effectively precipitates the product as a salt or free base depending on the pH manipulation. For the coupling stage, the choice of solvent system, often a mixture of acetonitrile and ethyl acetate, plays a pivotal role in solubility and reaction rate. Detailed standardized operating procedures for scaling this specific pathway from laboratory benchtop to commercial manufacturing are provided in the structured guide below.
- Convert the amide group of the Boc-protected starting material into a cyano group while simultaneously removing the Boc protecting group using aryl sulfonyl chloride and organic base.
- Condense the resulting aminonitrile intermediate with the adamantane amino acid derivative using a peptide coupling agent like EDC.HCl-HOBt or HBTU.
- Perform the final acidic deprotection of the Boc group on the coupled intermediate to yield the target Saxagliptin API.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route translates directly into enhanced operational resilience and cost efficiency. The primary economic driver is the drastic simplification of the purification train. By eliminating the use of ethyl nicotinate and trifluoroacetic anhydride, the manufacturer avoids the expensive and time-consuming extraction cycles previously required to remove these stubborn residues. This reduction in downstream processing time leads to shorter batch cycle times and increased facility throughput. Moreover, the shift to room temperature reactions for the cyanation and condensation steps significantly lowers utility costs associated with heating and cooling, contributing to a leaner manufacturing cost structure. The use of commodity chemicals like benzenesulfonyl chloride and pyridine, which are widely available and cost-effective, further stabilizes the raw material supply chain against market volatility.
- Cost Reduction in Manufacturing: The elimination of complex purification steps and the reduction in total process steps from four to three inherently lower the cost of goods sold (COGS). By avoiding the generation of difficult-to-remove byproducts, the yield loss associated with extensive washing and recrystallization is minimized. The process relies on readily available reagents rather than specialized or hazardous dehydrating agents, which reduces raw material procurement costs. Additionally, the simplified workflow requires less labor and fewer man-hours per batch, allowing for more competitive pricing strategies in the global API market without compromising margin.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures a more consistent supply of high-quality Saxagliptin intermediates and API. The mild reaction conditions reduce the risk of batch failures due to thermal runaway or equipment limitations, which is a common concern with exothermic dehydration reactions using TFAA. The reliance on stable, shelf-stable reagents like benzenesulfonyl chloride mitigates the risk of supply disruptions caused by the limited availability of niche catalysts. This reliability allows pharmaceutical partners to maintain tighter inventory controls and reduces the need for excessive safety stock, optimizing working capital across the supply network.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this process offers substantial advantages for large-scale production. The avoidance of fluorinated reagents like TFAA reduces the generation of fluorinated waste streams, simplifying wastewater treatment and lowering disposal costs. The ability to run key steps at room temperature reduces the carbon footprint of the manufacturing process by minimizing energy consumption. Furthermore, the high purity of the crude product reduces the solvent volume required for final purification, aligning with green chemistry principles and facilitating smoother regulatory audits and environmental compliance reporting for commercial scale-up.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Saxagliptin synthesis technology. These insights are derived directly from the experimental data and process descriptions within the patent documentation, providing clarity on the practical aspects of adoption. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into existing production lines or for procurement teams assessing the quality implications of the new method.
Q: How does the new saxagliptin synthesis route improve upon conventional methods?
A: The novel route shortens the process from four steps to three by combining cyanation and deprotection. It eliminates the use of difficult-to-remove reagents like ethyl nicotinate and avoids harsh trifluoroacetic anhydride conditions, leading to higher purity and easier industrial operation.
Q: What are the key reagents used in the optimized cyanation step?
A: The optimized process utilizes aryl sulfonyl chloride, specifically benzenesulfonyl chloride, in the presence of an organic base such as pyridine. This is followed by an acidification step using hydrochloric acid and diethyl ether to isolate the intermediate.
Q: Is this manufacturing process suitable for large-scale commercial production?
A: Yes, the process is designed for scalability. Key reactions such as cyanation and condensation occur at room temperature, reducing energy consumption. The simplified workup procedures and avoidance of complex extractions make it highly suitable for metric-ton scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Saxagliptin Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to a more efficient synthetic route requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in practice. Our state-of-the-art facilities are equipped to handle the specific solvent systems and reagent handling requirements of this process, while our stringent purity specifications and rigorous QC labs guarantee that every batch meets the highest pharmacopeial standards. We are committed to delivering Saxagliptin and its advanced intermediates with the consistency and quality required by top-tier pharmaceutical companies.
We invite you to collaborate with us to leverage this innovative technology for your supply chain. Our team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this 3-step route can optimize your budget. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a sustainable and cost-effective supply of high-purity Saxagliptin for your global markets.
