Advanced Synthesis of 2-Amino-3,5-Dichloro-N-Methylbenzamide for Scalable Agrochemical Production
The pharmaceutical and agrochemical industries are constantly seeking more efficient pathways to synthesize critical intermediates that drive the production of next-generation active ingredients. A pivotal development in this domain is documented in patent CN112142620A, which outlines a novel synthetic method for 2-amino-3,5-dichloro-N-methylbenzamide, a key precursor for bisamide pesticides such as tetrachlorantraniliprole. This specific intermediate serves as the structural backbone for potent insecticides that exhibit activity comparable to chlorantraniliprole but often require more streamlined manufacturing processes to be commercially viable. The innovation presented in this patent addresses long-standing challenges regarding cost, purity, and environmental impact, marking a significant shift away from traditional, less efficient chlorination protocols. For global supply chain leaders and R&D directors, understanding this technological leap is essential for securing a competitive edge in the agrochemical market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-amino-3,5-dichloro-N-methylbenzamide has been plagued by economic and technical inefficiencies that hinder large-scale adoption. Prior art, such as the methods disclosed in patent CN101298451B, relied on starting materials like 2-amino-N-methylbenzamide combined with N-chlorosuccinimide (NCS) for chlorination. This approach suffers from prohibitively high raw material costs, as both the starting amide and the chlorinating agent are expensive specialty chemicals. Furthermore, alternative routes utilizing dichlorohydantoin (DCDMH) often result in incomplete reactions, leaving behind significant quantities of monochlorinated impurities that are notoriously difficult to remove through standard recrystallization techniques. These legacy processes not only generate substantial solid waste in the form of hydantoin byproducts but also struggle to achieve the high purity levels required for modern pesticide registration, creating a bottleneck for manufacturers aiming to reduce their cost of goods sold.
The Novel Approach
In stark contrast, the methodology described in CN112142620A introduces a robust one-pot synthesis strategy that fundamentally restructures the production workflow to maximize efficiency and minimize waste. By utilizing isatoic anhydride as the starting material, which is cheap and readily available on the industrial market, the process bypasses the need for expensive pre-functionalized amines. The core innovation lies in the substitution of traditional chlorinating agents with trichloroisocyanuric acid (TCCA), a reagent known for its superior reactivity and economic profile. This novel approach ensures that the chlorination proceeds to completion, effectively eliminating the persistent monochlorinated impurities that plague older methods. Additionally, the unique chemical properties of the TCCA byproduct allow for a simplified workup procedure where impurities are dissolved away in an aqueous phase, leaving behind the target product with exceptional purity and yield, thereby setting a new standard for cost reduction in agrochemical manufacturing.
Mechanistic Insights into TCCA-Mediated Electrophilic Chlorination
The chemical elegance of this synthesis lies in the dual-functionality of the reaction sequence, beginning with a nucleophilic ring-opening followed by a highly selective electrophilic aromatic substitution. In the first stage, isatoic anhydride undergoes nucleophilic attack by methylamine in a solvent system such as ethyl acetate, typically maintained between 10°C and 50°C. This step efficiently generates the intermediate 2-amino-N-methylbenzamide in situ. The subsequent addition of trichloroisocyanuric acid initiates the chlorination phase, where the active chlorine species generated from TCCA act as powerful electrophiles. Unlike less reactive agents like DCDMH, TCCA provides sufficient driving force to overcome the deactivating effects of the initial chlorine substitution, ensuring that the reaction proceeds past the monochloro stage to the desired 3,5-dichloro configuration. This mechanistic advantage is critical for preventing the accumulation of partially chlorinated side products that would otherwise compromise the quality of the final agrochemical intermediate.
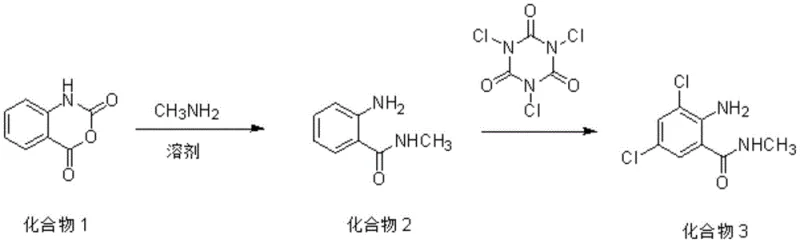
Beyond the reaction kinetics, the purification mechanism employed in this patent represents a significant advancement in process chemistry design. Upon completion of the chlorination, the reaction mixture contains the target dichloro-benzamide alongside isocyanuric acid, the reduced form of the chlorinating agent. The brilliance of this system is revealed during the workup phase: when the pH of the aqueous mixture is adjusted to an alkaline range of 8 to 13, the isocyanuric acid converts into a highly water-soluble salt. In contrast, the target product, 2-amino-3,5-dichloro-N-methylbenzamide, remains insoluble in the aqueous medium due to its lipophilic character. This differential solubility allows for a straightforward separation via suction filtration, where the pure white solid is retained while the soluble byproducts are washed away. This eliminates the need for complex chromatographic purification or multiple recrystallization steps, directly contributing to the high purity specifications of greater than 96% observed in the final product.
How to Synthesize 2-Amino-3,5-Dichloro-N-Methylbenzamide Efficiently
Implementing this synthesis route requires precise control over reaction parameters to ensure optimal yield and safety, particularly given the exothermic nature of the chlorination step. The process is designed as a telescoped one-pot operation, meaning the intermediate formed in the first step does not need to be isolated before proceeding to the second, which significantly reduces processing time and solvent consumption. Operators must monitor the reaction progress using liquid chromatography (HPLC) to confirm the complete disappearance of the starting anhydride in the first stage and the total consumption of both the mono-amide and monochloro-intermediate in the second stage. To guarantee the highest quality output, any excess oxidizing agent must be carefully quenched using reducing agents like sodium sulfite or ascorbic acid before the final isolation. The detailed standardized operating procedures for executing this synthesis at scale are provided below.
- React isatoic anhydride with methylamine solution in solvent A (e.g., ethyl acetate) at 10°C to 50°C to form 2-amino-N-methylbenzamide.
- Add trichloroisocyanuric acid (TCCA) to the organic phase at 30°C to 80°C to effect dichlorination.
- Quench excess oxidant, remove solvent, adjust aqueous pH to 8-13 to dissolve byproducts, and filter to isolate the pure white solid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this TCCA-based synthesis route offers compelling strategic advantages that extend far beyond simple technical metrics. The primary driver for adoption is the dramatic improvement in the cost structure of the final intermediate, achieved not through marginal tweaks but through fundamental changes in raw material selection. By replacing expensive chlorinating agents like DCDMH, which can cost significantly more per ton, with widely available industrial-grade TCCA, the direct material costs are substantially reduced. This shift also mitigates the risk of supply chain disruptions, as TCCA is a commodity chemical produced in vast quantities for water treatment and other industries, ensuring a stable and reliable supply line even during periods of market volatility. Furthermore, the simplified purification process reduces the demand for specialized solvents and energy-intensive distillation steps, leading to broader operational expenditure savings.
- Cost Reduction in Manufacturing: The economic benefits of this process are rooted in the elimination of high-cost reagents and the minimization of waste disposal fees. Traditional methods generate large volumes of solid hydantoin waste that require costly handling and disposal, whereas the byproducts of the TCCA method are water-soluble and environmentally benign, decomposing naturally into harmless substances like carbon dioxide and ammonia. This reduction in hazardous waste generation translates directly into lower environmental compliance costs and reduced liability for the manufacturing facility. Additionally, the high yield of over 90% means that less raw material is required to produce the same amount of finished product, maximizing the return on investment for every kilogram of isatoic anhydride purchased.
- Enhanced Supply Chain Reliability: Securing a consistent supply of high-purity intermediates is critical for maintaining uninterrupted production of downstream pesticides. This synthesis method enhances reliability by utilizing starting materials that are not subject to the same supply constraints as specialty fine chemicals. Isatoic anhydride and methylamine are foundational building blocks in the chemical industry, available from multiple global suppliers, which prevents single-source dependency. The robustness of the reaction conditions, which tolerate a range of temperatures and solvent choices including ethyl acetate and dichloromethane, further ensures that production can continue smoothly even if specific utility conditions fluctuate, providing a buffer against operational risks.
- Scalability and Environmental Compliance: As regulatory pressures on chemical manufacturing intensify globally, the ability to scale production while maintaining a low environmental footprint is a key competitive differentiator. This process is inherently scalable due to its one-pot nature, which reduces the number of unit operations and the associated equipment footprint required in the plant. The use of TCCA avoids the introduction of heavy metals or persistent organic pollutants, aligning with green chemistry principles and simplifying the permitting process for capacity expansion. The ease of separating the product via pH-controlled precipitation means that wastewater treatment loads are significantly lower, facilitating compliance with strict discharge regulations and supporting long-term sustainability goals for the enterprise.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the experimental data and beneficial effects reported in the patent literature. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers provided reflect the specific advantages of the TCCA-mediated pathway over conventional alternatives, focusing on purity profiles, impurity management, and operational simplicity. These insights are intended to assist decision-makers in assessing the potential for process optimization and cost savings within their specific organizational context.
Q: Why is Trichloroisocyanuric Acid (TCCA) preferred over DCDMH for this synthesis?
A: TCCA offers higher reaction activity and significantly lower cost compared to dichlorohydantoin (DCDMH). Furthermore, its byproduct, isocyanuric acid, forms water-soluble salts under alkaline conditions, facilitating easier purification and higher product purity (>96%).
Q: How does this process address the issue of monochlorinated impurities?
A: The high reactivity of TCCA ensures complete conversion of the intermediate. By monitoring via HPLC until both the starting amide and the monochlorinated intermediate disappear, the process guarantees minimal residual impurities without needing complex recrystallization steps.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method utilizes cheap, widely available industrial raw materials like isatoic anhydride and TCCA. The one-pot nature, simple workup involving pH adjustment and filtration, and high yields (>90%) make it highly scalable and economically viable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Amino-3,5-Dichloro-N-Methylbenzamide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced agrochemicals depends on the availability of high-quality intermediates produced via robust and scalable pathways. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical techniques to verify every batch. By leveraging the advanced synthetic strategies outlined in recent patents, we can offer our partners a supply of 2-amino-3,5-dichloro-N-methylbenzamide that is not only chemically superior but also economically optimized for the demands of the global market.
We invite you to collaborate with us to explore how this optimized synthesis route can enhance your supply chain resilience and profitability. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical needs. We encourage you to contact us directly to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on concrete technical evidence. Let us be your partner in driving efficiency and innovation in the agrochemical sector, ensuring that your production lines remain competitive and compliant in an evolving regulatory landscape.
