Advanced Catalytic Synthesis of Polysubstituted Gamma-Butyrolactones for Commercial Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex heterocyclic scaffolds that serve as critical building blocks for next-generation therapeutics. A significant breakthrough in this domain is documented in patent CN110240579B, which discloses a highly efficient preparation method for polysubstituted gamma-butyrolactone compounds. These structural motifs are ubiquitous in natural products and drug molecules, exhibiting potent biological activities ranging from antibacterial to antitumor properties. The innovation lies in a novel catalytic system that enables the direct coupling of N-substituted maleimides with allyl alcohol derivatives, streamlining what was traditionally a laborious synthetic challenge. By leveraging earth-abundant transition metals, this technology offers a pathway to high-purity intermediates that align perfectly with the demands of modern drug discovery pipelines. As a reliable pharmaceutical intermediate supplier, understanding such technological shifts is crucial for maintaining a competitive edge in the global market.

The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of polysubstituted gamma-butyrolactone cores has been plagued by synthetic inefficiencies that hinder large-scale adoption. Traditional routes often rely on multi-step sequences involving harsh reaction conditions, stoichiometric amounts of toxic reagents, or the use of precious metal catalysts that drive up production costs significantly. Furthermore, conventional methods frequently suffer from poor regioselectivity and stereoselectivity, leading to complex mixtures of isomers that are difficult and expensive to separate. The low atom economy associated with these older processes generates substantial chemical waste, creating environmental compliance burdens and increasing the overall carbon footprint of manufacturing. For procurement managers, these inefficiencies translate into volatile pricing and extended lead times, as the supply chain struggles to accommodate the complexities of purification and waste disposal. Consequently, there has been a persistent industry demand for a more streamlined, cost-effective, and environmentally benign synthetic strategy.
The Novel Approach
The methodology outlined in the patent data represents a paradigm shift by utilizing a one-step reductive cyclization strategy that directly addresses the bottlenecks of legacy processes. By employing an electron-deficient olefin acceptor, specifically N-substituted maleimide, and reacting it with an electron-rich olefin donor like allyl alcohol, the process achieves rapid bond formation under mild conditions. The use of non-precious metal catalysts such as cobalt or nickel acetylacetonate drastically reduces raw material costs while maintaining high catalytic activity and selectivity. This approach not only simplifies the operational workflow by eliminating intermediate isolation steps but also enhances the overall yield, ensuring that more starting material is converted into valuable product. The versatility of this system allows for a wide range of substituents on both the maleimide and the alcohol components, enabling the rapid generation of diverse chemical libraries for biological screening. This flexibility is essential for R&D directors who require access to varied structural analogs to optimize drug candidates effectively.
Mechanistic Insights into Co/Ni-Catalyzed Reductive Cyclization
At the heart of this technological advancement is a sophisticated catalytic cycle that orchestrates the formation of multiple bonds in a single operation. The mechanism likely involves the activation of the allyl alcohol by the transition metal center, generating a reactive species that undergoes conjugate addition to the maleimide double bond. Subsequent intramolecular cyclization and reduction steps, facilitated by the hydride source, lock in the gamma-butyrolactone architecture with high fidelity. The choice of ligand environment around the metal center plays a pivotal role in stabilizing key intermediates and preventing undesired side reactions such as polymerization or over-reduction. Understanding these mechanistic nuances allows chemists to fine-tune reaction parameters, such as solvent polarity and temperature, to maximize efficiency. The ability to control the electronic properties of the catalyst ensures that even sterically hindered substrates can be processed effectively, broadening the scope of accessible chemical space.
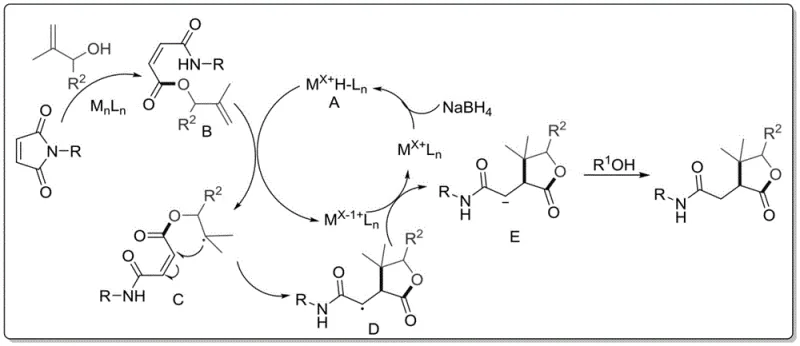
Impurity control is another critical aspect where this mechanism excels, providing significant advantages for quality assurance teams. The high selectivity of the catalytic system minimizes the formation of by-products that typically arise from non-specific radical pathways or competing nucleophilic attacks. By carefully selecting the reducing agent, such as phenylsilane or sodium borohydride, the process avoids the generation of metallic residues that are difficult to remove and can be toxic in final drug products. The reaction conditions are optimized to prevent decomposition of sensitive functional groups, ensuring that the integrity of the molecular scaffold is maintained throughout the synthesis. This results in a crude product profile that is much cleaner than those obtained from traditional methods, reducing the burden on downstream purification units. For supply chain heads, this means fewer batch failures and more consistent delivery schedules, as the risk of encountering unmanageable impurity spikes is substantially mitigated.
How to Synthesize Polysubstituted Gamma-Butyrolactone Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires adherence to specific operational protocols to ensure safety and reproducibility. The process begins with the precise weighing and charging of N-substituted maleimide and the chosen allyl alcohol derivative into a suitable reaction vessel equipped with stirring and temperature control. A solvent system, preferably acetonitrile or a mixture with ether solvents, is added to dissolve the reactants, creating a homogeneous phase that facilitates efficient mass transfer. The catalyst and reducing agent are then introduced, initiating the exothermic reaction which must be monitored to maintain the temperature within the optimal range of room temperature to 100°C. Detailed standardized synthesis steps see the guide below.
- Charge a reaction vessel with N-substituted maleimide and allyl alcohol derivatives in a molar ratio optimized for maximum conversion, typically ranging from 1: 1 to 1:4 depending on substrate sterics.
- Introduce an earth-abundant transition metal catalyst such as cobalt acetylacetonate or nickel acetylacetonate along with a compatible hydride reducing agent like phenylsilane or sodium borohydride.
- Maintain the reaction mixture at mild temperatures between room temperature and 100°C in a polar aprotic solvent like acetonitrile, followed by aqueous quenching and recrystallization to isolate the high-purity solid product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented technology offers profound benefits that extend beyond mere technical feasibility, directly impacting the bottom line and operational resilience. The shift away from precious metal catalysts to abundant base metals like cobalt and nickel represents a strategic move to decouple production costs from the volatile pricing of commodities like palladium and platinum. This substitution not only lowers the direct material cost but also simplifies the regulatory landscape regarding heavy metal residues in pharmaceutical ingredients. Furthermore, the simplicity of the one-pot procedure reduces labor hours and energy consumption, contributing to substantial cost savings in manufacturing overheads. For procurement managers, this translates into a more stable pricing model and the ability to negotiate better terms with suppliers who utilize such efficient technologies. The overall economic viability of producing these high-value intermediates is significantly enhanced, making them accessible for a broader range of therapeutic applications.
- Cost Reduction in Manufacturing: The elimination of expensive noble metal catalysts and the reduction of synthetic steps lead to a drastic simplification of the production process, thereby lowering the total cost of goods sold. By utilizing readily available and inexpensive reagents such as allyl alcohols and maleimides, the raw material expenditure is minimized without compromising on the quality of the final output. The high yield associated with this method ensures that less starting material is wasted, further optimizing the resource utilization efficiency. Additionally, the reduced need for complex purification techniques lowers the consumption of solvents and chromatography media, which are often significant cost drivers in fine chemical synthesis. These cumulative effects result in a leaner manufacturing operation that is better positioned to withstand market fluctuations and price pressures.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals that are widely produced and stocked globally ensures a robust supply chain that is less susceptible to disruptions caused by geopolitical events or single-source dependencies. The mild reaction conditions allow for the use of standard glass-lined or stainless steel reactors, meaning that production can be easily transferred between different manufacturing sites without the need for specialized equipment. This flexibility enhances the agility of the supply network, enabling rapid scaling up or down in response to market demand changes. Moreover, the stability of the reagents and the simplicity of the process reduce the risk of shipping hazards and storage complications, facilitating smoother logistics operations. For supply chain heads, this reliability is paramount in maintaining continuous production schedules and meeting strict delivery commitments to downstream pharmaceutical clients.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, with reaction parameters that can be linearly translated from gram-scale experiments to multi-ton industrial batches without losing efficiency. The use of greener solvents and the generation of minimal hazardous waste align with increasingly stringent environmental regulations, reducing the liability and cost associated with waste treatment and disposal. The absence of high-pressure or cryogenic requirements simplifies the engineering controls needed for safe operation, lowering the capital investment required for plant expansion. This environmental friendliness also enhances the corporate sustainability profile, which is becoming a key criterion for partnerships with major multinational pharmaceutical companies. Consequently, this technology supports long-term business growth by ensuring compliance with global standards while maximizing production throughput.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this synthesis technology, based on the detailed disclosures within the patent documentation. These insights are intended to clarify the operational advantages and potential applications for stakeholders evaluating this method for their own production needs. Understanding these aspects is vital for making informed decisions about integrating this chemistry into existing manufacturing portfolios. The answers provided reflect the consensus of technical analysis regarding the process capabilities and limitations.
Q: What are the primary advantages of this catalytic method over traditional lactone synthesis?
A: This method utilizes a one-step reductive cyclization that bypasses complex multi-step sequences, significantly reducing processing time and eliminating the need for expensive precious metal catalysts like palladium in favor of cost-effective cobalt or nickel systems.
Q: How does this process impact impurity profiles for pharmaceutical applications?
A: The controlled radical or cationic mechanism facilitated by the specific catalyst system ensures high regioselectivity, minimizing the formation of side-products and simplifying downstream purification to meet stringent pharmaceutical purity specifications.
Q: Is this synthesis route scalable for industrial manufacturing?
A: Yes, the reaction operates under atmospheric pressure and mild thermal conditions using commercially available solvents and reagents, making it highly amenable to scale-up from kilogram to multi-ton production without requiring specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Gamma-Butyrolactone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the one described in CN110240579B for accelerating drug development timelines. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the bench to the market. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications required for clinical and commercial grade materials. We understand that consistency and quality are non-negotiable in the pharmaceutical industry, and our team is committed to delivering intermediates that meet the highest international standards. By leveraging our technical expertise, you can mitigate the risks associated with process scale-up and focus on your core competencies in drug discovery.
We invite you to engage with our technical procurement team to discuss how we can tailor this synthesis route to your specific project requirements. Request a Customized Cost-Saving Analysis to understand the economic benefits of switching to this catalytic method for your supply chain. We are ready to provide specific COA data and route feasibility assessments to demonstrate our capability to deliver high-quality polysubstituted gamma-butyrolactone derivatives efficiently. Partner with us to secure a reliable supply of critical intermediates that will drive the success of your pharmaceutical pipeline.
