Revolutionizing Alpha-Aryl Carbonyl Synthesis for Scalable Pharmaceutical Manufacturing
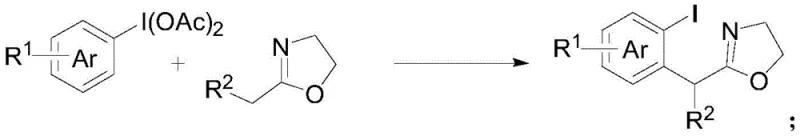
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable methodologies for constructing complex molecular architectures, particularly alpha-aryl carbonyl motifs which serve as critical scaffolds in numerous bioactive molecules. Patent CN113135869A introduces a groundbreaking synthetic strategy that addresses long-standing challenges in this domain by leveraging hypervalent iodine chemistry. This innovation details a novel method for preparing alpha-aryl carbonyl compounds through the activation of substituted iodobenzene diacetate by a Lewis acid, followed by a reaction with substituted oxazoline compounds in the presence of a base. This approach represents a significant paradigm shift from traditional transition-metal-dependent routes, offering a pathway that is not only chemically elegant but also commercially viable for the production of high-purity pharmaceutical intermediates. The technology promises to enhance the efficiency of synthesizing key building blocks used in the development of new drugs, providing a reliable foundation for both early-stage discovery and late-stage process optimization.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alpha-aryl carbonyl compounds has relied heavily on methods that present substantial operational and economic hurdles for large-scale manufacturing. Traditional protocols often involve the reaction of cyclic ketones with diaryl iodonium salts mediated by stoichiometric amounts of metal reagents, which necessitates the use of strong bases and results in poor functional group compatibility. Furthermore, alternative pathways utilizing carbonyl compounds and haloaromatics catalyzed by metallic palladium, nickel, or copper are plagued by issues such as narrow substrate scope and selectivity challenges. These transition-metal-catalyzed processes frequently require rigorous purification steps to remove trace metal residues, a critical requirement for pharmaceutical ingredients that adds significant cost and complexity to the supply chain. Additionally, methods involving [3,3] shift rearrangements of aryl sulfoxides are often sensitive to moisture and require specialized substrates, limiting their general applicability in diverse synthetic campaigns.
The Novel Approach
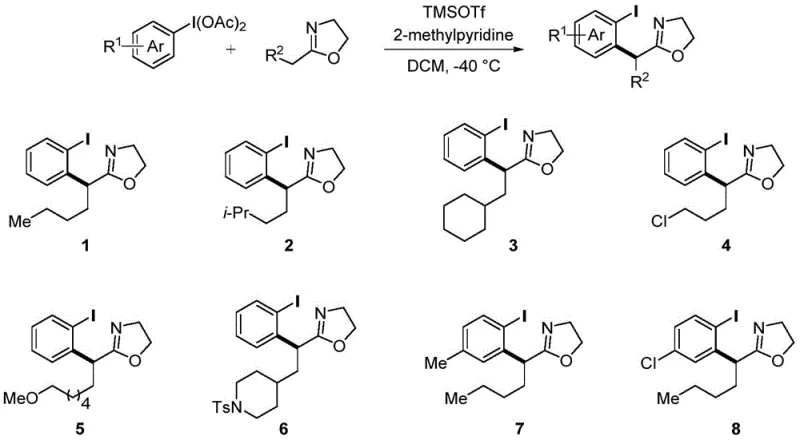
In stark contrast to these legacy techniques, the methodology disclosed in CN113135869A utilizes a transition-metal-free system driven by the reactivity of iodobenzene diacetate and oxazolines. This novel route operates under remarkably mild conditions, typically ranging from -40°C to 25°C, which preserves sensitive functional groups that might otherwise degrade under harsh thermal or basic conditions. The core transformation involves a [3,3]-sigmatropic rearrangement that efficiently constructs the carbon-carbon bond necessary for the alpha-aryl motif without the need for expensive precious metal catalysts. By employing readily available starting materials and avoiding the complexities associated with metal catalysis, this method streamlines the synthetic workflow, making it an attractive option for cost reduction in pharmaceutical intermediate manufacturing. The simplicity of the operation and the ease of product separation further underscore its potential for adoption in industrial settings where throughput and purity are paramount.
Mechanistic Insights into Lewis Acid-Catalyzed [3,3]-Sigmatropic Rearrangement
The chemical elegance of this process lies in the precise activation of the hypervalent iodine species by a Lewis acid, such as trimethylsilyl trifluoromethanesulfonate (TMSOTf). This activation step generates a highly reactive electrophilic species that facilitates the nucleophilic attack by the oxazoline nitrogen or carbon center, depending on the specific mechanistic pathway, ultimately triggering the [3,3]-sigmatropic rearrangement. The use of TMSOTf is particularly advantageous due to its high Lewis acidity, which allows the reaction to proceed rapidly even at cryogenic temperatures like -40°C, thereby suppressing side reactions and polymerization that could compromise yield. The subsequent addition of a base, such as 2-methylpyridine, serves to neutralize the acidic byproducts and drive the equilibrium towards the formation of the desired alpha-aryl carbonyl product. This delicate balance of activation and neutralization ensures high conversion rates while maintaining the integrity of the oxazoline ring, which is crucial for downstream transformations.
From an impurity control perspective, the mild nature of this reaction mechanism offers distinct advantages over traditional high-temperature or strong-base mediated processes. The low-temperature regime minimizes the formation of thermal degradation products and prevents the racemization of chiral centers if chiral substrates are employed. Furthermore, the absence of transition metals eliminates the risk of metal-induced oxidative side reactions, leading to a cleaner crude reaction profile. This inherent cleanliness simplifies the downstream purification process, often allowing for straightforward isolation via column chromatography or crystallization. The robustness of the mechanism across a wide range of substituents on both the aromatic ring and the oxazoline side chain demonstrates the versatility of this catalytic system, making it a powerful tool for generating diverse libraries of analogues for structure-activity relationship studies.
How to Synthesize Alpha-Aryl Carbonyl Compounds Efficiently
The practical implementation of this synthesis involves a straightforward sequence of reagent addition and temperature control that is well-suited for standard laboratory and pilot plant equipment. The process begins with the activation of the iodobenzene diacetate precursor, followed by the introduction of the oxazoline partner and the base to initiate the rearrangement. Detailed standardized operating procedures regarding stoichiometry, addition rates, and quenching protocols are essential for ensuring reproducible results and maximizing yield. For a comprehensive guide on the specific execution of this protocol, including precise molar ratios and workup details, please refer to the standardized synthesis steps provided below.
- Activate the substituted iodobenzene diacetate with a Lewis acid such as TMSOTf in anhydrous DCM at low temperature (-40°C).
- Add the substituted oxazoline substrate and a base like 2-methylpyridine to initiate the [3,3]-sigmatropic rearrangement reaction.
- Monitor reaction completion via TLC, then perform aqueous workup and column chromatography to isolate the high-purity alpha-aryl carbonyl product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers compelling strategic benefits that extend beyond mere chemical efficiency. The elimination of precious metal catalysts such as palladium and platinum directly translates to significant cost savings, as these materials are subject to volatile market pricing and supply constraints. Moreover, the removal of heavy metals from the process flow drastically reduces the regulatory burden associated with residual metal testing and clearance, accelerating the timeline for regulatory filings and commercial release. The use of commodity chemicals like iodobenzene diacetate and simple oxazolines ensures a stable and secure supply of raw materials, mitigating the risk of production delays caused by sourcing bottlenecks. This reliability is critical for maintaining continuous manufacturing operations and meeting the demanding delivery schedules of global pharmaceutical clients.
- Cost Reduction in Manufacturing: The transition from transition-metal catalysis to a Lewis acid-mediated organic transformation fundamentally alters the cost structure of producing alpha-aryl carbonyl intermediates. By removing the need for expensive ligands and metal salts, the direct material costs are substantially lowered. Additionally, the simplified workup procedure reduces the consumption of scavengers and specialized filtration media required for metal removal, leading to further operational expenditure savings. The overall process efficiency is enhanced by the high atom economy of the rearrangement reaction, minimizing waste generation and disposal costs associated with heavy metal sludge.
- Enhanced Supply Chain Reliability: The reliance on widely available organic reagents rather than specialized organometallic complexes enhances the resilience of the supply chain. Iodobenzene diacetate and substituted oxazolines are accessible from multiple global suppliers, reducing dependency on single-source vendors. This diversification of the supply base ensures that production schedules remain unaffected by geopolitical disruptions or logistical challenges that often impact the availability of rare earth metals or specialized catalysts. Consequently, manufacturers can offer more reliable lead times and guarantee consistent supply continuity for their customers.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of toxic heavy metals make this process inherently safer and more environmentally friendly, aligning with modern green chemistry principles. Scaling up from gram to kilogram quantities does not require specialized high-pressure or high-temperature equipment, as the reaction proceeds efficiently at ambient or sub-ambient pressures. The reduced environmental footprint facilitates easier compliance with increasingly stringent environmental regulations, avoiding the costly permitting and waste treatment processes associated with metal-containing effluents. This sustainability advantage positions the technology as a future-proof solution for long-term commercial manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic technology. These insights are derived directly from the experimental data and scope defined in the patent literature, providing a clear understanding of the method's capabilities and limitations. Understanding these details is crucial for R&D teams evaluating the feasibility of integrating this route into their existing process workflows and for procurement teams assessing the value proposition.
Q: What are the primary advantages of this method over traditional transition-metal catalysis?
A: Unlike conventional methods requiring palladium, nickel, or copper catalysts which often suffer from poor selectivity and heavy metal contamination risks, this novel approach utilizes hypervalent iodine reagents and Lewis acids. This eliminates the need for expensive transition metals and stringent heavy metal removal processes, significantly simplifying purification and reducing environmental impact.
Q: What are the optimal reaction conditions for this synthesis?
A: The reaction proceeds optimally under mild conditions, typically between -40°C and 25°C, using dichloromethane as the solvent. The process employs trimethylsilyl trifluoromethanesulfonate (TMSOTf) as the Lewis acid activator and 2-methylpyridine as the base, ensuring high functional group compatibility and minimizing substrate decomposition.
Q: Can the resulting products be further derivatized for drug development?
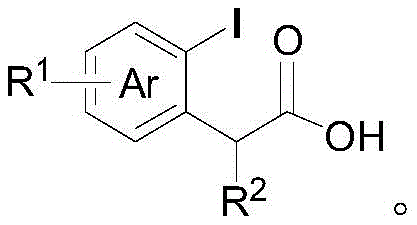
A: Yes, the synthesized alpha-aryl carbonyl compounds, particularly the oxazoline derivatives, serve as versatile intermediates. They can be readily hydrolyzed under acidic conditions to yield corresponding carboxylic acids, which are valuable precursors for amides, esters, and complex pharmaceutical scaffolds, offering significant flexibility in medicinal chemistry campaigns.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Aryl Carbonyl Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this advanced synthetic methodology in accelerating drug discovery and development programs. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop to plant floor is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific requirements of Lewis acid-catalyzed reactions, including precise temperature control and inert atmosphere handling, guaranteeing stringent purity specifications for every batch. With our rigorous QC labs and commitment to quality, we deliver high-purity alpha-aryl carbonyl intermediates that meet the exacting standards of the global pharmaceutical industry.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific molecule, demonstrating how this metal-free route can optimize your budget without compromising quality. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and a comprehensive proposal for your custom synthesis needs. Let us help you secure a competitive advantage through superior chemistry and reliable supply chain execution.
