Advanced Organocatalytic Synthesis of Optically Active Beta-Amino Aldehydes for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing chiral building blocks, particularly those serving as critical intermediates for complex therapeutic agents. Patent CN103189352A introduces a groundbreaking process for the preparation of optically active β-aminoaldehyde compounds, addressing long-standing challenges in stereoselective synthesis. This technology leverages a specialized organocatalytic system to facilitate asymmetric Mannich reactions with unprecedented efficiency. For R&D directors and procurement specialists, understanding the nuances of this patent is vital, as it represents a shift towards more sustainable and cost-effective manufacturing of high-value intermediates used in treatments for conditions such as diabetes and Alzheimer's disease. The core innovation lies in the ability to utilize both aromatic and aliphatic imine derivatives, a significant expansion over prior art which was largely limited to aromatic systems.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of optically active β-amino aldehydes has been fraught with significant technical hurdles that impact both yield and stereochemical purity. Conventional methods, such as those cited in Non-Patent Document 1 (Angewandte Chemie International Edition, 2007), relied heavily on the use of (S)-proline as a catalyst. While effective for aromatic imine compounds, these traditional protocols demonstrated a critical failure when applied to aliphatic imine substrates, resulting in the inability to obtain the target optically active compounds. This limitation severely restricts the structural diversity available to medicinal chemists, forcing reliance on more expensive or multi-step alternative routes. Furthermore, many legacy processes require harsh reaction conditions, toxic heavy metal catalysts, or strictly anhydrous organic solvents, which complicate waste management and increase the overall cost of goods sold (COGS). The instability of the resulting β-amino aldehydes also poses a challenge, often leading to racemization or decomposition during isolation and purification steps if not handled with extreme care.
The Novel Approach
The methodology disclosed in CN103189352A overcomes these barriers through the introduction of a novel optically active pyrrolidine compound acting as an organocatalyst. This new approach enables the successful reaction of imine compounds, including previously problematic aliphatic variants, with aldehyde compounds to produce the desired β-amino aldehydes with excellent enantioselectivity and diastereoselectivity. 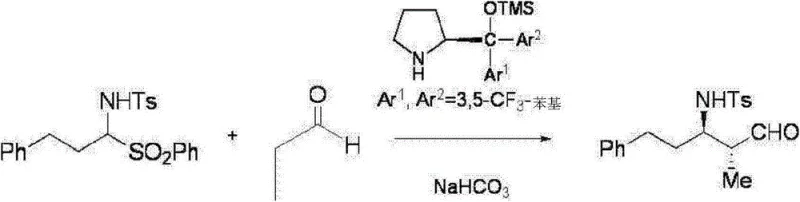 The process is remarkably versatile regarding solvent choice, demonstrating high efficacy even in environmentally benign media such as water containing inorganic salts (e.g., saturated saline) or ether solvents like 1,4-dioxane. This flexibility allows manufacturers to optimize for either performance or sustainability depending on specific project requirements. By utilizing a sulfone compound as a stable precursor that eliminates a sulfonyl group during the reaction, the method ensures a clean transformation pathway. The ability to operate under mild conditions (0-40°C) further enhances the safety profile and energy efficiency of the process, making it highly attractive for large-scale commercial production of reliable pharmaceutical intermediate supplier networks.
The process is remarkably versatile regarding solvent choice, demonstrating high efficacy even in environmentally benign media such as water containing inorganic salts (e.g., saturated saline) or ether solvents like 1,4-dioxane. This flexibility allows manufacturers to optimize for either performance or sustainability depending on specific project requirements. By utilizing a sulfone compound as a stable precursor that eliminates a sulfonyl group during the reaction, the method ensures a clean transformation pathway. The ability to operate under mild conditions (0-40°C) further enhances the safety profile and energy efficiency of the process, making it highly attractive for large-scale commercial production of reliable pharmaceutical intermediate supplier networks.
Mechanistic Insights into Organocatalytic Asymmetric Mannich Reaction
The heart of this technological advancement is the specific structure of the optically active pyrrolidine catalyst, represented generally by formula (4) in the patent. 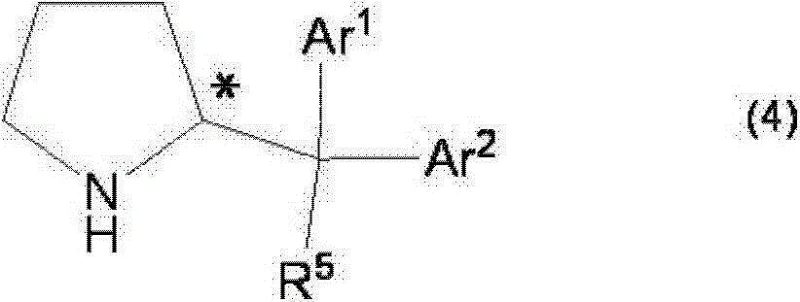 This catalyst features a pyrrolidine ring substituted with bulky aryl groups (Ar1 and Ar2), often 3,5-bis(trifluoromethyl)phenyl groups, and a silyloxy group (OTMS). These steric and electronic features are crucial for inducing high levels of stereocontrol. The mechanism likely involves the formation of an enamine intermediate between the catalyst and the aldehyde donor, which then attacks the imine electrophile in a highly organized transition state. The bulky aryl groups create a chiral environment that shields one face of the reactive species, directing the incoming nucleophile to attack from the preferred trajectory. This precise spatial arrangement is what allows the reaction to distinguish between pro-chiral faces, resulting in the preferential formation of specific enantiomers (e.g., (2R, 3R) or (2S, 3S) configurations) with ee values often exceeding 90%. The use of a sulfone precursor (formula 1-2) adds another layer of mechanistic elegance; the elimination of the sulfonyl group occurs concomitantly with the C-C bond formation, driving the equilibrium forward and simplifying the reaction mixture by avoiding the need for pre-formed, unstable imines.
This catalyst features a pyrrolidine ring substituted with bulky aryl groups (Ar1 and Ar2), often 3,5-bis(trifluoromethyl)phenyl groups, and a silyloxy group (OTMS). These steric and electronic features are crucial for inducing high levels of stereocontrol. The mechanism likely involves the formation of an enamine intermediate between the catalyst and the aldehyde donor, which then attacks the imine electrophile in a highly organized transition state. The bulky aryl groups create a chiral environment that shields one face of the reactive species, directing the incoming nucleophile to attack from the preferred trajectory. This precise spatial arrangement is what allows the reaction to distinguish between pro-chiral faces, resulting in the preferential formation of specific enantiomers (e.g., (2R, 3R) or (2S, 3S) configurations) with ee values often exceeding 90%. The use of a sulfone precursor (formula 1-2) adds another layer of mechanistic elegance; the elimination of the sulfonyl group occurs concomitantly with the C-C bond formation, driving the equilibrium forward and simplifying the reaction mixture by avoiding the need for pre-formed, unstable imines.
Impurity control is inherently built into this catalytic cycle. The high diastereoselectivity means that unwanted diastereomers (cis vs. trans) are minimized at the source, reducing the burden on downstream purification processes. When the aldehyde component contains a substituent at the alpha-position (R2 is not hydrogen), the reaction preferentially yields the trans-isomer, which is often the desired configuration for biological activity. The patent data indicates that by selecting the appropriate enantiomer of the catalyst (either the S-configuration or R-configuration at the chiral center), manufacturers can access either enantiomer of the product on demand. This switchable stereochemistry is a powerful tool for process development, allowing for the rapid synthesis of analog libraries without changing the core reaction infrastructure. Furthermore, the stability issues associated with free β-amino aldehydes are mitigated by the option to reduce the crude reaction mixture directly to the corresponding amide alcohol using sodium borohydride, locking in the stereochemistry and facilitating accurate quality control analysis.
How to Synthesize Optically Active Beta-Amino Aldehydes Efficiently
Implementing this synthesis route requires careful attention to the preparation of precursors and reaction conditions to maximize yield and optical purity. The process begins with the preparation of the sulfone compound, which serves as a stable surrogate for the reactive imine. This is achieved by reacting an aldehyde with an arenesulfonamide and sodium benzenesulfinate in an acidic aqueous medium. Once the precursor is secured, the core Mannich reaction is executed by combining the sulfone compound, the aldehyde partner, a base such as sodium bicarbonate, and the chiral pyrrolidine catalyst in a suitable solvent. 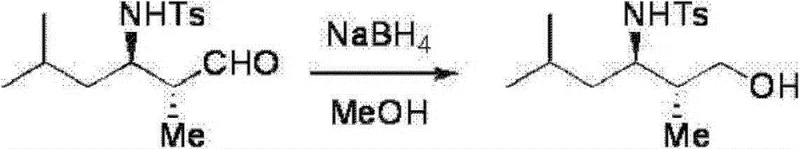 The reaction mixture is typically stirred at temperatures ranging from 0°C to 40°C for periods extending from 10 to 50 hours, depending on the specific substrates involved. Following the reaction, standard workup procedures involving extraction and chromatography are employed to isolate the product. For applications requiring long-term storage or further derivatization, the unstable aldehyde functionality is often reduced to a primary alcohol using sodium borohydride in methanol, yielding a stable amino alcohol derivative. Detailed standardized synthesis steps see the guide below.
The reaction mixture is typically stirred at temperatures ranging from 0°C to 40°C for periods extending from 10 to 50 hours, depending on the specific substrates involved. Following the reaction, standard workup procedures involving extraction and chromatography are employed to isolate the product. For applications requiring long-term storage or further derivatization, the unstable aldehyde functionality is often reduced to a primary alcohol using sodium borohydride in methanol, yielding a stable amino alcohol derivative. Detailed standardized synthesis steps see the guide below.
- Prepare the sulfone compound precursor by reacting an aldehyde with arenesulfonamide and sodium benzenesulfinate dihydrate in formic acid and water.
- Conduct the asymmetric Mannich reaction by mixing the sulfone compound, an aldehyde, sodium bicarbonate, and the optically active pyrrolidine catalyst in a solvent such as saturated brine or 1,4-dioxane.
- Isolate the product via extraction and purification, optionally reducing the unstable aldehyde to a stable amide alcohol using sodium borohydride for analysis or further use.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the technology described in CN103189352A offers substantial strategic benefits beyond mere chemical novelty. The shift towards organocatalysis eliminates the need for expensive and potentially toxic transition metal catalysts, which simplifies regulatory compliance and reduces the cost associated with heavy metal removal and disposal. This transition directly contributes to cost reduction in API manufacturing by streamlining the purification train and minimizing waste treatment expenses. The ability to use water or saturated saline as a reaction medium is a game-changer for green chemistry initiatives, significantly lowering the volume of volatile organic compounds (VOCs) released into the environment and reducing solvent procurement costs. This environmental compatibility aligns perfectly with modern corporate sustainability goals and increasingly stringent global regulations.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts removes a major variable cost driver and supply risk associated with fluctuating metal prices. Additionally, the high selectivity of the reaction minimizes the formation of byproducts, leading to higher overall yields and less material loss during purification. The use of inexpensive bases like sodium bicarbonate and common solvents further drives down the raw material costs. By consolidating the imine generation and Mannich addition into a streamlined sequence using stable sulfone precursors, the process reduces the number of unit operations required, thereby saving labor and energy costs.
- Enhanced Supply Chain Reliability: The reagents required for this process, including the sulfone precursors, aldehydes, and the organocatalyst, are derived from readily available commodity chemicals. This broad availability ensures a robust supply chain that is less susceptible to disruptions compared to processes relying on exotic or single-source reagents. The stability of the sulfone intermediates allows for batch production and inventory stocking, providing flexibility in scheduling and reducing lead times for high-purity pharmaceutical intermediates. Manufacturers can scale production up or down rapidly in response to market demand without facing bottlenecks related to reagent scarcity.
- Scalability and Environmental Compliance: The reaction conditions are mild and safe, operating at near-ambient temperatures and pressures, which facilitates easy scale-up from laboratory to pilot and commercial plant scales. The potential to run the reaction in aqueous media significantly reduces the fire hazard and explosion risk associated with large volumes of flammable organic solvents. This safety profile lowers insurance premiums and infrastructure investment requirements. Moreover, the reduced solvent usage and absence of heavy metals simplify wastewater treatment, ensuring compliance with environmental discharge standards and avoiding potential fines or shutdowns.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These answers are derived directly from the experimental data and specifications provided in the patent documentation to ensure accuracy and reliability for potential partners and licensees. Understanding these details is crucial for assessing the feasibility of integrating this technology into existing production lines.
Q: What is the key advantage of the catalyst described in CN103189352A?
A: The specialized optically active pyrrolidine catalyst enables high enantioselectivity and diastereoselectivity even with aliphatic imine compounds, overcoming limitations of previous methods that were restricted to aromatic substrates.
Q: Can this process be performed in aqueous media?
A: Yes, the patent explicitly demonstrates that the reaction proceeds effectively in water containing inorganic salts, such as saturated saline, which offers significant environmental and cost benefits over organic solvents.
Q: How is the stability of the beta-amino aldehyde product managed?
A: Since the beta-amino aldehyde can be unstable, the process often includes an in-situ or post-reaction reduction step using sodium borohydride to convert it into a stable amide alcohol derivative for isolation and quality control.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Amino Aldehyde Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic routes disclosed in CN103189352A for the production of high-value chiral intermediates. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, utilizing our rigorous QC labs to verify enantiomeric excess and chemical purity at every stage of the manufacturing process. Our facility is equipped to handle the specific requirements of organocatalytic reactions, including precise temperature control and specialized containment for sensitive intermediates.
We invite you to collaborate with us to leverage this advanced technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can accelerate your development timeline and optimize your supply chain. Let us be your partner in bringing these critical pharmaceutical intermediates to market with speed, quality, and economic efficiency.
