Advanced Catalytic Synthesis of 3-Iodo-2-Bromotoluene for Commercial Scale-Up
The chemical industry is constantly evolving towards greener and more efficient synthetic pathways, a trend vividly exemplified by the innovations disclosed in patent CN113214041A. This patent introduces a robust and novel method for preparing 3-iodo-2-bromotoluene, a critical building block extensively utilized in the synthesis of complex pharmaceutical agents and advanced agrochemical formulations. The disclosed methodology represents a significant departure from legacy processes that have long plagued manufacturers with environmental burdens and operational inefficiencies. By leveraging a strategic sequence of diazotization, catalytic hydrogenation, and halogen exchange, the inventors have established a route that prioritizes both atomic economy and process safety. For R&D directors and procurement specialists alike, understanding the nuances of this patent is essential, as it offers a blueprint for reducing waste generation while maintaining high product integrity. The transition from hazardous reductants to catalytic systems not only aligns with modern regulatory standards but also promises substantial improvements in yield consistency and downstream purification efforts.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of halogenated toluene derivatives like 3-iodo-2-bromotoluene has relied heavily on reduction steps utilizing iron powder in acidic media. While chemically effective, this traditional approach generates massive quantities of iron mud, a solid waste byproduct that poses severe disposal challenges and environmental hazards. The handling of iron sludge requires extensive centrifugal operations which are often difficult to automate and prone to mechanical failure, leading to inconsistent batch quality and increased labor costs. Furthermore, the presence of residual iron species can complicate subsequent reaction steps, necessitating rigorous and costly purification protocols to meet the stringent purity specifications required for pharmaceutical intermediates. The safety risks associated with handling large volumes of reactive metal powders and the potential for exothermic runaway reactions further diminish the attractiveness of these legacy methods for modern, large-scale manufacturing facilities seeking to minimize their carbon footprint and operational liabilities.
The Novel Approach
In stark contrast, the methodology outlined in patent CN113214041A circumvents these issues by employing a palladium-catalyzed hydrogenation strategy for the critical reduction step. This innovative shift eliminates the generation of solid metal waste entirely, replacing it with a clean catalytic cycle that produces water as the primary byproduct. The use of 2-amino-3-nitrotoluene as a starting material allows for a highly controlled sequence where functional groups are introduced with precision, minimizing the formation of regio-isomers and other difficult-to-remove impurities. By integrating mild reaction conditions and commercially available reagents such as hydrobromic acid and alkali metal iodides, the new process ensures that the synthesis remains economically viable without compromising on safety or environmental standards. This approach not only streamlines the workflow but also significantly enhances the overall throughput of the manufacturing line, making it an ideal candidate for companies aiming to optimize their supply chain resilience and reduce total cost of ownership.
Mechanistic Insights into Pd/C-Catalyzed Hydrogenation and Halogenation
The core of this synthetic breakthrough lies in the seamless integration of electrophilic aromatic substitution and catalytic reduction mechanisms. The process initiates with the diazotization of 2-amino-3-nitrotoluene, where the amino group is converted into a diazonium salt using sodium nitrite and hydrobromic acid, followed immediately by a Sandmeyer-type bromination to yield 2-bromo-3-nitrotoluene. This intermediate then undergoes a pivotal transformation via heterogeneous catalysis, where hydrogen gas interacts with the nitro group on the surface of the palladium-carbon catalyst. This hydrogenation step is meticulously controlled at pressures between 0.2 to 0.6 MPa, ensuring selective reduction of the nitro group to an amine without affecting the sensitive bromine substituent, a feat that requires precise catalyst selection and reaction monitoring. The final stage involves a second diazotization followed by iodination using potassium iodide, effectively swapping the diazonium moiety for an iodine atom to complete the target molecule.
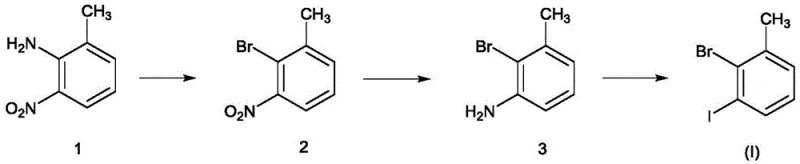
Impurity control is inherently built into this mechanistic design, as the stepwise nature of the synthesis allows for intermediate purification before proceeding to the next stage. The avoidance of iron powder prevents the introduction of metallic contaminants that could catalyze unwanted side reactions or degrade product stability during storage. Moreover, the use of specific molar ratios, such as maintaining a 1:1.0-2.0 ratio of substrate to nitrite, ensures that diazotization proceeds to completion without excessive accumulation of unstable diazonium species, which could otherwise lead to tar formation or decomposition. The careful regulation of temperature during the exothermic diazotization phases, typically kept between 0-20°C, further suppresses the formation of phenolic byproducts, ensuring that the final 3-iodo-2-bromotoluene achieves the high purity levels demanded by downstream coupling reactions in drug discovery and development pipelines.
How to Synthesize 3-Iodo-2-Bromotoluene Efficiently
Executing this synthesis requires strict adherence to the optimized parameters defined in the patent to maximize yield and safety. The process begins with the preparation of the brominated nitro-intermediate, followed by the critical catalytic hydrogenation which demands careful pressure management, and concludes with the iodination step which requires precise temperature control to manage the evolution of iodine vapors. Operators must ensure that all reagents, particularly the nitrite solutions and acids, are added at controlled rates to prevent thermal spikes that could compromise reaction selectivity. The detailed standardized synthesis steps, including specific solvent choices like dioxane and ethanol for solubility management, are critical for reproducibility and are outlined in the comprehensive guide below for technical teams ready to implement this superior route.
- React 2-amino-3-nitrotoluene with hydrobromic acid, nitrite, and alkali metal bromide to form 2-bromo-3-nitrotoluene.
- Perform hydrogenation reduction on 2-bromo-3-nitrotoluene using a palladium catalyst to obtain 2-bromo-3-aminotoluene.
- React 2-bromo-3-aminotoluene with nitrite, concentrated acid, and alkali metal iodide to finalize the synthesis of 3-iodo-2-bromotoluene.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route translates directly into tangible operational benefits and risk mitigation. The elimination of iron powder not only removes a hazardous material from the inventory but also drastically reduces the logistical burden associated with waste disposal and treatment. By switching to a catalytic hydrogenation process, manufacturers can achieve a cleaner production profile that aligns with increasingly strict environmental regulations, thereby avoiding potential fines and shutdowns that plague facilities relying on outdated heavy metal chemistry. The reliance on commodity chemicals like hydrobromic acid and potassium iodide ensures that raw material sourcing remains stable and cost-effective, shielding the supply chain from the volatility often seen with specialized or scarce reagents. This stability is crucial for maintaining continuous production schedules and meeting the just-in-time delivery requirements of global pharmaceutical clients.
- Cost Reduction in Manufacturing: The transition away from iron powder reduction eliminates the significant costs associated with the disposal of toxic iron mud and the maintenance of equipment damaged by abrasive slurries. Furthermore, the high selectivity of the palladium-catalyzed step reduces the need for extensive chromatographic purification, lowering solvent consumption and energy usage during the isolation phase. The overall simplification of the workup procedure, which avoids complex filtration of solid wastes, leads to faster batch turnover times and reduced labor overhead, contributing to a leaner and more cost-efficient manufacturing operation that enhances profit margins without sacrificing quality.
- Enhanced Supply Chain Reliability: Utilizing widely available starting materials such as 2-amino-3-nitrotoluene ensures that production is not bottlenecked by the scarcity of exotic precursors. The robustness of the reaction conditions, which tolerate standard industrial equipment and do not require cryogenic temperatures or ultra-high pressures, means that the process can be easily transferred between different manufacturing sites or scaled up without significant capital investment in specialized infrastructure. This flexibility allows supply chain managers to diversify their production base and secure continuity of supply even in the face of regional disruptions, ensuring that critical intermediates remain available for downstream API synthesis.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, with reaction exotherms that are manageable through standard cooling systems and reagent addition rates that are compatible with large-volume reactors. The absence of heavy metal waste streams simplifies the environmental permitting process and reduces the long-term liability associated with site contamination. By adopting this greener chemistry, companies can improve their sustainability metrics and corporate social responsibility profiles, which are becoming increasingly important criteria for partnerships with major multinational pharmaceutical corporations that prioritize green supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These answers are derived directly from the experimental data and advantageous effects described in patent CN113214041A, providing clarity on how this method outperforms traditional techniques in terms of safety, yield, and environmental impact. Understanding these details is vital for technical teams evaluating the feasibility of adopting this process for their own manufacturing portfolios.
Q: How does the new method improve upon traditional iron powder reduction?
A: The novel method replaces hazardous iron powder reduction with palladium-catalyzed hydrogenation, eliminating toxic iron mud waste and simplifying post-reaction filtration, thereby enhancing environmental compliance and operational safety.
Q: What are the critical reaction conditions for the hydrogenation step?
A: The hydrogenation step utilizes a palladium-carbon catalyst under mild pressure conditions ranging from 0.2 to 0.6 MPa and temperatures between 45-55°C, ensuring high conversion rates without requiring extreme thermal inputs.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the process utilizes readily available raw materials like 2-amino-3-nitrotoluene and common reagents such as hydrobromic acid and potassium iodide, making it highly scalable and economically viable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Iodo-2-Bromotoluene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your drug development programs. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the laboratory bench to full-scale manufacturing. Our state-of-the-art facilities are equipped to handle the specific requirements of halogenated aromatic synthesis, including the safe management of hydrogenation reactions and diazotization processes, while our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets your exacting standards. We are committed to delivering not just a chemical product, but a reliable supply solution that supports your timeline and regulatory goals.
We invite you to engage with our technical procurement team to discuss how our capabilities align with your specific needs for 3-iodo-2-bromotoluene and related intermediates. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized processes can reduce your overall manufacturing expenses. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to being your trusted partner in chemical innovation and supply chain excellence.
