Advanced Nickel-Catalyzed Synthesis of Monofluoroethyl Aromatic Compounds for Commercial Scale
Introduction to Advanced Fluoroethyl Synthesis Technology
The strategic incorporation of fluorine atoms into organic molecules remains a cornerstone of modern medicinal chemistry and agrochemical design, profoundly influencing the metabolic stability and bioavailability of active pharmaceutical ingredients. Patent CN107628926B introduces a transformative preparation method for monofluoroethyl substituted aromatic compounds, addressing critical bottlenecks in the supply of these high-value intermediates. This technology leverages a robust nickel-catalyzed cross-coupling system that directly links a monofluoroethyl fragment to aromatic rings, bypassing the multi-step and hazardous protocols traditionally associated with fluorination. By utilizing 1-fluoro-2-iodoethane and aryl boronic acids under mild conditions, this process offers a streamlined pathway that aligns with the rigorous demands of contemporary pharmaceutical manufacturing. The significance of this innovation extends beyond mere chemical novelty, representing a tangible shift towards safer, more cost-effective, and scalable production methodologies for fluorinated building blocks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of monofluoroethyl substituted aromatic compounds has relied heavily on the nucleophilic fluorination of beta-hydroxyethyl aromatic precursors, a strategy fraught with significant operational and safety challenges. As illustrated in the reaction pathway below, conventional methods often necessitate the use of highly toxic and expensive fluorinating reagents such as diethylaminosulfur trifluoride (DAST) or sulfur tetrafluoride (SF4), which pose severe handling risks and require specialized containment infrastructure. Furthermore, alternative routes involving the conversion of hydroxyl groups into leaving groups like tosylates prior to fluorination introduce additional synthetic steps, leading to cumulative yield losses and increased waste generation. These inefficiencies not only escalate the cost of goods sold but also complicate the supply chain due to the regulatory scrutiny surrounding hazardous reagents, making them less desirable for large-scale commercial applications.
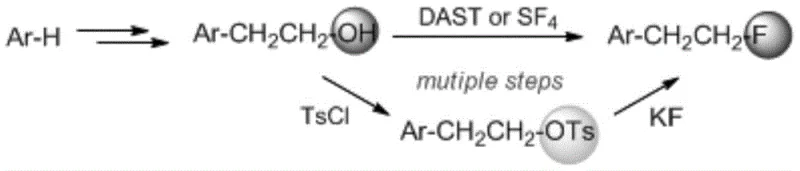
The Novel Approach
In stark contrast to legacy technologies, the novel approach detailed in the patent employs a direct cross-coupling strategy that eliminates the need for pre-functionalized hydroxyethyl intermediates and dangerous fluorinating agents. By utilizing a nickel catalyst in conjunction with bipyridine ligands, the method facilitates the efficient coupling of 1-fluoro-2-iodoethane with a wide array of aryl boronic acids. This paradigm shift simplifies the synthetic route to a single catalytic step, significantly reducing the operational complexity and the overall environmental footprint of the manufacturing process. The use of base metal nickel instead of precious metals like palladium further enhances the economic viability of the process, while the mild reaction conditions ensure compatibility with sensitive functional groups often present in complex drug candidates. This streamlined methodology represents a substantial advancement in process chemistry, offering a reliable and scalable solution for the production of high-purity fluoroethyl intermediates.

Mechanistic Insights into Ni-Catalyzed Cross-Coupling
The efficacy of this synthesis relies on a sophisticated catalytic cycle where the nickel center orchestrates the activation of the carbon-iodine bond in 1-fluoro-2-iodoethane and the subsequent transmetalation with the aryl boronic acid species. The bipyridine ligand plays a critical role in stabilizing the nickel catalyst, modulating its electronic properties to facilitate oxidative addition and reductive elimination steps with high fidelity. This ligand-accelerated catalysis ensures that the reaction proceeds with minimal formation of side products, such as homocoupling byproducts or defluorinated species, which are common pitfalls in fluorinated compound synthesis. The weak base present in the reaction mixture activates the boronic acid by forming a boronate complex, thereby enhancing its nucleophilicity and promoting efficient transmetalation to the nickel center. This precise control over the mechanistic pathway is essential for achieving the high yields and purity profiles required for pharmaceutical grade intermediates.
Furthermore, the reaction design incorporates specific measures to manage the volatility of 1-fluoro-2-iodoethane, ensuring that the reagent remains available for the catalytic cycle throughout the reaction duration. The choice of solvent, particularly glyme-based solvents like diglyme or DME, provides an optimal environment for solubilizing the inorganic bases and stabilizing the catalytic species without interfering with the fluorine moiety. The robustness of this system is evidenced by its tolerance to a diverse range of substituents on the aromatic ring, including electron-withdrawing and electron-donating groups, which demonstrates the versatility of the catalytic manifold. Such mechanistic robustness is a key indicator of a process that can be reliably transferred from the laboratory to pilot and commercial scales without significant re-optimization.
How to Synthesize Monofluoroethyl Substituted Aromatic Compound Efficiently
The practical implementation of this synthesis involves a straightforward sequence of operations that can be readily adapted to standard reactor configurations used in fine chemical manufacturing. The process begins with the activation of the aryl boronic acid in a solvent under an inert atmosphere, followed by the controlled addition of the fluoroethyl iodide and the catalytic system. Maintaining strict temperature control and monitoring the reaction progress via fluorine NMR ensures that the endpoint is accurately determined, maximizing yield while minimizing resource consumption. The detailed standardized synthesis steps are provided in the guide below for technical reference.
- Activate aryl boronic acid with a weak base in a reaction solvent under a nitrogen atmosphere.
- Add 1-fluoro-2-iodoethane, bipyridine ligand, and nickel catalyst sequentially to the mixture.
- Heat the sealed reaction mixture to 80°C, monitor progress, and purify the product via extraction and chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this technology offers compelling advantages by fundamentally altering the cost structure and risk profile associated with fluorinated intermediate production. The substitution of expensive and hazardous fluorinating reagents with commercially abundant 1-fluoro-2-iodoethane results in a drastic simplification of the raw material portfolio, reducing both acquisition costs and the regulatory burden associated with handling toxic substances. Additionally, the use of nickel catalysts instead of palladium significantly lowers the catalyst cost, which is a major driver of expense in cross-coupling reactions, thereby enhancing the overall margin potential for downstream products. The simplicity of the workup procedure, which involves standard extraction and purification techniques, further contributes to operational efficiency by reducing processing time and solvent consumption.
- Cost Reduction in Manufacturing: The elimination of high-cost fluorinating agents like DAST and the replacement of precious metal catalysts with base metal nickel creates a substantially more economical process architecture. This shift reduces the direct material costs and minimizes the need for specialized waste treatment protocols required for hazardous byproducts, leading to significant overall cost savings in the manufacturing of fluoroethyl intermediates. The streamlined single-step nature of the reaction also reduces labor and utility costs associated with multi-step synthetic sequences.
- Enhanced Supply Chain Reliability: The reliance on readily available and stable starting materials such as aryl boronic acids and 1-fluoro-2-iodoethane ensures a robust supply chain that is less susceptible to disruptions caused by the scarcity of specialized reagents. The stability of the nickel catalyst system allows for easier storage and handling compared to air-sensitive palladium catalysts, further securing the continuity of supply. This reliability is crucial for maintaining consistent production schedules and meeting the just-in-time delivery requirements of global pharmaceutical clients.
- Scalability and Environmental Compliance: The mild reaction conditions and the absence of highly toxic reagents make this process inherently safer and easier to scale from kilogram to multi-ton production volumes. The reduced generation of hazardous waste aligns with increasingly stringent environmental regulations, facilitating smoother permitting processes and reducing the long-term liability associated with chemical manufacturing. The simplicity of the purification process also supports high throughput, enabling manufacturers to respond quickly to market demand fluctuations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this nickel-catalyzed fluorination technology. These insights are derived directly from the patent data to provide accurate guidance for R&D and procurement teams evaluating this synthetic route for their supply chains. Understanding these details is essential for assessing the feasibility of integrating this method into existing manufacturing workflows.
Q: What are the primary advantages of this nickel-catalyzed method over traditional fluorination?
A: This method avoids the use of hazardous and expensive fluorinating agents like DAST or SF4, utilizing commercially available 1-fluoro-2-iodoethane instead, which significantly enhances safety and reduces raw material costs.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent highlights mild reaction conditions, simple product separation, and the use of stable base metal catalysts, all of which are critical factors for successful commercial scale-up and process safety.
Q: What is the scope of substrate compatibility for this reaction?
A: The method demonstrates broad functional group tolerance, successfully reacting with aryl boronic acids containing alkyl, alkoxy, acyl, cyano, and heterocyclic substituents, making it versatile for diverse intermediate synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Monofluoroethyl Substituted Aromatic Compound Supplier
NINGBO INNO PHARMCHEM stands at the forefront of adopting such advanced synthetic methodologies to deliver high-quality fluorinated intermediates to the global market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this nickel-catalyzed process are fully realized at an industrial level. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of monofluoroethyl substituted aromatic compound meets the exacting standards required for pharmaceutical applications. Our commitment to process innovation allows us to offer competitive pricing without compromising on the quality or reliability of our supply.
We invite procurement leaders and technical directors to engage with our Customized Cost-Saving Analysis service to evaluate how this technology can optimize your specific supply chain. By contacting our technical procurement team, you can request specific COA data and route feasibility assessments tailored to your project needs. Let us partner with you to secure a sustainable and cost-effective source of critical fluorinated building blocks for your next generation of therapeutic agents.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
