Advanced Dibenzofuran Synthesis: Visible Light Coupling and Palladium Cyclization for Commercial Scale
Introduction to Next-Generation Dibenzofuran Manufacturing
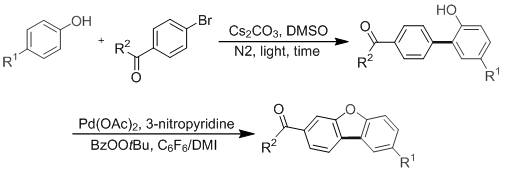
The landscape of fine chemical manufacturing is undergoing a significant transformation driven by the demand for greener, more efficient synthetic routes, particularly for complex heterocyclic scaffolds like dibenzofurans. Patent CN114560832A introduces a groundbreaking methodology that leverages visible light photocatalysis to construct biaryl ether linkages, followed by a palladium-catalyzed intramolecular cyclization to yield diverse dibenzofuran derivatives. This dual-catalytic approach represents a paradigm shift from traditional thermal processes, offering a sustainable pathway that aligns with modern environmental regulations and cost-efficiency goals. By utilizing benign energy sources such as blue LEDs or even natural sunlight, this technology minimizes the carbon footprint associated with high-temperature reactions while maintaining high conversion rates. For R&D directors and procurement specialists, understanding the nuances of this patent is crucial for evaluating its potential impact on supply chain resilience and production costs in the pharmaceutical intermediate sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of diaryl ethers and subsequent dibenzofuran structures has been plagued by inefficiencies inherent to multi-step protection strategies. Conventional wisdom dictated that the phenolic hydroxyl group must be masked with directing groups such as esters, carbamates, or ethers to facilitate regioselective arylation using precious metal catalysts like palladium, ruthenium, or gold. This necessity for protection and subsequent deprotection not only extends the synthetic timeline but also introduces additional reagents, solvents, and purification stages that inflate the overall cost of goods sold (COGS). Furthermore, the use of stoichiometric amounts of protecting group reagents generates substantial chemical waste, posing challenges for waste management and environmental compliance. These繁琐 steps often result in lower overall yields due to material loss at each isolation stage, making the final API intermediate less economically viable for large-scale manufacturing.
The Novel Approach
In stark contrast, the methodology disclosed in CN114560832A eliminates the need for hydroxyl protection entirely, enabling a direct C-O coupling between halogenated aromatics and phenols under visible light irradiation. This innovative strategy utilizes cesium carbonate as a base in dimethyl sulfoxide (DMSO) solvent, activated by blue light LEDs with a wavelength maximum of 455 nm, to generate reactive radical species that forge the biaryl ether bond at room temperature. The subsequent cyclization step employs a optimized palladium catalytic system with 3-nitropyridine as a ligand and organic peroxides as oxidants, ensuring high efficiency in closing the furan ring. This streamlined two-step sequence drastically reduces the number of unit operations, thereby enhancing the atom economy and simplifying the process workflow for chemical engineers. The ability to utilize sunlight as an energy source further underscores the sustainability profile of this novel approach, offering a distinct competitive advantage in green chemistry metrics.

Mechanistic Insights into Visible Light Coupling and Pd-Catalyzed Cyclization
The mechanistic underpinning of the first step involves a photo-induced electron transfer (PET) or energy transfer process that activates the aryl halide or the phenol substrate without the need for transition metal photocatalysts. Under blue light irradiation, the interaction between the base and the substrate likely generates a radical anion or a charge-transfer complex that facilitates the homolytic cleavage of the carbon-halogen bond. This generates an aryl radical which subsequently attacks the phenolic oxygen or undergoes a radical substitution mechanism to form the C-O bond. The absence of transition metals in this initial coupling phase is particularly advantageous for pharmaceutical applications, as it reduces the risk of heavy metal contamination in the intermediate, simplifying downstream purification requirements. The reaction proceeds efficiently under inert atmosphere (nitrogen), preventing unwanted side reactions with oxygen while maintaining high selectivity for the desired biaryl ether product.
Following the formation of the aryl phenol intermediate, the second stage involves a sophisticated palladium-catalyzed C-H activation and cyclization mechanism. The palladium(II) acetate catalyst, coordinated by the electron-deficient 3-nitropyridine ligand, activates the ortho-C-H bond adjacent to the ether linkage. The presence of tert-butyl peroxybenzoate (BzOOtBu) serves as a terminal oxidant, regenerating the active palladium species and driving the catalytic cycle forward through a Pd(II)/Pd(IV) manifold. This oxidative cyclization effectively closes the furan ring to yield the rigid dibenzofuran core. The use of a composite solvent system comprising hexafluorobenzene and 1,3-dimethyl-2-imidazolidinone (DMI) is critical for solubilizing the intermediates and stabilizing the transition states, ensuring optimal reaction kinetics at 90°C. This precise control over the catalytic environment allows for the tolerance of various functional groups, including esters, ketones, and nitriles, expanding the scope of accessible chemical space.
How to Synthesize 2-Acetyldibenzofuran Efficiently
The practical implementation of this synthesis route offers a robust protocol for producing high-purity dibenzofuran derivatives suitable for drug discovery and development. The process begins with the preparation of the aryl phenol precursor via the visible light-mediated coupling, followed by the palladium-catalyzed cyclization to finalize the heterocyclic structure. Detailed operational parameters, including reagent stoichiometry, light intensity, and temperature profiles, are critical for reproducing the high yields reported in the patent literature. For process chemists looking to adopt this technology, understanding the specific workup procedures and purification techniques is essential to maximize recovery and minimize impurity levels. The following guide outlines the standardized synthesis steps derived from the patent examples to ensure consistent quality and performance.
- Perform visible light-induced coupling of halogenated aromatic hydrocarbons and phenols using Cs2CO3 in DMSO under blue LED irradiation to form aryl phenol intermediates.
- Conduct intramolecular cyclization of the aryl phenol intermediate using Pd(OAc)2, 3-nitropyridine, and BzOOtBu in a C6F6/DMI solvent mixture at 90°C.
- Purify the final dibenzofuran product via silica gel column chromatography to achieve high purity specifications suitable for pharmaceutical applications.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this visible light-driven synthesis platform offers compelling economic benefits that extend beyond simple yield improvements. By removing the requirement for hydroxyl protecting groups, manufacturers can eliminate the purchase of expensive reagents such as acid chlorides, chloroformates, and specialized deprotection agents, leading to substantial cost savings in raw material expenditures. The simplification of the synthetic route also translates to reduced labor costs and shorter production cycles, allowing facilities to increase throughput without significant capital investment in new equipment. Furthermore, the use of commodity chemicals like cesium carbonate and DMSO ensures a stable and reliable supply chain, mitigating risks associated with the sourcing of exotic or highly regulated catalysts. This stability is crucial for maintaining continuous production schedules and meeting tight delivery deadlines for global pharmaceutical clients.
- Cost Reduction in Manufacturing: The elimination of protection and deprotection steps fundamentally alters the cost structure of dibenzofuran production by reducing the total number of synthetic operations. This reduction in step count directly correlates with lower solvent consumption, decreased energy usage for heating and cooling, and minimized waste disposal fees, all of which contribute to a leaner manufacturing budget. Additionally, the avoidance of transition metal catalysts in the first coupling step reduces the burden on metal scavenging processes, further lowering operational expenses. These cumulative efficiencies enable suppliers to offer more competitive pricing for high-purity intermediates while maintaining healthy profit margins.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials such as bromoacetophenone and phenol derivatives ensures that production is not bottlenecked by the availability of niche reagents. The robustness of the reaction conditions, which tolerate ambient temperatures and common solvents, means that the process can be easily transferred between different manufacturing sites without extensive re-validation. This flexibility enhances supply chain resilience, allowing for rapid scaling in response to market demand fluctuations. Moreover, the ability to utilize sunlight as an energy source in certain contexts provides an additional layer of security against energy price volatility, ensuring consistent operational continuity.
- Scalability and Environmental Compliance: The patent demonstrates successful gram-scale synthesis, proving the feasibility of translating this laboratory method to pilot and commercial scales. The green chemistry attributes, including the use of visible light and the generation of less hazardous byproducts, align perfectly with increasingly stringent environmental regulations globally. This compliance reduces the regulatory burden on manufacturers and facilitates faster approval times for new process validations. The simplified waste stream, devoid of complex protecting group fragments, makes effluent treatment more straightforward and cost-effective, supporting sustainable manufacturing practices that are highly valued by end-user pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this dibenzofuran synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity on reaction scope, scalability, and operational requirements. Understanding these details is vital for stakeholders evaluating the feasibility of integrating this method into their existing production portfolios. The answers reflect the specific advantages and limitations observed during the development of this novel synthetic route.
Q: What are the primary advantages of this visible light method over traditional dibenzofuran synthesis?
A: Unlike conventional methods requiring hydroxyl protection and deprotection steps, this patent describes a direct coupling route that significantly reduces synthetic steps and waste generation.
Q: Can this synthesis method be scaled for industrial production?
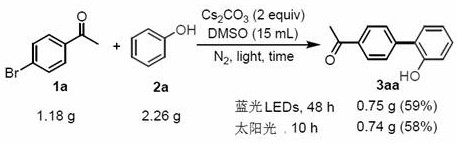
A: Yes, the patent demonstrates successful gram-scale synthesis yielding 0.75 g of product under blue light, indicating feasibility for larger commercial scale-up operations.
Q: What specific catalysts and conditions are required for the cyclization step?
A: The cyclization utilizes a palladium catalyst (Pd(OAc)2) with 3-nitropyridine as a ligand and tert-butyl peroxybenzoate as an oxidant in a composite solvent system at 90°C.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Acetyldibenzofuran Supplier
As the demand for complex heterocyclic intermediates continues to rise in the pharmaceutical and agrochemical sectors, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM ensures access to cutting-edge synthetic technologies. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, leveraging our state-of-the-art facilities to deliver compounds with stringent purity specifications. Our rigorous QC labs employ advanced analytical techniques to verify the identity and quality of every batch, ensuring that our dibenzofuran derivatives meet the exacting standards required for clinical and commercial applications. By integrating innovative methods such as the visible light coupling described in CN114560832A, we optimize our processes to provide superior value and reliability to our global partners.
We invite you to engage with our technical procurement team to discuss how our capabilities can support your specific project needs. Request a Customized Cost-Saving Analysis to understand how adopting this streamlined synthesis route can impact your bottom line. Our experts are ready to provide specific COA data and route feasibility assessments tailored to your target molecules, ensuring a seamless transition from development to commercial supply. Contact us today to explore collaborative opportunities that drive innovation and efficiency in your supply chain.
