Advanced Heterogeneous Cobalt Catalysis for High-Purity Alpha-Vinyl Silane Manufacturing and Commercial Scale-Up
Introduction to Breakthrough Hydrosilylation Technology
The landscape of organosilicon chemistry is undergoing a significant transformation driven by the urgent need for sustainable and highly selective synthetic methodologies. Patent CN111187292B introduces a pioneering approach to the synthesis of alpha-vinyl silanes, a critical structural motif widely utilized in the construction of complex pharmaceutical intermediates and advanced polymeric materials. This technology leverages a unique high-nuclearity cobalt cluster, specifically formulated as [Co14(μ3-OH)4(H2dpbt)6Cl12]•23C6H10, to achieve unprecedented levels of chemical, regio-, and stereoselectivity under heterogeneous conditions. By shifting away from expensive noble metals towards earth-abundant transition metals, this innovation addresses both economic and environmental imperatives in modern chemical manufacturing. The ability to perform these transformations at room temperature with minimal byproduct formation represents a substantial leap forward in process efficiency.
For R&D directors and process chemists, the implications of this patent are profound, offering a robust alternative to traditional hydrosilylation protocols that often suffer from poor selectivity or harsh reaction requirements. The heterogeneous nature of the catalyst not only simplifies downstream processing but also enhances the overall safety profile of the operation by eliminating the need for pyrophoric or highly toxic homogeneous catalysts. As the demand for high-purity silicon-containing building blocks continues to rise in the fine chemical sector, mastering this cobalt-catalyzed pathway becomes a strategic asset for any organization aiming to secure a reliable supply chain for next-generation materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alpha-vinyl silanes via hydrosilylation of alkynes has been plagued by significant challenges related to regiocontrol and catalyst recovery. Traditional methods frequently rely on homogeneous catalysts based on platinum, rhodium, or ruthenium, which, while effective, introduce severe bottlenecks in large-scale production due to the difficulty of removing trace metal residues from the final product. Furthermore, these conventional processes often lack the necessary selectivity, leading to complex mixtures containing beta-(Z), beta-(E), and over-hydrogenated byproducts that necessitate costly and time-consuming purification steps. Previous attempts to utilize cobalt catalysts have yielded disappointing results, with reported yields for alkyl alpha-vinyl silanes hovering around merely 38%, rendering them commercially unviable for high-volume manufacturing. The inability to effectively control the reaction selectivity has long been a persistent hurdle, limiting the utility of alpha-vinyl silanes in sensitive applications such as API synthesis where impurity profiles are strictly regulated.
The Novel Approach
The methodology disclosed in patent CN111187292B fundamentally disrupts this status quo by employing a specialized heterogeneous cobalt cluster that delivers exceptional alpha-regioselectivity. This novel catalytic system operates under mild conditions, typically at room temperature, and utilizes readily available silanes such as phenylsilane or diphenylsilane to convert a wide array of alkynes into the desired alpha-vinyl silane products with high fidelity. The heterogeneous nature of the catalyst allows for straightforward separation via filtration, effectively eliminating the need for complex metal scavenging procedures that drive up operational costs. As illustrated in the general reaction scheme below, the process achieves high atom utilization while minimizing the formation of unwanted isomers, thereby streamlining the entire production workflow.
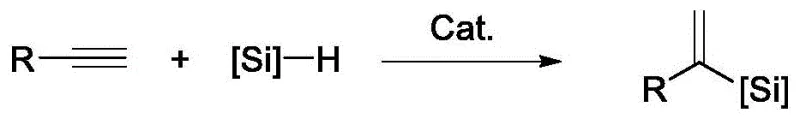
By overcoming the selectivity issues that have hindered previous cobalt-based systems, this approach opens new avenues for the efficient synthesis of structurally diverse vinyl silanes. The robustness of the catalyst is further evidenced by its ability to maintain performance over multiple recycling runs, a feature that is rarely achieved in non-precious metal catalysis. This combination of high selectivity, operational simplicity, and catalyst reusability positions the technology as a superior alternative for the cost reduction in pharmaceutical intermediate manufacturing, providing a scalable solution that aligns with green chemistry principles.
Mechanistic Insights into Co14 Cluster-Catalyzed Hydrosilylation
The exceptional performance of this synthetic route can be attributed to the unique electronic and structural properties of the high-nuclearity Co14 cluster catalyst. Unlike mononuclear cobalt complexes, this polynuclear assembly creates a distinct coordination environment that favors the activation of the Si-H bond and the subsequent insertion of the alkyne in a specific orientation. The presence of multiple metal centers likely facilitates a cooperative mechanism that stabilizes the transition state leading to the alpha-vinyl product, effectively suppressing the pathways that would otherwise lead to beta-isomers or reduced alkanes. The ligand framework, incorporating pyridyl and triazole moieties, plays a crucial role in modulating the electron density at the active sites, ensuring that the reaction proceeds with high chemoselectivity even in the presence of sensitive functional groups. Understanding this mechanistic nuance is vital for process optimization, as it explains why electronically unbiased alkylalkynes, which are notoriously difficult substrates, react efficiently under these conditions.
From an impurity control perspective, the heterogeneous nature of the catalyst provides an additional layer of quality assurance by preventing metal leaching into the product stream. In homogeneous catalysis, trace metals can often catalyze secondary decomposition reactions or interact with product functionalities, leading to stability issues during storage. By confining the catalytic activity to the solid phase, this method ensures that the resulting alpha-vinyl silanes possess a clean impurity profile, which is essential for their use in downstream cross-coupling reactions like the Hiyama coupling. The rigorous control over stereochemistry further reduces the burden on analytical teams, as the absence of E/Z isomer mixtures simplifies characterization and specification setting. This level of precision underscores the suitability of the technology for producing high-purity OLED material precursors and other specialty chemicals where structural integrity is paramount.
How to Synthesize Alpha-Vinyl Silane Efficiently
Implementing this synthesis protocol requires careful attention to reaction conditions to maximize yield and selectivity, although the procedure itself is designed to be operationally simple. The process begins with the preparation of the reaction mixture under an inert atmosphere to prevent catalyst deactivation by oxygen or moisture. Key parameters such as the ratio of silane to alkyne and the amount of base (sodium tert-butoxide) are optimized to drive the reaction to completion within a short timeframe, typically around 4 hours at ambient temperature. The detailed standardized synthesis steps see the guide below for specific molar ratios and workup procedures tailored to different substrate classes.
- Under an inert argon atmosphere, charge a reaction vessel with the heterogeneous Co14 cluster catalyst, the target alkyne substrate, and anhydrous ether or tetrahydrofuran solvent.
- Introduce the silane reagent (phenylsilane or diphenylsilane) and sodium tert-butoxide base to the mixture, ensuring strict exclusion of moisture and oxygen.
- Stir the reaction at room temperature for approximately 4 hours, then remove the solvent and purify the crude alpha-vinyl silane product via flash column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this heterogeneous cobalt catalysis technology offers tangible benefits that extend far beyond the laboratory bench. The shift from precious metals to earth-abundant cobalt significantly mitigates the risk associated with volatile raw material pricing, ensuring more stable long-term costing models for key intermediates. Furthermore, the simplified workup procedure, which eliminates the need for expensive metal scavengers and extensive purification trains, drastically reduces the overall manufacturing cycle time. This efficiency gain translates directly into improved throughput and lower operational expenditures, making the production of complex organosilicon compounds more economically attractive. The ability to recycle the catalyst multiple times further amplifies these cost savings, reducing the consumption of catalytic material per kilogram of product produced.
- Cost Reduction in Manufacturing: The elimination of noble metal catalysts such as platinum or rhodium removes a major cost driver from the bill of materials, while the heterogeneous nature of the system avoids the expense of specialized metal removal resins. By operating at room temperature, the process also minimizes energy consumption associated with heating and cooling, contributing to a leaner manufacturing footprint. The high selectivity reduces waste generation, lowering the costs associated with solvent usage and waste disposal, which are increasingly significant factors in total production costs. Consequently, this technology enables substantial cost savings in electronic chemical manufacturing without compromising on product quality or purity standards.
- Enhanced Supply Chain Reliability: Relying on cobalt, a more abundant and geographically diverse resource than platinum group metals, reduces supply chain vulnerability and geopolitical risk. The robustness of the catalyst allows for flexible production scheduling, as the reaction is not sensitive to minor fluctuations in temperature or pressure, ensuring consistent output. The simplified purification process shortens the lead time for high-purity pharmaceutical intermediates, allowing suppliers to respond more rapidly to market demand spikes. Additionally, the stability of the catalyst during storage and handling simplifies logistics, reducing the need for specialized containment or transport conditions that often delay shipments of sensitive reagents.
- Scalability and Environmental Compliance: The heterogeneous format of the catalyst is inherently scalable, facilitating the transition from gram-scale laboratory synthesis to multi-ton commercial production with minimal process re-engineering. The reduction in heavy metal waste aligns with stringent environmental regulations, simplifying the permitting process for new manufacturing lines and reducing the liability associated with hazardous waste management. The high atom economy of the hydrosilylation reaction ensures that raw materials are utilized efficiently, supporting sustainability goals and improving the overall carbon footprint of the chemical process. This alignment with green chemistry principles enhances the corporate social responsibility profile of the supply chain, appealing to end-users who prioritize sustainable sourcing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this heterogeneous hydrosilylation technology. These answers are derived directly from the experimental data and technical disclosures found within the patent literature, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this method into existing production workflows.
Q: What distinguishes this heterogeneous cobalt catalyst from traditional homogeneous systems?
A: Unlike traditional homogeneous noble metal catalysts that require complex removal steps, this high-nuclearity Co14 cluster operates heterogeneously, allowing for simple filtration and reuse while maintaining high alpha-regioselectivity.
Q: Is the catalyst recyclable for industrial applications?
A: Yes, the patent data demonstrates that the heterogeneous catalyst can be recovered and reused for at least four consecutive cycles without significant loss of catalytic activity or selectivity.
Q: What is the substrate scope for this hydrosilylation method?
A: The method exhibits broad compatibility, successfully converting electronically unbiased alkylalkynes, arylalkynes, and heteroarylalkynes into alpha-vinyl silanes with high yields and stereoselectivity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Vinyl Silane Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the one described in patent CN111187292B for the production of high-value organosilicon intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch meets the exacting standards required by the global pharmaceutical and agrochemical industries. We are uniquely positioned to leverage this cobalt-catalyzed route to deliver cost-effective and sustainable solutions for your complex synthesis needs.
We invite you to engage with our technical procurement team to discuss how this technology can be adapted to your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this heterogeneous protocol for your supply chain. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions based on real-world performance metrics. Let us collaborate to optimize your synthesis of alpha-vinyl silanes and secure a competitive advantage in the marketplace.
