Scalable Synthesis of Functionalized Alkynes Using Earth-Abundant Metal Catalysts for Industrial Applications
Scalable Synthesis of Functionalized Alkynes Using Earth-Abundant Metal Catalysts for Industrial Applications
The landscape of organic synthesis is continuously evolving towards more sustainable and cost-effective methodologies, particularly for the construction of carbon-carbon triple bonds which are ubiquitous in pharmaceutical and material science applications. A significant breakthrough in this domain is detailed in Chinese Patent CN108840785B, which discloses a novel and efficient method for synthesizing alkyne compounds through the oxidative cross-coupling of hydrazine or sulfonylhydrazide derivatives with terminal alkynes. This technology represents a paradigm shift from traditional precious metal-catalyzed processes, offering a robust alternative that utilizes inexpensive iron or copper catalysts under aerobic conditions. For R&D directors and procurement managers in the fine chemical sector, this innovation addresses critical pain points regarding catalyst cost, reaction stability, and environmental compliance, positioning it as a highly attractive route for the commercial production of high-purity pharmaceutical intermediates and advanced electronic materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of internal alkyne frameworks has relied heavily on the Sonogashira coupling reaction, which necessitates the use of palladium catalysts alongside copper co-catalysts and often requires expensive, air-sensitive phosphine ligands. While effective, this traditional approach presents substantial challenges for large-scale manufacturing, including the high cost of palladium, the stringent requirement for inert atmospheres to prevent catalyst deactivation, and the difficulty in removing trace heavy metal residues from the final active pharmaceutical ingredients. Furthermore, alternative methods such as the Glaser coupling often suffer from poor selectivity, leading to significant amounts of homocoupled byproducts that complicate downstream purification and reduce overall yield. These limitations result in inflated production costs and extended lead times, creating a bottleneck for the reliable supply of complex alkyne-based building blocks required by the global pharmaceutical and agrochemical industries.
The Novel Approach
In stark contrast to these legacy methods, the technology described in patent CN108840785B introduces a streamlined oxidative coupling strategy that leverages earth-abundant metal catalysts such as ferric chloride or copper acetate. This novel approach eliminates the dependency on precious metals and sensitive ligands, operating effectively under an atmospheric oxygen environment which drastically simplifies reactor requirements and operational safety protocols. The reaction proceeds via a radical mechanism where the sulfonylhydrazide serves as a precursor for aryl radicals, which then selectively couple with terminal alkynes to form the desired carbon-carbon triple bond. This methodology not only reduces raw material costs but also enhances process safety by avoiding the use of unstable diazo compounds or harsh halogenated reagents, thereby offering a superior pathway for the cost reduction in fine chemical manufacturing.
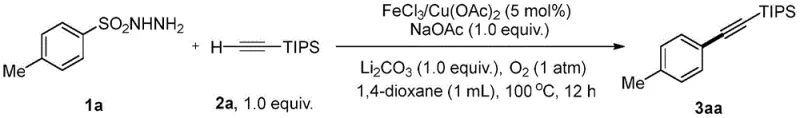
Mechanistic Insights into FeCl3-Catalyzed Oxidative Cross-Coupling
The mechanistic elegance of this synthesis lies in its ability to generate aryl radicals in situ from readily available sulfonylhydrazides under mild oxidative conditions. Upon heating in the presence of a metal catalyst like FeCl3 or Cu(OAc)2 and a base such as lithium carbonate, the sulfonylhydrazide undergoes single-electron transfer oxidation to release nitrogen gas and sulfur dioxide, generating a highly reactive aryl radical species. This radical intermediate then engages in a selective cross-coupling with the terminal alkyne substrate, effectively bypassing the competing homocoupling pathways that typically plague radical alkyne chemistry. The use of molecular oxygen as the terminal oxidant ensures that the catalytic cycle is sustained efficiently, with water being the only stoichiometric byproduct alongside nitrogen, thus adhering to the principles of green chemistry and minimizing the environmental footprint of the synthesis process.
Furthermore, the substrate scope of this reaction is exceptionally broad, accommodating a wide array of functional groups that are often problematic in transition metal-catalyzed couplings. As demonstrated in the patent examples, the system tolerates electron-rich and electron-deficient aromatic rings, heterocycles such as thiophene, and sensitive functionalities like free amino groups and halogens without the need for protective group manipulation. This high level of chemoselectivity is crucial for the synthesis of complex molecular architectures found in modern drug candidates, where the presence of multiple reactive sites can lead to impurity formation in less robust systems. The ability to synthesize diverse alkyne derivatives, including those with silyl protections or aliphatic chains, underscores the versatility of this platform for generating high-purity OLED material precursors and pharmaceutical intermediates.
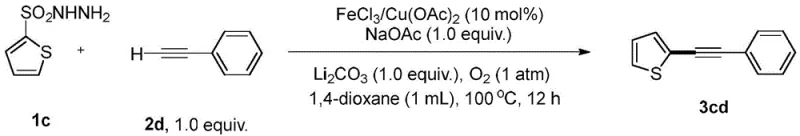
How to Synthesize Functionalized Alkynes Efficiently
Implementing this synthetic route in a laboratory or pilot plant setting involves a straightforward protocol that balances reaction efficiency with operational simplicity. The process typically begins with the charging of a reaction vessel with the sulfonylhydrazide substrate and the terminal alkyne in a molar ratio optimized for maximum conversion, often ranging from 1:1 to 2:1 depending on the specific electronic nature of the substrates. The reaction mixture is supplemented with the iron or copper catalyst, appropriate base additives like sodium acetate, and an inert solvent such as 1,4-dioxane, after which it is heated to approximately 110°C under an oxygen atmosphere. Detailed standardized synthesis steps for specific derivatives are provided in the technical guide below, ensuring reproducibility and consistency for quality control teams aiming to validate this technology for commercial adoption.
- Charge a reaction vessel with the sulfonylhydrazide compound, terminal alkyne, iron or copper catalyst (e.g., FeCl3 or Cu(OAc)2), base additives (Li2CO3, NaOAc), and 1,4-dioxane solvent.
- Heat the reaction mixture to 110°C under an atmospheric oxygen or air atmosphere for 12 to 15 hours to facilitate the oxidative cross-coupling.
- Upon completion, cool to room temperature, filter through diatomite to remove metal residues, concentrate the filtrate, and purify the crude product via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this iron or copper-catalyzed alkyne synthesis offers transformative economic and logistical benefits that directly impact the bottom line. By shifting away from palladium-based catalysis, manufacturers can achieve substantial cost savings on raw materials, as iron and copper salts are orders of magnitude cheaper than precious metal complexes and do not require specialized handling or recovery infrastructure. Additionally, the elimination of expensive phosphine ligands and the ability to run reactions under air or oxygen rather than strict inert gas conditions significantly lowers the barrier to entry for scale-up, allowing for faster turnaround times and more flexible production scheduling. These factors collectively contribute to a more resilient supply chain capable of meeting the demanding volume requirements of the global pharmaceutical market without compromising on quality or delivery reliability.
- Cost Reduction in Manufacturing: The replacement of palladium catalysts with inexpensive iron or copper salts results in a drastic reduction in catalyst loading costs, which is particularly significant for large-volume production runs. Moreover, the simplified workup procedure, which involves basic filtration and chromatography without the need for complex metal scavenging resins, further reduces processing expenses and solvent consumption. This economic efficiency allows suppliers to offer more competitive pricing structures for key alkyne intermediates, enhancing the overall profitability of downstream drug synthesis campaigns.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable starting materials, such as sulfonylhydrazides and terminal alkynes, ensures a consistent and secure supply of feedstock, mitigating the risks associated with the scarcity of specialized reagents. The robustness of the reaction conditions, which tolerate moisture and oxygen to a greater extent than traditional methods, reduces the likelihood of batch failures due to environmental fluctuations, thereby guaranteeing a steady flow of high-quality intermediates to customers. This reliability is critical for maintaining continuous manufacturing operations and avoiding costly production delays in the pharmaceutical value chain.
- Scalability and Environmental Compliance: The generation of benign byproducts like nitrogen and water aligns perfectly with increasingly stringent environmental regulations, simplifying waste treatment processes and reducing the regulatory burden on manufacturing facilities. The absence of toxic heavy metal waste streams facilitates easier disposal and recycling of solvents, supporting sustainability goals and corporate social responsibility initiatives. Furthermore, the scalability of the process from gram to kilogram scales has been validated through diverse substrate examples, confirming its readiness for commercial scale-up of complex polymer additives and electronic chemical precursors.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this alkyne synthesis technology, providing clarity for technical teams evaluating its potential integration into existing workflows. These insights are derived directly from the experimental data and claims presented in the patent literature, ensuring accuracy and relevance for decision-makers. Understanding these nuances is essential for optimizing reaction parameters and maximizing yield when transitioning from bench-scale discovery to pilot production.
Q: What are the primary advantages of this method over traditional Sonogashira coupling?
A: This method eliminates the need for expensive palladium catalysts and phosphine ligands, utilizing earth-abundant iron or copper instead. It operates under aerobic conditions without strict inert gas protection, significantly reducing operational costs and complexity while producing only nitrogen and water as byproducts.
Q: Does this synthesis tolerate sensitive functional groups like amines or halogens?
A: Yes, the protocol demonstrates excellent functional group tolerance. Specific embodiments confirm compatibility with amino groups, bromo substituents, cyano groups, and heterocycles like thiophene, making it highly suitable for synthesizing complex pharmaceutical intermediates without extensive protecting group strategies.
Q: What is the atom economy and environmental impact of this process?
A: The process exhibits high atom economy as the hydrazide moiety is expelled as harmless nitrogen gas, and the oxidation byproduct is water. This aligns with green chemistry principles, minimizing hazardous waste generation compared to traditional halogen-based coupling methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alkyne Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of innovative synthetic methodologies like the one described in CN108840785B for advancing the development of next-generation therapeutics and materials. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of intermediates that meet stringent purity specifications. Our state-of-the-art rigorous QC labs are equipped to analyze and verify the quality of alkyne compounds synthesized via this iron-copper catalytic route, guaranteeing that every batch adheres to the highest industry standards for impurity profiles and chemical identity.
We invite pharmaceutical and chemical companies to collaborate with us to leverage this cost-effective and green synthesis platform for their specific project needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your target molecule, along with specific COA data and route feasibility assessments. Let us help you optimize your supply chain and accelerate your time-to-market with our reliable alkyne compound supply solutions, combining cutting-edge chemistry with unwavering commitment to quality and service excellence.
