Advanced Synthesis of Ferrocenyl Aminopyrimidines for Commercial Scale-Up and Drug Discovery
Introduction to Novel Ferrocenyl Pyrimidine Technology
The pharmaceutical landscape is constantly evolving, driven by the need for novel scaffolds that offer enhanced biological activity and favorable safety profiles. Patent CN109081856B introduces a significant advancement in the synthesis of 4-ferrocenyl-6-aryl-2-aminopyrimidine compounds, a class of heterocyclic molecules that merge the unique redox properties of ferrocene with the pharmacophore versatility of the pyrimidine ring. This technology addresses critical challenges in medicinal chemistry by providing a robust, high-yield pathway to access these complex structures, which have demonstrated promising inhibitory activity against prostate cancer PC-3 cells. For R&D directors and procurement specialists, understanding the nuances of this synthetic route is essential for securing a reliable pharmaceutical intermediate supplier capable of delivering high-quality materials for drug discovery programs. The innovation lies not just in the final molecule, but in the efficiency and scalability of the preparation method, which utilizes readily available starting materials and mild reaction conditions to achieve superior outcomes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of substituted pyrimidine rings often involves harsh reaction conditions, including the use of strong acids, high temperatures, or expensive transition metal catalysts that complicate downstream purification. Conventional routes may suffer from poor atom economy, generating significant amounts of hazardous waste that increase disposal costs and environmental liability. Furthermore, the introduction of sensitive organometallic groups like ferrocene typically requires stringent anhydrous conditions and inert atmospheres to prevent decomposition, leading to lower overall yields and inconsistent batch-to-batch reproducibility. These factors create bottlenecks in the supply chain, extending lead times for high-purity pharmaceutical intermediates and inflating the cost of goods sold. For procurement managers, these inefficiencies translate into higher raw material costs and greater supply risk, making it difficult to secure a stable source of complex heterocyclic building blocks for clinical trial materials.
The Novel Approach
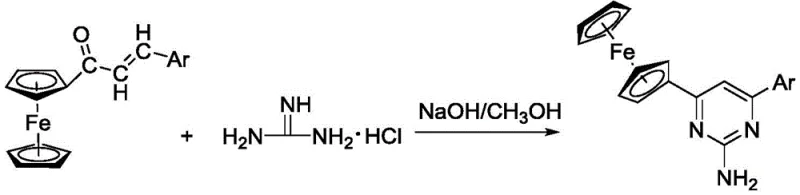
In contrast, the methodology disclosed in CN109081856B offers a streamlined alternative that leverages the nucleophilicity of guanidine hydrochloride under basic conditions to drive the cyclization efficiently. By employing methanol as a solvent and heating to reflux, the process achieves excellent conversion rates without the need for exotic reagents or extreme pressures. The reaction tolerates a wide range of substituents on the aryl ring, including electron-withdrawing groups like nitro and halogens, as well as electron-donating groups, demonstrating remarkable substrate scope. This flexibility allows for the rapid generation of diverse compound libraries, accelerating the structure-activity relationship (SAR) studies crucial for modern drug development. The simplicity of the workup, involving basic aqueous quenching and standard extraction, further enhances the operational ease, making it an ideal candidate for cost reduction in API manufacturing where process robustness is paramount.
Mechanistic Insights into Base-Promoted Cyclization
The reaction mechanism proceeds through a sophisticated sequence of nucleophilic attacks and rearrangements that ensure high regioselectivity and minimal byproduct formation. Initially, the lone pair electrons on the nitrogen atoms of the guanidine derivative act as a potent nucleophile, attacking the electrophilic beta-carbon of the ferrocenyl chalcone alpha,beta-unsaturated ketone system. This Michael-type addition generates a key intermediate that positions the reactive centers in close proximity for the subsequent intramolecular cyclization. The presence of the alkali base facilitates the deprotonation steps necessary to drive the equilibrium forward, while the ferrocene moiety remains stable throughout the process due to the mild pH environment. Understanding this mechanistic pathway is vital for R&D teams aiming to optimize reaction parameters, as it highlights the importance of maintaining the correct stoichiometric ratio between the chalcone, guanidine, and base to prevent side reactions such as polymerization or hydrolysis.
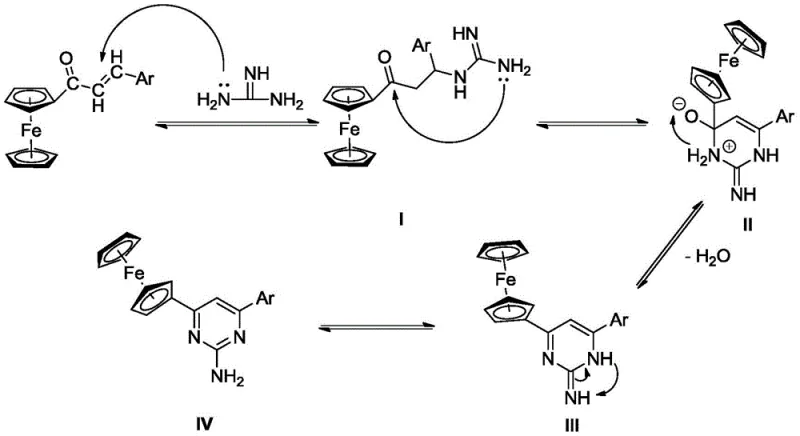
Following the initial addition, the intermediate undergoes an intramolecular condensation where the amino group attacks the carbonyl carbon, forming the six-membered pyrimidine ring. The final step involves a dehydration and aromatization process, driven by the thermodynamic stability of the aromatic pyrimidine system and the ferrocene unit. This cascade effectively locks the structure into the desired 2-aminopyrimidine configuration, minimizing the formation of isomeric impurities that are common in less controlled syntheses. The mechanism also explains the observed tolerance for various functional groups; since the reaction does not rely on aggressive oxidants or reductants, sensitive moieties like nitro groups or halides remain intact. This level of control over the impurity profile is critical for meeting the stringent purity specifications required by regulatory agencies for pharmaceutical ingredients, ensuring that the final product is safe for biological evaluation.
How to Synthesize 4-Ferrocenyl-6-aryl-2-aminopyrimidine Efficiently
The practical execution of this synthesis is designed for operational simplicity, allowing chemists to reproduce the results with high fidelity across different scales. The process begins with the dissolution of the ferrocenyl chalcone precursor in methanol, ensuring a homogeneous solution that maximizes contact between reactants. Upon the addition of guanidine hydrochloride and the alkali base, the mixture is heated to reflux, a condition that provides sufficient thermal energy to overcome the activation barrier for cyclization while keeping the solvent volume manageable. Monitoring the reaction progress via Thin Layer Chromatography (TLC) allows for precise determination of the endpoint, preventing over-reaction or degradation of the product. Once complete, the addition of water induces precipitation or phase separation, facilitating the isolation of the crude product which is then purified to pharmaceutical grade.
- Dissolve ferrocenyl chalcone in methanol at room temperature with stirring.
- Add guanidine hydrochloride and alkali base, then heat to reflux while monitoring by TLC.
- Quench with water, extract organic phase, dry, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers substantial benefits that directly impact the bottom line and supply chain resilience for pharmaceutical manufacturers. The reliance on commodity chemicals like methanol, potassium hydroxide, and guanidine hydrochloride means that raw material costs are kept low and sourcing is not dependent on single-supplier bottlenecks. The elimination of expensive transition metal catalysts removes the need for costly metal scavenging steps and rigorous testing for residual metals, which are significant cost drivers in API production. Furthermore, the high yields reported in the patent examples, often exceeding 70% and reaching nearly 95% in optimized cases, translate to better material throughput and reduced waste generation. For supply chain heads, this efficiency means shorter production cycles and the ability to respond quickly to fluctuating demand without compromising on quality or delivery schedules.
- Cost Reduction in Manufacturing: The process utilizes inexpensive, widely available reagents and avoids the use of precious metal catalysts, which significantly lowers the direct material costs associated with production. Additionally, the simplified workup procedure reduces labor hours and solvent consumption, leading to substantial cost savings in utility and waste disposal expenses. By streamlining the synthesis into fewer steps with high conversion rates, the overall cost of goods is minimized, making the final intermediate more competitive in the global market.
- Enhanced Supply Chain Reliability: Since the starting materials are bulk chemicals with established supply chains, the risk of raw material shortages is drastically reduced compared to routes requiring specialized or custom-synthesized reagents. The robustness of the reaction conditions ensures consistent output even with minor variations in input quality, providing a stable supply of critical intermediates for downstream drug formulation. This reliability is essential for maintaining continuous manufacturing operations and meeting strict delivery deadlines for clinical and commercial batches.
- Scalability and Environmental Compliance: The use of methanol as a primary solvent and the absence of hazardous heavy metals make this process environmentally friendly and easier to permit for large-scale production. The reaction can be safely scaled from gram to kilogram quantities using standard stainless steel reactors, facilitating the commercial scale-up of complex pharmaceutical intermediates. Moreover, the reduced generation of toxic byproducts aligns with green chemistry principles, helping companies meet increasingly stringent environmental regulations and sustainability goals.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating this technology for their specific applications, we have compiled answers to common questions regarding the synthesis and properties of these ferrocenyl pyrimidines. These insights are derived directly from the experimental data and mechanistic understanding provided in the patent literature, ensuring accuracy and relevance for process development. Whether you are concerned about reaction safety, purification methods, or biological activity, the following information addresses the key technical considerations for implementing this route in your facility.
Q: What are the key advantages of this synthesis method over traditional routes?
A: This method utilizes mild reaction conditions (methanol reflux) and inexpensive reagents like guanidine hydrochloride, avoiding harsh catalysts and significantly simplifying the workup procedure compared to traditional high-temperature cyclizations.
Q: What is the expected yield and purity profile for these intermediates?
A: The patent data indicates yields consistently exceeding 70%, with specific examples reaching up to 94.8%. The use of column chromatography ensures high-purity specifications suitable for downstream pharmaceutical applications.
Q: Can this process be scaled for industrial production?
A: Yes, the process relies on standard unit operations such as reflux, liquid-liquid extraction, and distillation, which are easily transferable from laboratory scale to multi-ton commercial production without requiring specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Ferrocenyl-6-aryl-2-aminopyrimidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of high-quality intermediates in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can support your project from early-stage discovery through to full-scale manufacturing. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 4-ferrocenyl-6-aryl-2-aminopyrimidine meets the highest industry standards. Our commitment to technical excellence allows us to navigate the complexities of organometallic chemistry, delivering products that enable your research to proceed without interruption.
We invite you to collaborate with us to leverage this advanced synthetic technology for your drug discovery pipeline. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your supply chain efficiency and reduce your overall development costs.
