Advanced Palladium-Catalyzed Synthesis of 2-Phenyl Quinazolinones for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust synthetic methodologies for nitrogen-containing heterocycles, particularly quinazolinones, due to their profound biological activities ranging from antimicrobial to anticancer properties. Patent CN108129402B introduces a groundbreaking approach for synthesizing 2-phenyl quinazolinone compounds by utilizing readily available tolane compounds and 2-aminobenzamide as starting materials. This innovative protocol leverages a palladium-catalyzed oxidative cleavage strategy under an oxygen atmosphere, effectively breaking the carbon-carbon triple bond of the alkyne to generate a transient benzaldehyde intermediate which subsequently condenses with the amide. This method represents a significant departure from classical condensation reactions that often require pre-functionalized aldehydes, offering a streamlined pathway that enhances atom economy and operational simplicity for the production of high-value pharmaceutical intermediates.
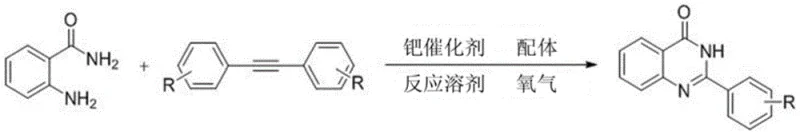
The development of efficient synthetic routes for quinazolinone derivatives is critical for drug discovery programs targeting resistant bacterial strains and various cancers. By employing molecular oxygen as the terminal oxidant, this technology not only reduces the reliance on stoichiometric heavy metal oxidants but also aligns with modern green chemistry principles. The versatility of this method is demonstrated by its tolerance to various substituents on the aromatic rings, including electron-donating and electron-withdrawing groups, ensuring broad applicability across diverse medicinal chemistry campaigns. For reliable pharmaceutical intermediate suppliers, mastering such transformative C-C bond activation techniques is essential for maintaining a competitive edge in the global supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic strategies for constructing the quinazolinone scaffold often rely on the condensation of 2-aminobenzamide with pre-formed benzaldehydes or benzonitriles, which can present significant logistical and economic challenges. The preparation of substituted benzaldehydes frequently involves multi-step sequences requiring harsh oxidation conditions or the use of toxic reagents, thereby increasing the overall cost of goods and environmental footprint. Furthermore, many classical methods suffer from limited substrate scope, where sensitive functional groups may not survive the rigorous reaction conditions, leading to complex impurity profiles that are difficult to purge. In industrial settings, the handling of volatile aldehydes poses safety risks, and the need for stoichiometric oxidants generates substantial waste streams, complicating downstream processing and regulatory compliance for cost reduction in pharmaceutical manufacturing.
The Novel Approach
The methodology disclosed in the patent circumvents these hurdles by utilizing stable diphenylacetylene (tolane) derivatives as latent aldehyde equivalents, which are activated in situ through a sophisticated palladium-catalyzed cycle. This approach eliminates the need to isolate reactive aldehyde intermediates, thereby simplifying the operational workflow and reducing the potential for side reactions associated with free aldehydes. The use of molecular oxygen as the sole oxidant ensures that the only byproduct is water, drastically minimizing waste generation and enhancing the sustainability profile of the process. Moreover, the reaction proceeds under relatively mild thermal conditions (60-110°C) with high efficiency, achieving yields up to 90% for certain substrates, which underscores its potential for commercial scale-up of complex pharmaceutical intermediates. This strategic shift from pre-functionalized building blocks to direct alkyne activation marks a pivotal advancement in heterocyclic chemistry.
Mechanistic Insights into Pd-Catalyzed Oxidative Cleavage and Cyclization
The core of this transformation lies in the unique ability of the palladium catalyst to mediate the oxidative cleavage of the carbon-carbon triple bond in the tolane substrate. Under an oxygen atmosphere, the palladium species coordinates with the alkyne, facilitating the insertion of oxygen and subsequent bond scission to generate a benzaldehyde species within the reaction matrix. This transient aldehyde immediately engages in a nucleophilic attack by the amino group of the 2-aminobenzamide, initiating a cascade of condensation and cyclization events that forge the quinazolinone ring system. The presence of a bidentate nitrogen ligand, specifically 2,2'-bipyridine, is crucial for stabilizing the active palladium center and modulating its electronic properties to favor the desired cleavage pathway over competing polymerization or homocoupling reactions. Understanding this mechanistic nuance allows chemists to fine-tune reaction parameters for optimal performance.
Impurity control in this system is inherently managed by the specificity of the catalytic cycle and the choice of reaction conditions. The use of Pd(TFA)2 as the catalyst precursor, rather than simpler salts like PdCl2, appears to enhance the solubility and reactivity of the metal center in the organic phase, leading to cleaner conversion profiles. Experimental data suggests that deviations from the optimal ligand-to-catalyst ratio or the use of inappropriate solvents can lead to incomplete conversion or the formation of side products, emphasizing the importance of precise process control. The reaction's dependence on oxygen pressure and temperature further dictates the kinetics of the oxidative cleavage step, ensuring that the rate of aldehyde generation matches the rate of condensation, thereby preventing the accumulation of reactive intermediates that could lead to oligomerization. This delicate balance ensures the production of high-purity pharmaceutical intermediates suitable for stringent regulatory standards.
How to Synthesize 2-Phenyl Quinazolinone Efficiently
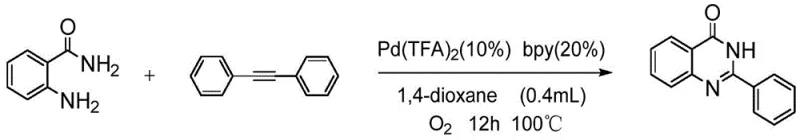
The practical execution of this synthesis requires careful attention to the order of addition and the maintenance of an oxygen-rich environment to drive the catalytic cycle forward. The standard protocol involves charging the reactor with the solid reagents and catalyst system, followed by degassing and backfilling with oxygen to ensure an inert yet oxidative atmosphere. The selection of 1,4-dioxane as the solvent is paramount, as screening data indicates that polar aprotic solvents significantly outperform non-polar alternatives like toluene or halogenated solvents in terms of yield and reaction rate. Once the reaction reaches completion, typically monitored by TLC or HPLC, the workup is straightforward, involving solvent removal and purification via standard chromatographic techniques. For detailed operational parameters and safety guidelines, please refer to the standardized synthesis steps outlined below.
- Charge the reactor with 2-aminobenzamide, tolane substrate, Pd(TFA)2 catalyst, and bipyridine ligand, then perform vacuum-oxygen exchange cycles.
- Add 1,4-dioxane solvent and heat the mixture to 100°C under an oxygen atmosphere for approximately 12 hours to facilitate oxidative cleavage and cyclization.
- Remove the solvent under reduced pressure and purify the crude residue via silica gel column chromatography to isolate the high-purity quinazolinone product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this synthetic route offers compelling advantages by leveraging commodity chemicals as starting materials. Tolane derivatives and 2-aminobenzamide are widely available in the global chemical market, ensuring a stable supply chain that is less susceptible to the volatility often seen with specialized reagents. The elimination of expensive stoichiometric oxidants and the use of ambient oxygen significantly reduce the raw material costs associated with the oxidation step. Furthermore, the simplified workup procedure, which avoids complex aqueous extractions or hazardous quenching steps, translates to reduced labor costs and shorter batch cycle times. These factors collectively contribute to a more economical manufacturing process, allowing for cost reduction in pharmaceutical manufacturing without compromising on product quality or yield.
- Cost Reduction in Manufacturing: The utilization of molecular oxygen as a green oxidant removes the financial burden associated with purchasing and disposing of traditional heavy metal oxidants or peroxides. Additionally, the high atom economy of the reaction, where the majority of the substrate atoms are incorporated into the final product, minimizes waste disposal costs. The robustness of the catalyst system allows for efficient conversion even at moderate loadings, further optimizing the cost structure. By streamlining the synthesis from multiple steps to a single pot operation, manufacturers can achieve significant savings in energy consumption and equipment usage, driving down the overall cost of goods sold for quinazolinone-based active ingredients.
- Enhanced Supply Chain Reliability: Sourcing stability is a critical concern for supply chain managers, and this method mitigates risk by relying on bulk chemicals with established production capacities. Unlike specialized aldehyde intermediates that may have limited suppliers, tolane compounds are produced on a large scale for various industries, ensuring consistent availability and competitive pricing. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, reducing the likelihood of batch failures. This reliability ensures reducing lead time for high-purity pharmaceutical intermediates and supports just-in-time manufacturing strategies essential for meeting tight project deadlines in drug development.
- Scalability and Environmental Compliance: The transition from laboratory to pilot and commercial scale is facilitated by the simplicity of the reaction setup, which does not require specialized high-pressure equipment or cryogenic conditions. The use of oxygen gas is easily managed with standard sparging techniques, and the absence of toxic byproducts simplifies effluent treatment processes. This alignment with green chemistry principles not only reduces the environmental impact but also eases the regulatory burden associated with waste management and emissions. Consequently, the process is highly scalable, enabling manufacturers to rapidly ramp up production volumes to meet market demand while maintaining strict adherence to environmental, health, and safety (EHS) standards.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this palladium-catalyzed synthesis. These insights are derived directly from the experimental data and comparative studies presented in the patent documentation, providing a clear understanding of the method's capabilities and limitations. Addressing these points helps stakeholders evaluate the feasibility of adopting this technology for their specific production needs.
Q: What is the primary advantage of using tolane compounds over traditional aldehyde precursors?
A: Tolane compounds serve as stable, inexpensive precursors that undergo in situ oxidative cleavage to generate the reactive aldehyde species, eliminating the need for handling unstable or expensive benzaldehyde derivatives directly.
Q: Which palladium catalyst system provides the optimal yield for this transformation?
A: Experimental data indicates that Palladium Trifluoroacetate (Pd(TFA)2) combined with 2,2'-bipyridine (bpy) as a ligand delivers superior yields (up to 82%) compared to other palladium sources like acetate or chloride.
Q: Is this synthetic route suitable for large-scale manufacturing?
A: Yes, the process utilizes molecular oxygen as a green oxidant and simple workup procedures like solvent evaporation and chromatography, making it highly amenable to scale-up for industrial production of quinazolinone APIs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Phenyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient synthetic routes like the one described in CN108129402B for the rapid development of novel therapeutic agents. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless. We are committed to delivering stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 2-phenyl quinazolinone intermediate meets the highest industry standards. Our state-of-the-art facilities are equipped to handle complex palladium-catalyzed reactions safely and efficiently, providing you with a dependable source for your critical building blocks.
We invite you to collaborate with us to leverage this advanced technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized processes can accelerate your timeline and enhance your bottom line. Let us be your partner in turning innovative chemistry into commercial success.
