Advanced Synthesis of 1-Alkylpyrazole-4-Boronic Acid Pinacol Esters for Commercial Pharma Applications
The pharmaceutical industry continuously seeks robust and scalable pathways for constructing heterocyclic scaffolds, particularly pyrazole derivatives which serve as critical building blocks in modern drug discovery. Patent CN103601749A discloses a transformative synthetic methodology for producing 1-alkylpyrazole-4-boronic acid pinacol esters, addressing significant bottlenecks found in traditional manufacturing processes. This innovation leverages a strategic three-step sequence involving electrophilic iodination, N-alkylation, and a mild halogen-magnesium exchange to deliver high-purity intermediates suitable for Suzuki-Miyaura cross-coupling. By shifting away from cryogenic lithiation and expensive transition metal catalysis, this technology offers a compelling value proposition for reliable pharmaceutical intermediates supplier networks aiming to optimize their supply chains. The ability to operate at manageable temperatures while maintaining exceptional chemical fidelity makes this approach highly attractive for the commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of pyrazole boronic esters has been plagued by severe operational constraints and economic inefficiencies that hinder large-scale production. One prevalent legacy method relies on the use of n-butyllithium to effect lithiation at the C4 position, necessitating ultra-low temperature conditions around -90°C to prevent decomposition and ensure regioselectivity. 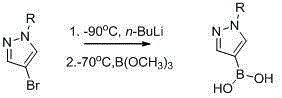 Such cryogenic requirements impose a massive burden on reactor infrastructure, demanding specialized cooling systems and significantly increasing energy consumption and operational risk. Furthermore, alternative palladium-catalyzed borylation routes, while avoiding extreme cold, introduce prohibitive costs due to the reliance on precious metal catalysts like PdCl2(dppf)CH2Cl2 and expensive bis(pinacolato)diboron reagents. These transition metal methods also create substantial downstream processing challenges, requiring rigorous purification steps to reduce residual metal content to ppm levels acceptable for API manufacturing, thereby complicating the cost reduction in pharmaceutical intermediates manufacturing.
Such cryogenic requirements impose a massive burden on reactor infrastructure, demanding specialized cooling systems and significantly increasing energy consumption and operational risk. Furthermore, alternative palladium-catalyzed borylation routes, while avoiding extreme cold, introduce prohibitive costs due to the reliance on precious metal catalysts like PdCl2(dppf)CH2Cl2 and expensive bis(pinacolato)diboron reagents. These transition metal methods also create substantial downstream processing challenges, requiring rigorous purification steps to reduce residual metal content to ppm levels acceptable for API manufacturing, thereby complicating the cost reduction in pharmaceutical intermediates manufacturing.
The Novel Approach
The patented methodology introduces a streamlined and economically superior pathway that circumvents the drawbacks of both cryogenic lithiation and palladium catalysis. 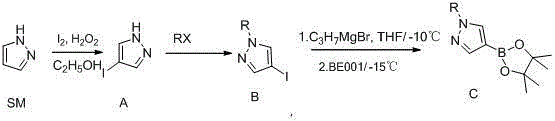 By utilizing a halogen-magnesium exchange strategy with isopropyl Grignard reagent, the reaction can proceed efficiently at temperatures between 0°C and -30°C, a range that is easily achievable with standard industrial cooling media rather than liquid nitrogen. This shift not only enhances process safety but also drastically simplifies the engineering controls required for production. The route begins with the inexpensive iodination of pyrazole using molecular iodine and hydrogen peroxide, followed by straightforward alkylation, before the final boronation step. This logical progression ensures that high-purity 1-alkylpyrazole-4-boronic acid pinacol ester is obtained without the need for exotic reagents, positioning this technology as a benchmark for reducing lead time for high-purity pharmaceutical intermediates.
By utilizing a halogen-magnesium exchange strategy with isopropyl Grignard reagent, the reaction can proceed efficiently at temperatures between 0°C and -30°C, a range that is easily achievable with standard industrial cooling media rather than liquid nitrogen. This shift not only enhances process safety but also drastically simplifies the engineering controls required for production. The route begins with the inexpensive iodination of pyrazole using molecular iodine and hydrogen peroxide, followed by straightforward alkylation, before the final boronation step. This logical progression ensures that high-purity 1-alkylpyrazole-4-boronic acid pinacol ester is obtained without the need for exotic reagents, positioning this technology as a benchmark for reducing lead time for high-purity pharmaceutical intermediates.
Mechanistic Insights into Grignard-Mediated Halogen Exchange
The core chemical innovation lies in the selective halogen-magnesium exchange at the C4 position of the pyrazole ring, which is facilitated by the use of isopropylmagnesium bromide. Unlike lithium-halogen exchange which often requires kinetic control at extremely low temperatures to avoid side reactions such as nucleophilic attack on the ring or deprotonation, the magnesium species exhibits a favorable thermodynamic profile that allows the exchange to occur rapidly at moderate sub-zero temperatures. The mechanism involves the coordination of the magnesium center to the iodine atom, followed by a concerted transfer of the alkyl group to the magnesium and the iodine to the isopropyl group, generating the reactive pyrazolyl-magnesium intermediate in situ. This intermediate is then immediately trapped by isopropyl pinacol borate (BE001), forming the stable boronic ester bond. The choice of isopropyl Grignard is critical as it minimizes competing nucleophilic addition to the electron-deficient pyrazole ring, ensuring high chemoselectivity and yield.
Impurity control is meticulously managed through the specific sequence of reagents and workup conditions defined in the patent. The initial iodination step utilizes hydrogen peroxide as a green oxidant, which minimizes the formation of poly-iodinated byproducts that are common when using stronger oxidizing agents. During the final boronation step, the reaction mixture is quenched and subjected to aqueous washing protocols that effectively remove magnesium salts and unreacted boron species. The use of n-heptane for slurry purification in the final isolation step is particularly effective at rejecting organic impurities that possess different solubility profiles than the target boronic ester. This multi-stage purification strategy ensures that the final product consistently achieves purity levels greater than 98%, meeting the stringent quality standards required for high-purity pharmaceutical intermediates used in sensitive cross-coupling reactions.
How to Synthesize 1-Alkylpyrazole-4-Boronic Acid Pinacol Ester Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing precise temperature control and stoichiometric balance to maximize yield. The process begins with the preparation of 4-iodopyrazole, followed by N-alkylation to install the desired substituent, and concludes with the Grignard exchange and boronation. Operators must strictly adhere to the specified temperature ranges, particularly during the exothermic iodination and the moisture-sensitive Grignard steps, to ensure reproducibility. For detailed operational parameters, reagent grades, and specific workup instructions, please refer to the standardized synthesis guide below which encapsulates the critical process parameters for successful execution.
- Prepare 4-iodopyrazole intermediate by reacting pyrazole with iodine and hydrogen peroxide in ethanol at controlled temperatures below 70°C.
- Perform N-alkylation of the 4-iodopyrazole intermediate using haloalkanes and a base to generate the 1-alkyl-4-iodopyrazole precursor.
- Execute halogen-magnesium exchange using isopropyl Grignard reagent at 0 to -30°C, followed by quenching with isopropyl pinacol borate to yield the final ester.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this synthetic route offers profound advantages that directly address the pain points of cost volatility and supply chain fragility in the fine chemical sector. By eliminating the dependency on palladium catalysts, manufacturers can insulate themselves from the fluctuating market prices of precious metals and avoid the long lead times often associated with sourcing high-grade catalytic complexes. Additionally, the replacement of n-butyllithium with Grignard reagents reduces the safety hazards associated with pyrophoric materials, leading to lower insurance premiums and reduced regulatory compliance burdens. The use of commodity chemicals such as iodine, hydrogen peroxide, and simple haloalkanes ensures a robust and diversified supply base, significantly enhancing supply chain reliability and reducing the risk of production stoppages due to raw material shortages.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the substitution of expensive catalytic systems with stoichiometric reagents that are readily available at bulk commodity prices. Removing the palladium catalyst not only saves on the initial reagent cost but also eliminates the downstream expense of metal scavenging resins and the associated yield losses during purification. Furthermore, the ability to run the critical boronation step at 0 to -30°C instead of -90°C results in substantial energy savings, as standard glycol cooling systems can be utilized instead of cryogenic liquid nitrogen setups. These cumulative efficiencies translate into a significantly lower cost of goods sold, allowing for more competitive pricing in the global market for pharmaceutical building blocks.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials such as pyrazole, iodine, and common alkyl halides ensures that production is not bottlenecked by niche suppliers. Unlike specialized boron reagents or air-sensitive organolithiums which may have limited global production capacity, the reagents used in this protocol are produced by multiple chemical manufacturers worldwide. This diversification of the supply base mitigates the risk of single-source failure and ensures consistent availability of raw materials even during periods of market disruption. Consequently, partners can rely on a stable and continuous supply of the final boronic ester intermediates, supporting uninterrupted drug development timelines and commercial manufacturing schedules.
- Scalability and Environmental Compliance: The mild reaction conditions and simplified workup procedures make this process inherently scalable from kilogram to multi-ton production volumes without requiring disproportionate increases in capital expenditure. The avoidance of heavy metals aligns with increasingly strict environmental regulations regarding waste disposal and residual limits in active pharmaceutical ingredients. By generating less hazardous waste and avoiding persistent organic pollutants associated with certain catalytic ligands, this method supports greener manufacturing practices. The straightforward isolation via crystallization or slurry further reduces solvent consumption compared to extensive chromatographic purifications, contributing to a reduced environmental footprint and easier compliance with sustainability mandates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing clarity on yield expectations, purity profiles, and operational requirements. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their specific project needs.
Q: Why is the Grignard exchange method preferred over n-BuLi lithiation for pyrazole boronic esters?
A: The Grignard exchange method operates at significantly milder temperatures (0 to -30°C) compared to the ultra-low cryogenic conditions (-90°C) required for n-BuLi lithiation. This reduces energy consumption, minimizes safety risks associated with cryogenic operations, and simplifies the engineering requirements for commercial scale-up.
Q: How does this synthesis route impact the cost of goods for pharmaceutical intermediates?
A: This route eliminates the need for expensive palladium catalysts and precious metal ligands typically used in Suzuki-Miyaura borylation. Furthermore, it utilizes commodity chemicals like iodine and hydrogen peroxide for the initial functionalization, substantially lowering raw material costs and removing the need for costly heavy metal scavenging steps.
Q: What purity levels can be achieved with this synthetic methodology?
A: The described process consistently achieves purity levels exceeding 97% to 98% as confirmed by GC and 1H-NMR analysis. The specific workup procedures, including aqueous washes and slurry purification, effectively remove inorganic salts and organic byproducts, ensuring the material meets stringent specifications for downstream cross-coupling reactions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Alkylpyrazole-4-Boronic Acid Pinacol Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality heterocyclic building blocks play in accelerating drug discovery and development. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify every batch. Our facility is equipped to handle the specific thermal and safety requirements of Grignard chemistry, guaranteeing a safe and compliant manufacturing environment for your most sensitive projects.
We invite you to collaborate with us to leverage this advanced synthetic technology for your upcoming programs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing processes can enhance your supply chain efficiency and reduce overall project costs.
