Advanced Synthetic Route for Rebamipide Intermediates: Enhancing Purity and Scalability
Introduction to Advanced Intermediate Manufacturing
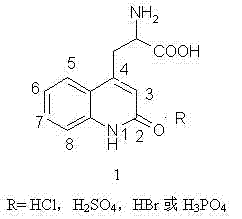
The pharmaceutical industry continuously seeks robust synthetic pathways that balance high purity with economic viability, particularly for gastroprotective agents like Rebamipide. Patent CN102153512B introduces a significant technological advancement in the preparation of 2-amino-3-(2(1H)-quinolone-4-yl) propionic acid salts, which serve as the critical backbone for Rebamipide synthesis. This innovation addresses longstanding inefficiencies in heterocyclic chemistry by replacing harsh, single-acid hydrolysis conditions with a sophisticated mixed-acid system. By integrating organic acids such as acetic or formic acid with inorganic mineral acids, the process fundamentally alters the solvation dynamics of the reaction medium. This strategic modification not only enhances the solubility of key intermediates but also mitigates the risk of uncontrolled decarboxylation, a common failure mode in prior art methodologies. For global procurement teams, this represents a shift towards more predictable and scalable supply chains for high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical approaches to synthesizing this quinolone-based intermediate have been plagued by operational complexities and suboptimal yields that hinder industrial adoption. Early methodologies, such as those described in JP2004131506, relied on a cumbersome sequence involving two distinct hydrolysis steps, which significantly increased processing time and solvent consumption. Furthermore, alternative routes like US4578381 attempted to streamline the process but failed to resolve critical kinetic issues, often resulting in incomplete reactions and substantial material loss known as 'wash-away' tendencies. In these traditional single-acid environments, typically utilizing concentrated hydrochloric acid alone, the intermediate species exhibits poor solubility. This physical limitation leads to heterogeneous reaction conditions where localized hotspots can trigger sudden, exothermic decarboxylation events. Consequently, manufacturers faced yields stagnating around 70%, coupled with purification challenges that drove up the cost of goods sold and complicated waste management protocols.
The Novel Approach
The patented methodology overcomes these thermodynamic and kinetic barriers through the implementation of a binary acid system during the critical hydrolysis and salt formation stage. Instead of relying solely on mineral acids, the process employs a tailored mixture of organic acids (e.g., acetic, formic) and inorganic acids (e.g., HCl, H2SO4) in specific volumetric ratios. This dual-acid strategy dramatically improves the solubility profile of the precursor compound, ensuring a homogeneous reaction phase that proceeds smoothly without violent thermal spikes. The result is a controlled transformation that preserves the structural integrity of the sensitive quinolone ring while efficiently cleaving the protecting groups. By optimizing the reaction environment, this approach eliminates the 'rushing material' phenomenon observed in older techniques, thereby securing a total recovery rate that significantly exceeds previous benchmarks. This refinement transforms a previously erratic chemical step into a reliable, high-yielding unit operation suitable for continuous manufacturing.
Mechanistic Insights into Mixed-Acid Catalyzed Hydrolysis
The core mechanistic advantage of this invention lies in the modulation of proton activity and solvation power within the reaction matrix. In a standard hydrochloric acid medium, the zwitterionic nature of the amino acid intermediate often leads to precipitation before full conversion is achieved, effectively halting the reaction progress. The introduction of an organic co-acid acts as a co-solvent and a buffering agent, maintaining the intermediate in solution long enough for the hydrolysis of the ester and amide functionalities to reach completion. This solubility enhancement is critical because it prevents the formation of insoluble aggregates that trap unreacted starting materials. Furthermore, the organic acid component helps to moderate the acidity strength locally, preventing the aggressive protonation that can lead to ring degradation or unwanted side reactions on the quinolone scaffold. This delicate balance ensures that the decarboxylation step, if it occurs, happens in a controlled manner rather than as a sudden, yield-destroying event.
From an impurity control perspective, the mixed-acid system offers a cleaner reaction profile by suppressing the formation of polymeric byproducts and degradation species. Traditional high-temperature acid hydrolysis often generates colored impurities and tars that are difficult to remove via crystallization, necessitating expensive chromatographic purification. The milder yet effective conditions provided by the organic-inorganic acid blend minimize thermal stress on the molecule, resulting in a crude product with inherently higher purity. This reduces the burden on downstream processing units, allowing for simpler recrystallization protocols to achieve pharmaceutical-grade specifications. For R&D directors, this implies a more robust design space where minor fluctuations in temperature or stoichiometry do not catastrophically impact the final impurity profile, ensuring consistent batch-to-batch quality essential for regulatory filings.
How to Synthesize 2-Amino-3-(2(1H)-quinolone-4-yl) Propionic Acid Salt Efficiently
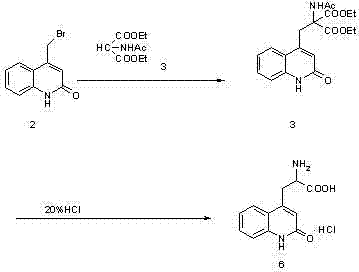
The synthesis protocol outlined in the patent provides a clear, two-stage pathway that transitions seamlessly from condensation to final salt formation. The initial step involves the coupling of a bromo-quinolone derivative with a malonate precursor in a fatty alcohol solvent, facilitated by a strong base like sodium ethylate. Following this condensation, the reaction mixture is carefully neutralized and cooled to precipitate the protected intermediate, which is then isolated via filtration. The subsequent and most critical stage involves subjecting this isolated intermediate to the novel mixed-acid hydrolysis conditions. Detailed standardized operating procedures, including specific temperature ramps, stirring rates, and acid addition profiles required to replicate this high-yield process, are provided in the technical guide below.
- Condense 4-bromoethyl-1H-2-quinolinone with acetamido diethyl malonate in fatty alcohol using sodium ethylate, then adjust pH and filter.
- React the resulting intermediate with a mixture of organic acid (e.g., acetic acid) and inorganic acid (e.g., hydrochloric acid) at elevated temperatures.
- Cool the reaction mixture to crystallize the final salt product, followed by filtration and drying to achieve high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this mixed-acid synthesis route translates directly into enhanced operational efficiency and reduced supply risk. The elimination of tedious multi-step hydrolysis sequences simplifies the manufacturing workflow, reducing the number of unit operations and the associated labor and utility costs. By moving away from processes prone to low yields and material loss, manufacturers can maximize the output from every kilogram of raw material input, effectively lowering the variable cost per unit of the active pharmaceutical ingredient. This efficiency gain is particularly valuable in the context of volatile raw material markets, where maximizing atom economy is a primary driver of profitability. Furthermore, the use of common, commodity-grade acids and solvents ensures that the supply chain remains resilient against shortages of exotic reagents.
- Cost Reduction in Manufacturing: The transition to a mixed-acid system eliminates the need for complex purification steps often required to remove stubborn impurities generated by harsh single-acid hydrolysis. By achieving higher crude purity directly from the reactor, the reliance on expensive chromatographic media or multiple recrystallization cycles is drastically reduced. This streamlining of the downstream process significantly lowers the consumption of solvents and energy, contributing to substantial cost savings in the overall production budget. Additionally, the improved yield means less waste disposal cost, further enhancing the economic viability of the process.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures consistent production schedules, as the reaction is less susceptible to failure due to solubility issues or thermal runaways. This reliability allows for more accurate forecasting and inventory planning, reducing the need for safety stock buffers that tie up working capital. The use of widely available reagents like acetic acid and hydrochloric acid minimizes the risk of supply disruptions associated with specialized catalysts or reagents. Consequently, partners can rely on a steady flow of high-quality intermediates to support their own API manufacturing timelines without unexpected delays.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind, utilizing standard reactor configurations and avoiding extreme pressure or temperature conditions that require specialized equipment. The simplified workup procedure, which relies on straightforward crystallization and filtration, facilitates easy scale-up from pilot plant to commercial tonnage production. Moreover, the reduction in solvent usage and waste generation aligns with increasingly stringent environmental regulations, helping manufacturers maintain compliance while minimizing their ecological footprint. This green chemistry aspect is becoming a key differentiator for suppliers seeking to partner with sustainability-focused pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Rebamipide intermediates using this advanced methodology. These insights are derived directly from the comparative data and experimental examples provided in the patent literature, highlighting the practical benefits of the mixed-acid approach. Understanding these nuances is essential for technical teams evaluating potential technology transfers or sourcing strategies for this critical building block.
Q: Why is a mixed acid system preferred over pure hydrochloric acid for this synthesis?
A: Pure hydrochloric acid often leads to poor solubility of the intermediate, causing sudden decarboxylation and low yields. A mixed acid system improves solubility and controls reaction severity.
Q: What represents the primary impurity challenge in traditional Rebamipide intermediate synthesis?
A: Traditional methods often suffer from incomplete reactions and 'wash-away' tendencies where materials are lost during processing, leading to lower overall mass balance and purity issues.
Q: Can this process be scaled for industrial production of API intermediates?
A: Yes, the method eliminates tedious multi-step hydrolysis and uses common solvents and acids, making it highly suitable for large-scale commercial manufacturing with consistent quality.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rebamipide Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the quality of the final API is inextricably linked to the precision of its precursor synthesis. Our technical team has extensively analyzed the mixed-acid hydrolysis technology described in CN102153512B and possesses the expertise to implement this route with rigorous process control. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a GMP-compliant manufacturing environment. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 2-amino-3-(2(1H)-quinolone-4-yl) propionic acid salt meets the exacting standards required for global pharmaceutical registration.
We invite procurement directors and R&D leaders to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging our optimized manufacturing capabilities, we can help you secure a stable supply of high-purity intermediates while reducing your overall cost of goods. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to chemical excellence can support your long-term strategic goals.
