Advanced Synthesis of P,O-Hybrid Ligands: A Safer Path to High-Purity Catalytic Intermediates
Advanced Synthesis of P,O-Hybrid Ligands: A Safer Path to High-Purity Catalytic Intermediates
The landscape of homogeneous catalysis is undergoing a significant transformation driven by the demand for safer, more efficient ligand synthesis protocols. Patent CN116178428A introduces a groundbreaking methodology for the preparation of P,O-hybrid ligands, a critical class of compounds widely utilized in transition metal-catalyzed reactions such as carbonylation and cross-coupling. Unlike conventional approaches that depend heavily on hazardous organometallic reagents, this invention leverages a robust two-step sequence involving nucleophilic substitution followed by metal salt-catalyzed coupling. For R&D directors and procurement specialists in the fine chemical sector, this represents a pivotal shift towards processes that offer enhanced safety profiles without compromising on the structural integrity or purity required for high-performance catalytic applications. The ability to synthesize complex bidentate ligands under mild conditions addresses long-standing bottlenecks in the supply chain of specialized pharmaceutical and agrochemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of phosphorus-containing ligands has been plagued by severe operational hazards and stringent environmental controls. Traditional pathways typically necessitate the use of Grignard reagents or organolithium compounds, which are notoriously pyrophoric and extremely sensitive to moisture and oxygen. These reagents require reaction environments that are absolutely dry and anaerobic, often demanding cryogenic conditions ranging from 0°C down to -78°C to control exothermic reactions and prevent decomposition. Such苛刻 conditions not only escalate capital expenditure for specialized equipment like Schlenk lines and low-temperature chillers but also introduce significant safety risks, including potential fires and explosions during reagent handling. Furthermore, the quenching and disposal of residual metal reagents post-reaction generate substantial hazardous waste, complicating downstream processing and increasing the overall environmental footprint of the manufacturing process. These factors collectively limit the scalability and economic viability of producing P,O-hybrid ligands on a commercial tonnage scale.
The Novel Approach
The methodology disclosed in CN116178428A circumvents these critical vulnerabilities by employing a transition metal-catalyzed coupling strategy that operates under significantly milder conditions. Instead of generating reactive organometallic species in situ, the process utilizes stable dialkylphosphine hydrogens as the phosphorus source, reacting them with halogenated ether intermediates in the presence of catalytic amounts of metal salts such as PdCl2 or Pd(OAc)2. This innovation allows the reaction to proceed at temperatures ranging from room temperature up to 150°C, completely eliminating the need for cryogenic cooling. The use of common organic solvents like DMSO or DMF, combined with standard alkaline compounds, simplifies the reaction setup to basic round-bottom or three-necked flasks under nitrogen protection. This shift not only drastically reduces the risk of thermal runaway incidents but also streamlines the workup procedure, enabling easier isolation of the target ligands through standard column chromatography. By removing the dependency on high-risk reagents, this approach opens the door for safer, more cost-effective manufacturing of high-value catalytic ligands.
Mechanistic Insights into Metal Salt-Catalyzed Coupling
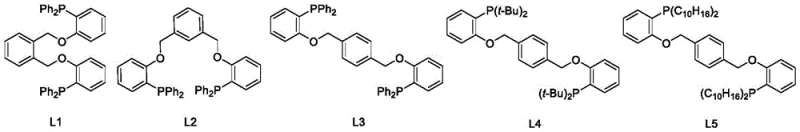
The core of this synthetic breakthrough lies in the efficient formation of the Carbon-Phosphorus (C-P) bond via a transition metal-catalyzed mechanism. In the second step of the synthesis, the di-[(halogenated phenoxy) methyl] benzene intermediate undergoes a coupling reaction with dialkylphosphine hydrogen. The metal salt catalyst, such as Palladium(II) chloride, likely facilitates an oxidative addition into the carbon-halogen bond of the substrate, forming a transient organopalladium species. Subsequent coordination and insertion of the phosphine hydrogen, followed by reductive elimination, forge the stable C-P bond while regenerating the active catalyst species. This catalytic cycle is highly advantageous because it avoids the stoichiometric consumption of expensive and hazardous metal reagents, relying instead on turnover numbers that maximize atom economy. The presence of the oxygen atom in the P,O-hybrid structure further stabilizes the metal center through hemilabile coordination, a feature that is preserved and optimized through this gentle synthetic route, ensuring the final ligand retains its unique electronic properties essential for catalytic activity.
Impurity control is another critical aspect where this mechanistic pathway excels over traditional lithiation methods. In Grignard-based syntheses, side reactions such as Wurtz coupling or protonation by trace moisture often lead to complex impurity profiles that are difficult to separate. In contrast, the nucleophilic substitution in the first step and the catalytic coupling in the second step are highly selective. The use of specific metal salts and controlled temperatures (e.g., 80°C to 130°C depending on the substrate) minimizes thermal degradation and non-specific side reactions. For instance, the patent reports yields as high as 82% for ligand L2 and 76% for L1, indicating a clean reaction profile with minimal byproduct formation. This high selectivity translates directly to reduced purification burdens, allowing manufacturers to achieve high-purity specifications required for sensitive pharmaceutical applications with fewer processing steps and lower solvent consumption.
How to Synthesize P,O-Hybrid Ligands Efficiently
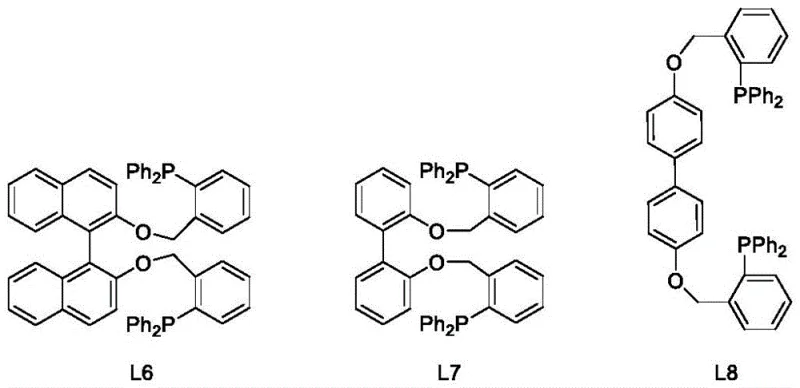
The synthesis protocol outlined in the patent provides a clear, reproducible roadmap for producing a variety of P,O-hybrid ligands, from simple phenyl derivatives to complex binaphthyl structures. The process begins with the preparation of the ether backbone via nucleophilic substitution, followed by the crucial phosphination step. This modular approach allows for the easy variation of substituents (R groups) on the phosphorus atom, such as phenyl, tert-butyl, or adamantyl groups, enabling the fine-tuning of steric and electronic properties for specific catalytic needs. The detailed standardized synthesis steps below outline the precise conditions, reagent ratios, and workup procedures necessary to replicate these high-yield results in a laboratory or pilot plant setting.
- Perform nucleophilic substitution between halogenated phenols and di(halomethyl)benzenes under alkaline conditions at elevated temperatures (up to 150°C) to form ether intermediates.
- Conduct a coupling reaction between the ether intermediate and dialkylphosphine hydrogen using a metal salt catalyst (e.g., PdCl2) and base in organic solvent.
- Purify the final P,O-hybrid ligand product via column chromatography to achieve high purity specifications suitable for catalytic applications.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers profound strategic advantages beyond mere technical feasibility. The elimination of pyrophoric reagents fundamentally alters the risk profile of the manufacturing facility, leading to substantial reductions in insurance premiums and safety compliance costs. Moreover, the ability to run reactions at ambient or moderately elevated temperatures removes the energy burden associated with maintaining cryogenic conditions, resulting in significant operational expenditure savings. The robustness of the process ensures consistent batch-to-batch quality, reducing the incidence of failed runs and material waste. This reliability is paramount for maintaining continuous supply chains for downstream API manufacturers who depend on timely delivery of high-quality intermediates. By simplifying the synthesis, companies can also reduce their dependency on specialized contract manufacturing organizations capable of handling hazardous chemistry, thereby bringing more production in-house or diversifying their supplier base with greater ease.
- Cost Reduction in Manufacturing: The removal of expensive and hazardous organolithium or Grignard reagents eliminates a major cost driver in raw material procurement. Additionally, the simplified workup and purification processes reduce solvent usage and waste disposal fees. The higher yields reported (up to 82%) mean less starting material is required per kilogram of finished product, directly improving the cost of goods sold (COGS) and enhancing overall margin potential for high-value ligand suppliers.
- Enhanced Supply Chain Reliability: Traditional methods are often prone to delays due to the strict handling requirements of sensitive reagents and the need for specialized equipment maintenance. This new method utilizes stable, commercially available starting materials and standard reactor setups, minimizing downtime and equipment failure risks. The milder conditions allow for faster turnaround times between batches, enabling suppliers to respond more agilely to fluctuating market demands and urgent orders from pharmaceutical clients without compromising safety or quality standards.
- Scalability and Environmental Compliance: Scaling hazardous Grignard reactions from gram to ton scale presents immense engineering challenges and regulatory hurdles. In contrast, this catalytic protocol is inherently scalable, as it avoids exothermic spikes and toxic byproducts associated with metal reagent quenching. The use of common solvents and the generation of less hazardous waste streams align with green chemistry principles, facilitating easier regulatory approval and permitting for expanded production capacity. This makes the technology ideal for long-term commercial scale-up of complex polymer additives or pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this P,O-hybrid ligand synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity on how this method outperforms legacy techniques in terms of safety, yield, and operational simplicity. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Why is this new synthesis method safer than traditional Grignard routes?
A: Traditional methods rely on pyrophoric Grignard or organolithium reagents requiring strict anhydrous/anaerobic conditions. This patent utilizes mild nucleophilic substitution and metal-salt catalysis, eliminating explosion risks and simplifying equipment requirements.
Q: What yields can be expected for P,O-hybrid ligands using this protocol?
A: The patent demonstrates significant yield improvements, with specific examples achieving up to 82% yield for ligands like L2, compared to often lower and inconsistent yields associated with hazardous organometallic pathways.
Q: Is this process scalable for industrial production of catalyst ligands?
A: Yes, the avoidance of cryogenic conditions (-78°C) and sensitive reagents allows for operation in standard reactors at temperatures up to 150°C, significantly enhancing commercial scale-up feasibility and supply chain reliability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable P,O-Hybrid Ligand Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced ligand technologies play in driving innovation in catalysis and drug discovery. Our team of expert chemists has thoroughly analyzed the potential of the synthetic route described in CN116178428A and is fully equipped to translate this laboratory-scale success into robust commercial production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. Our state-of-the-art facilities are designed to handle complex organic syntheses with stringent purity specifications, supported by rigorous QC labs that utilize advanced analytical techniques to verify the identity and quality of every batch of P,O-hybrid ligands we produce.
We invite you to collaborate with us to leverage this safer, more efficient synthesis technology for your specific catalytic applications. Whether you require custom synthesis of novel ligand derivatives or reliable supply of established intermediates, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis tailored to your project volume. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can optimize your supply chain and accelerate your time to market.
