Advanced Tetrapeptide Catalysis for High-Purity Pantolactone Intermediates and Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking more efficient, sustainable, and stereoselective methods for synthesizing complex chiral building blocks. A significant breakthrough in this domain is detailed in patent CN111848320A, which discloses a novel asymmetric Aldol reaction catalyzed by tetrapeptides (TP) or their enantiomers (ent-TP). This technology enables the precise synthesis of chiral 2-hydroxy-1,4-dicarbonyl compounds, which serve as critical precursors for the production of optically active pantolactone, a key intermediate in Vitamin B5 (pantothenic acid) manufacturing. Unlike traditional enzymatic or small-molecule approaches, this tetrapeptide-catalyzed method offers exceptional control over stereochemistry, allowing for the selective formation of either (R) or (S) configurations with high fidelity. The ability to access these high-value intermediates through a mild, organocatalytic pathway represents a substantial advancement for reliable pharmaceutical intermediate supplier networks aiming to reduce dependency on complex biocatalytic processes.
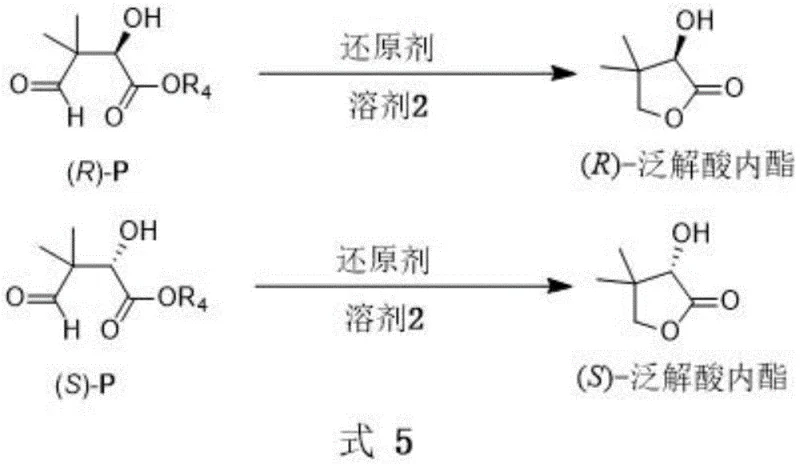
The strategic value of this invention lies in its direct applicability to the synthesis of pantolactone, a molecule of immense commercial importance. As illustrated in the reaction scheme, the chiral 2-hydroxy-1,4-dicarbonyl compounds generated via this Aldol reaction can be subsequently reduced to form pantolactone with retained optical purity. This two-step sequence—from simple aliphatic aldehydes and glyoxylates to the final lactone—streamlines the supply chain for vitamin manufacturers. By establishing a robust chemical route that bypasses the stability issues associated with natural enzymes, this technology ensures consistent quality and supply continuity. For procurement managers, this translates to a more predictable sourcing strategy for high-purity pantolactone precursors, mitigating risks associated with biological variability and fermentation bottlenecks often seen in alternative production methods.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of optically active 2-hydroxy-3-methyl-3-formylbutyrates, the direct precursors to pantolactone, has relied heavily on amino acid-based organocatalysts such as proline, histidine, or isoleucine. However, these conventional methods suffer from significant limitations in terms of stereoselectivity and efficiency. For instance, proline-catalyzed reactions have historically yielded enantiomeric excess (ee) values no higher than 42%, which is insufficient for high-grade pharmaceutical applications without extensive and costly purification steps. Even optimized protocols using histidine or isoleucine have struggled to surpass 80% ee, creating a bottleneck in the production of high-optical-purity intermediates. Furthermore, while biocatalysis offers green credentials, the complexity of enzyme protein structures, their susceptibility to inactivation, and narrow substrate specificity often restrict their rapid application in diverse chemical syntheses. These factors collectively drive up the cost of goods and complicate the commercial scale-up of complex chiral intermediates.
The Novel Approach
In stark contrast, the tetrapeptide-catalyzed approach described in the patent introduces a paradigm shift by utilizing designed short peptides that mimic enzymatic active sites while retaining the stability of small organic molecules. This novel methodology facilitates the asymmetric Aldol reaction of aliphatic aldehydes with glyoxylates or acylformaldehyde monohydrates under remarkably mild conditions. The use of tetrapeptide catalysts allows for the synthesis of both (R) and (S) configurations of the target 2-hydroxy-1,4-dicarbonyl compounds simply by selecting the appropriate catalyst enantiomer (TP or ent-TP). This flexibility is crucial for manufacturing diverse drug candidates that require specific stereochemical arrangements. Moreover, the reaction proceeds with high yields, potentially reaching up to 99%, and exceptional enantioselectivity, also achieving up to 99% ee. This dramatic improvement in performance metrics underscores the potential for significant cost reduction in API manufacturing by minimizing waste and maximizing the throughput of valuable chiral material.
Mechanistic Insights into Tetrapeptide-Catalyzed Asymmetric Aldol Reaction
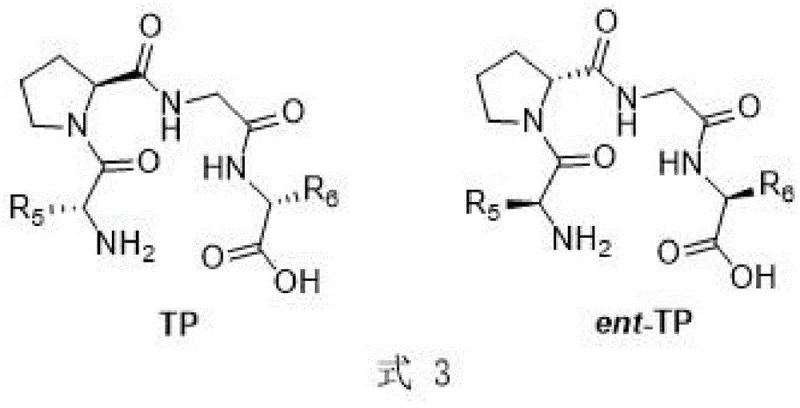
The efficacy of this synthetic route is rooted in the unique structural properties of the tetrapeptide catalysts. Unlike simple amino acids, these tetrapeptides possess a defined secondary structure that creates a chiral environment capable of distinguishing between prochiral faces of the substrate during the bond-forming event. The catalyst, generally represented by the structure in Formula 3, features specific amino acid residues (such as Valine, Leucine, Phenylalanine, etc.) that engage in hydrogen bonding and steric interactions with the aldehyde and glyoxylate substrates. This biomimetic design ensures that the nucleophilic attack occurs with high spatial precision, thereby locking in the desired stereochemistry. The robustness of the peptide backbone also contributes to the catalyst's stability under reaction conditions, preventing the degradation often observed with more fragile biological enzymes. This mechanistic stability is a key factor in ensuring reproducible results across different batches, a critical requirement for industrial adoption.
From an impurity control perspective, the high enantioselectivity of the tetrapeptide catalyst inherently limits the formation of unwanted stereoisomers. In traditional low-selectivity processes, the presence of the wrong enantiomer can act as a difficult-to-remove impurity that compromises the safety and efficacy of the final drug product. By achieving ee values approaching 99%, this process drastically simplifies the downstream purification workflow. The reduction in impurity load means fewer crystallization steps or chromatographic separations are required, which directly correlates to lower solvent consumption and reduced processing time. For R&D directors focused on process chemistry, this level of control offers a clear pathway to developing a robust, scalable manufacturing process that meets stringent regulatory standards for chiral purity without the need for expensive chiral resolution techniques post-synthesis.
How to Synthesize Chiral 2-Hydroxy-1,4-Dicarbonyl Compounds Efficiently
The practical implementation of this technology involves a straightforward protocol that is amenable to standard laboratory and pilot plant equipment. The process typically begins with the dissolution of the tetrapeptide catalyst in a suitable solvent, followed by the sequential addition of the aliphatic aldehyde and the electrophilic partner. Reaction monitoring is easily achieved via thin-layer chromatography (TLC), and the workup involves standard aqueous quenching and organic extraction. The simplicity of the operation, combined with the high performance of the catalyst, makes this an attractive option for process development teams looking to optimize their synthetic routes. For those interested in the specific operational parameters and stoichiometry, the detailed standardized synthesis steps are provided in the guide below.
- Prepare the reaction vessel by dissolving the specific tetrapeptide catalyst (TP or ent-TP) in a suitable organic solvent such as acetonitrile or dichloromethane under controlled temperature conditions.
- Sequentially add the aliphatic aldehyde and the electrophile (glyoxylate or acylformaldehyde monohydrate) to the catalyst solution while maintaining stirring to ensure homogeneous mixing and optimal catalytic activity.
- Monitor reaction progress via TLC, quench with saturated ammonium chloride, extract with ethyl acetate, and purify the resulting chiral 2-hydroxy-1,4-dicarbonyl compound using column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this tetrapeptide-catalyzed technology offers compelling economic and logistical benefits. The shift from low-efficiency amino acid catalysts or unstable enzymes to robust tetrapeptides fundamentally alters the cost structure of producing chiral intermediates. By eliminating the need for complex fermentation infrastructure or expensive metal-ligand systems, manufacturers can achieve substantial cost savings in raw materials and capital expenditure. The high yields and selectivity mean that less starting material is wasted, improving the overall atom economy of the process. Furthermore, the use of common, commercially available solvents like acetonitrile and dichloromethane ensures that the supply chain remains resilient and不受制于 specialized reagent shortages. This reliability is essential for maintaining continuous production schedules in a volatile global market.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and the avoidance of complex enzymatic fermentation processes lead to a drastically simplified production workflow. This simplification removes the need for expensive heavy metal removal steps and reduces the energy consumption associated with maintaining strict biological conditions. Consequently, the overall cost of goods sold (COGS) for the chiral intermediate is significantly lowered, providing a competitive pricing advantage for the final pharmaceutical product. The high catalytic efficiency also means that lower catalyst loadings may be sufficient, further driving down material costs.
- Enhanced Supply Chain Reliability: The starting materials for this reaction, such as aliphatic aldehydes and glyoxylates, are commodity chemicals with well-established global supply chains. Unlike specialized chiral pool materials that may have limited suppliers, these feedstocks are readily available from multiple sources, reducing the risk of supply disruption. Additionally, the stability of the tetrapeptide catalysts allows for easier storage and transportation compared to sensitive enzymes, ensuring that the critical catalytic component is always available when needed for production runs.
- Scalability and Environmental Compliance: The mild reaction conditions, often proceeding at room temperature or with simple cooling, facilitate easy scale-up from gram to ton scale without requiring exotic high-pressure or high-temperature equipment. This scalability is paired with improved environmental compliance, as the organocatalytic nature of the reaction avoids the generation of heavy metal waste streams. The reduced need for extensive purification also lowers the volume of solvent waste generated, aligning with modern green chemistry principles and reducing the burden on waste treatment facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this tetrapeptide-catalyzed synthesis. These insights are derived directly from the patent data and are intended to clarify the operational feasibility and strategic value of this technology for potential partners. Understanding these details is crucial for making informed decisions about integrating this route into existing manufacturing portfolios.
Q: What are the advantages of using tetrapeptide catalysts over traditional proline derivatives?
A: Tetrapeptide catalysts offer significantly higher enantioselectivity, achieving up to 99% ee compared to the limited 42-80% ee typically observed with proline or histidine derivatives. Additionally, they provide access to both R and S configurations simply by switching between the TP and ent-TP enantiomers.
Q: Can this method be used for the commercial production of pantolactone?
A: Yes, the synthetic products (chiral 2-hydroxy-1,4-dicarbonyl compounds) serve as direct precursors for pantolactone. The mild reaction conditions and high yields make this route highly suitable for scaling up in pharmaceutical manufacturing environments.
Q: What types of solvents are compatible with this asymmetric Aldol reaction?
A: The process demonstrates robust compatibility with a wide range of common organic solvents including acetonitrile, dichloromethane, chloroform, and ethers. Acetonitrile is often preferred for achieving optimal reaction rates and stereoselectivity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral 2-Hydroxy-1,4-Dicarbonyl Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced organocatalytic technologies like the tetrapeptide-mediated Aldol reaction for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are successfully translated into robust industrial processes. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs, guaranteeing that every batch of chiral 2-hydroxy-1,4-dicarbonyl compound or pantolactone precursor meets the highest international standards. We are equipped to handle the nuances of peptide-catalyzed reactions, optimizing conditions to maximize yield and enantioselectivity for your specific project needs.
We invite you to collaborate with us to leverage this cutting-edge synthesis method for your supply chain. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing you to evaluate how this efficient, high-purity manufacturing route can enhance your product portfolio and reduce time-to-market for your critical pharmaceutical ingredients.
