Advanced Pd-Catalyzed Synthesis of Diarylsultams: A Breakthrough for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to construct complex heterocyclic scaffolds, particularly those found in bioactive molecules. Patent CN108610304B, published in late 2021, introduces a transformative synthetic methodology for generating diarylsultam compounds, a privileged structural motif prevalent in numerous anti-inflammatory agents and biologically active natural products. This innovation leverages transition metal palladium catalysis to facilitate a direct ortho-C-H bond functionalization cyclization between N-methoxybenzenesulfonamide derivatives and aryne precursors. By bypassing the need for pre-functionalized halogenated starting materials, this technology addresses critical pain points in modern process chemistry, offering a streamlined route that aligns perfectly with the principles of green chemistry and atom economy. For R&D directors and procurement strategists, understanding the nuances of this patent is essential for evaluating next-generation supply chains for high-value pharmaceutical intermediates.
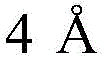
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the dibenzosultam backbone has relied heavily on intramolecular cyclization strategies that demand rigorous pre-functionalization of the substrate. Traditional literature describes methods involving intramolecular free radical cyclization or palladium-catalyzed intramolecular arylation of 2-halo-N-alkyl-N-aryl benzene sulfonamides. These legacy approaches suffer from inherent inefficiencies, primarily due to the necessity of introducing halogen atoms onto the aromatic ring prior to the cyclization event. This pre-functionalization step not only adds extra synthetic operations, thereby increasing the overall step count and reducing cumulative yield, but it also generates stoichiometric amounts of halogenated waste. Furthermore, many of these conventional routes require harsh reaction conditions, such as strong oxidants or high-energy radical initiators, which can compromise the integrity of sensitive functional groups elsewhere in the molecule. The reliance on specific halogenated substrates also severely restricts the scope of accessible chemical space, limiting the ability of medicinal chemists to rapidly explore structure-activity relationships (SAR) around the sultam core.
The Novel Approach
In stark contrast, the methodology disclosed in CN108610304B represents a paradigm shift by utilizing a direct C-H activation strategy. This novel approach employs a palladium catalyst to activate the ortho-C-H bond adjacent to the sulfonamide directing group, allowing for direct coupling with transient aryne intermediates generated in situ from triflate precursors. This eliminates the need for pre-installed halogen handles, effectively shortening the synthetic sequence and improving the overall mass balance of the process. The reaction operates under relatively mild thermal conditions in a mixed solvent system of dimethyl sulfoxide and dioxane, utilizing cesium fluoride as a key promoter for aryne generation. This transition from a step-wise, waste-generating protocol to a convergent, atom-economical cyclization significantly enhances the feasibility of producing complex diarylsultams. For process development teams, this means a drastic reduction in raw material costs and a simplified workflow that is far more amenable to scale-up.
Mechanistic Insights into Pd-Catalyzed C-H Activation Cyclization
The core of this technological breakthrough lies in the sophisticated interplay between the palladium catalyst and the unique reactivity of the N-methoxybenzenesulfonamide directing group. The mechanism initiates with the coordination of the palladium species to the nitrogen or oxygen atoms of the sulfonamide moiety, which directs the metal center to the proximal ortho-C-H bond. Through a concerted metalation-deprotonation (CMD) pathway facilitated by the base (sodium pivalate), a stable five-membered palladacycle intermediate is formed. This organometallic species is the pivotal junction where the C-H bond is effectively converted into a reactive C-Pd bond. Subsequently, the highly reactive aryne intermediate, generated from the triflate precursor via fluoride-induced elimination, inserts into this C-Pd bond. This insertion step is critical as it constructs the new carbon-carbon bond that links the two aromatic systems. Finally, a reductive elimination or protonolysis step releases the final diarylsultam product and regenerates the active palladium catalyst, closing the catalytic cycle. Understanding this mechanism is vital for R&D teams aiming to optimize reaction parameters or adapt the chemistry to novel substrates.
From an impurity control perspective, this mechanism offers distinct advantages over radical-based alternatives. Radical cyclizations often suffer from poor regioselectivity and the formation of polymeric byproducts due to the indiscriminate nature of radical species. In contrast, the palladium-catalyzed C-H activation described here is highly regioselective, driven by the chelation of the sulfonamide group. This intrinsic selectivity ensures that the cyclization occurs exclusively at the ortho-position, minimizing the formation of isomeric impurities that are notoriously difficult to separate. Additionally, the use of molecular sieves in the reaction mixture helps to sequester water, preventing the hydrolysis of the sensitive aryne intermediate or the catalyst, thereby further enhancing the purity profile of the crude product. This high level of control over the reaction trajectory translates directly into reduced downstream purification burdens, a key metric for commercial viability.
How to Synthesize 6H-dibenzo[c,e][1,2]thiazine-5,5-dioxide Efficiently
The practical implementation of this synthesis is straightforward and relies on readily available reagents, making it an attractive option for both laboratory discovery and pilot plant operations. The standard protocol involves charging a pressure-resistant reaction vessel with the N-methoxybenzenesulfonamide substrate, a catalytic amount of palladium acetate, and stoichiometric quantities of copper acetate and cesium fluoride. The reaction is conducted in a sealed system under an inert atmosphere to prevent oxidation of the catalyst or moisture sensitivity issues. Detailed standardized operating procedures for scaling this reaction from gram to kilogram quantities are provided in the technical guide below, ensuring reproducibility and safety.
- Prepare the reaction mixture by combining N-methoxybenzenesulfonamide, palladium acetate catalyst, anhydrous copper acetate, sodium pivalate hydrate, and molecular sieves in a pressure-resistant tube.
- Add the solvent system consisting of dioxane and dimethyl sulfoxide, followed by cesium fluoride and the phenylalkyne precursor under inert gas protection.
- Heat the sealed reaction system to 110°C for 24 hours, then cool, extract with organic solvents, and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers compelling economic and logistical benefits that extend beyond simple yield improvements. The shift away from pre-halogenated starting materials removes a significant cost driver from the bill of materials, as halogenated aromatics are often more expensive and subject to greater supply volatility than their non-halogenated counterparts. Furthermore, the high atom utilization rate of this C-H functionalization method means that a greater proportion of the input mass ends up in the final product, drastically reducing the volume of waste solvent and salts that require disposal. This reduction in waste handling costs contributes significantly to the overall cost reduction in pharmaceutical intermediate manufacturing, aligning with corporate sustainability goals.
- Cost Reduction in Manufacturing: The elimination of pre-functionalization steps inherently lowers the cost of goods sold (COGS) by reducing the number of unit operations required. By avoiding the use of stoichiometric amounts of expensive halogenating reagents and the subsequent purification of halogenated intermediates, the process achieves substantial cost savings. Additionally, the catalytic nature of the palladium system, combined with the use of inexpensive inorganic bases like cesium fluoride and sodium pivalate, ensures that reagent costs remain low even at large scales. The simplified workup procedure, which typically involves standard extraction and chromatography, further reduces labor and utility costs associated with complex isolation protocols.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as benzenesulfonamides and aryl triflates enhances supply chain resilience. Unlike specialized halogenated building blocks which may have limited suppliers, the starting materials for this process are widely produced and available from multiple global sources. This diversification of the supply base mitigates the risk of shortages and price spikes, ensuring a steady flow of materials for continuous production. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, further stabilizing the supply chain against upstream fluctuations.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing solvents and conditions that are compatible with standard stainless steel reactor infrastructure. The absence of hazardous radical initiators or explosive diazonium intermediates simplifies safety assessments and regulatory approvals for commercial plants. Moreover, the high atom economy and reduced waste generation facilitate compliance with increasingly stringent environmental regulations regarding solvent emissions and hazardous waste disposal. This environmental compatibility not only avoids potential fines but also enhances the company's reputation as a responsible manufacturer of specialty chemicals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis technology. These insights are derived directly from the experimental data and scope analysis presented in the patent documentation, providing a clear picture of the method's capabilities and limitations for potential adopters.
Q: What are the primary advantages of this Pd-catalyzed method over traditional radical cyclization?
A: Unlike traditional methods requiring pre-functionalized halogenated substrates which generate stoichiometric salt waste, this method utilizes direct C-H bond functionalization. This significantly improves atom economy and simplifies the purification process by eliminating the need for harsh radical initiators or pre-halogenation steps.
Q: What is the substrate scope for the aryne precursor in this synthesis?
A: The method demonstrates wide applicability, successfully accommodating various substituted aryl triflates including those with electron-donating groups like methyl and methoxy, as well as electron-withdrawing groups such as fluoro, chloro, bromo, cyano, and nitro. It also extends to fused ring systems like naphthalene derivatives.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process uses commercially available raw materials and standard reaction conditions (110°C in DMSO/dioxane). The absence of expensive transition metals beyond catalytic amounts of palladium and the use of robust inorganic bases make it highly scalable for commercial manufacturing of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diarylsultam Supplier
The synthesis of diarylsultams via palladium-catalyzed C-H activation represents a significant advancement in the field of heterocyclic chemistry, offering a robust platform for the production of high-purity pharmaceutical intermediates. NINGBO INNO PHARMCHEM stands at the forefront of translating such innovative academic and patent technologies into commercial reality. As a seasoned CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a manufacturing setting. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of diarylsultam delivered meets the exacting standards required for drug substance manufacturing.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this cutting-edge synthetic route for their pipeline projects. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise in complex organic synthesis can accelerate your development timelines and optimize your supply chain economics.
