Advanced Three-Step Synthesis of High-Purity cis-2-Aminocyclohexanol for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to access chiral building blocks, particularly beta-amino alcohols which serve as critical scaffolds in drug discovery. A recent technological breakthrough documented in patent CN114315609A introduces a robust and scalable process for the preparation of cis-2-aminocyclohexanol, a valuable intermediate often challenging to synthesize with high stereochemical fidelity. This innovative method departs from traditional multi-step sequences by utilizing cyclohexene as a readily available starting material, reacting it with common reagents such as N-halosuccinimides (NXS) or N-haloacetamides under catalytic conditions. The process streamlines the synthesis into three distinct operational stages: a cyclization reaction to form a key oxazoline intermediate, a subsequent ring-opening reaction, and a final salification and resolution step. By integrating these steps, the technology addresses long-standing issues regarding process complexity and purification difficulties, offering a compelling solution for manufacturers aiming to optimize their supply chains for high-purity chiral intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of cis-2-aminocyclohexanol has been fraught with operational inefficiencies that hinder large-scale production. Prior art, such as the methodology described in J. Org. Chem. (2006), relies on a cumbersome route starting from trans-2-benzylaminocyclohexanol. This conventional approach necessitates a nitrogen protection step using benzoyl chloride, followed by cyclization with excessive thionyl chloride to generate an oxazoline. Crucially, the subsequent hydrolysis step requires refluxing in 6M hydrochloric acid, which generates a mixture of cis and trans isomers in an 8:1 ratio. This lack of selectivity forces the reliance on column chromatography to separate the desired cis-isomer from the trans-byproduct, a technique that is notoriously difficult to translate from the laboratory to industrial manufacturing due to solvent consumption and throughput limitations. Furthermore, the final step involves palladium-carbon catalyzed hydrogenation for debenzylation, introducing expensive precious metal catalysts and safety concerns associated with high-pressure hydrogen gas, thereby inflating both capital expenditure and operational costs.
The Novel Approach
In stark contrast, the novel process disclosed in the patent data revolutionizes the synthetic landscape by establishing a direct route from cyclohexene that inherently favors the cis-configuration without generating difficult-to-separate trans-isomers. The strategy employs a cyclization reaction where cyclohexene reacts with NXS or CH3CONHX in the presence of specific catalysts like B(C6F5)3 or dimethyl sulfide, followed by ring closure with DBU to form a fused oxazoline intermediate. This intermediate structurally locks the relative stereochemistry, effectively bypassing the need for isomer separation via chromatography. The subsequent ring-opening is achieved under mild Lewis acid catalysis in an alcohol solvent, avoiding the harsh acidic conditions and gas evolution issues of the prior art. Finally, the process utilizes a classical resolution strategy with L-DBTA to secure enantiomeric purity, allowing for the recycling of the resolving agent. This streamlined approach not only shortens the reaction route but also replaces hazardous reagents and complex purification methods with standard unit operations suitable for ton-scale production.
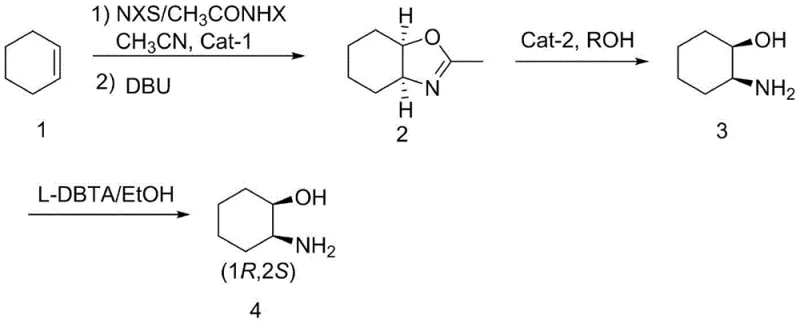
Mechanistic Insights into Oxazoline-Mediated Cyclization and Resolution
The core of this technological advancement lies in the stereoselective formation of the oxazoline ring system, which serves as a temporary scaffold to enforce the cis-relationship between the incoming functional groups. In the first step, the reaction of cyclohexene with N-halosuccinimide or N-haloacetamide in the presence of a Lewis acid catalyst initiates an electrophilic addition. The subsequent treatment with DBU (1,8-Diazabicyclo[5.4.0]undec-7-ene) promotes an intramolecular nucleophilic attack by the oxygen or nitrogen species onto the adjacent carbon, closing the ring to form the bicyclic oxazoline structure. This cyclization is highly stereospecific, ensuring that the substituents end up in a cis-configuration relative to the cyclohexane ring. The choice of catalyst, whether it be the strong Lewis acid tris(pentafluorophenyl)borane or the softer dimethyl sulfide, allows for fine-tuning of the reaction kinetics and yield, with reported yields reaching as high as 95.2% under optimized conditions. This high efficiency in the initial step sets the stage for a high-yielding overall process by minimizing material loss early in the synthesis.
Following the formation of the oxazoline intermediate, the second step involves a Lewis acid-catalyzed ring-opening reaction that liberates the free amine and alcohol functionalities. Unlike the harsh hydrolytic conditions of previous methods, this step utilizes alcohols like methanol or ethanol in the presence of catalysts such as BF3-Et2O or B(C6F5)3. The mechanism likely involves the coordination of the Lewis acid to the oxazoline nitrogen, increasing the electrophilicity of the ring carbon and facilitating nucleophilic attack by the solvent or trace water, ultimately leading to the ring-opened amino alcohol. The final stage employs chiral resolution using L-dibenzoyltartaric acid (L-DBTA), where the racemic amino alcohol forms a diastereomeric salt. Due to the differences in solubility between the diastereomers, the desired (1R,2S)-enantiomer can be isolated through crystallization. This resolution step is highly effective, achieving enantiomeric excess values of up to 98.3% ee, and the mother liquor containing the unwanted enantiomer and the resolving agent can be processed to recover and reuse the L-DBTA, further enhancing the atom economy and sustainability of the process.
How to Synthesize cis-2-Aminocyclohexanol Efficiently
The synthesis of this high-value chiral intermediate is structured around three critical operational phases that ensure both high yield and optical purity. The process begins with the activation of cyclohexene and subsequent cyclization to form the key oxazoline intermediate, followed by a controlled ring-opening to reveal the amino alcohol functionality. The final phase involves a precise resolution protocol to isolate the specific (1R,2S) enantiomer required for pharmaceutical applications. For detailed standard operating procedures, including specific molar ratios, temperature profiles, and workup instructions, please refer to the technical guide below.
- Cyclization Reaction: React cyclohexene with NXS or CH3CONHX in the presence of a catalyst (B(C6F5)3 or Me2S) followed by DBU treatment to form the oxazoline intermediate.
- Ring-Opening Reaction: Treat the oxazoline intermediate with an alcohol solvent and a Lewis acid catalyst (B(C6F5)3 or BF3-Et2O) to obtain the racemic amino alcohol.
- Chiral Resolution: Resolve the racemic mixture using L-DBTA in ethanol, followed by acid-base workup to isolate the pure (1R,2S)-enantiomer.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented process offers substantial strategic advantages by fundamentally altering the cost structure and risk profile of producing cis-2-aminocyclohexanol. The elimination of column chromatography is perhaps the most significant driver of cost reduction, as chromatographic separation is a major bottleneck in fine chemical manufacturing that consumes vast quantities of solvents and silica gel while limiting batch sizes. By replacing this with crystallization-based purification, the process enables true continuous or large-batch processing, drastically reducing solvent waste disposal costs and increasing throughput capacity. Furthermore, the removal of the palladium-catalyzed hydrogenation step eliminates the need for expensive precious metal catalysts and the associated specialized high-pressure equipment, leading to significant capital expenditure savings and reduced dependency on volatile metal markets. The use of commodity chemicals like cyclohexene and N-bromosuccinimide ensures a stable and diverse supply base, mitigating the risk of raw material shortages that often plague specialty syntheses reliant on exotic starting materials.
- Cost Reduction in Manufacturing: The process achieves cost efficiency primarily through the simplification of the synthetic route and the avoidance of expensive reagents. By removing the need for benzyl protection and subsequent hydrogenolysis, the manufacturer saves on the cost of benzylating agents and palladium catalysts, which are subject to significant price fluctuations. Additionally, the ability to recycle the chiral resolving agent, L-DBTA, means that the effective cost per kilogram of the resolving agent is significantly lowered over multiple batches. The high yields reported in the initial cyclization step, often exceeding 90%, minimize raw material waste, ensuring that a greater proportion of the input cyclohexene is converted into valuable product rather than byproducts. These factors combine to create a leaner manufacturing process with a lower variable cost per unit, making the final product more competitive in the global market.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the reliance on widely available industrial feedstocks. Cyclohexene is a bulk chemical produced in massive quantities for the nylon industry, ensuring a consistent and reliable supply stream that is less susceptible to the disruptions often seen with niche fine chemical intermediates. The reagents used, such as N-halosuccinimides and common Lewis acids, are standard inventory items for most chemical suppliers, reducing lead times for procurement. Moreover, the robustness of the reaction conditions, which tolerate a range of temperatures and do not require stringent anhydrous environments for all steps, reduces the risk of batch failures due to minor environmental variations. This operational robustness translates to more predictable production schedules and the ability to maintain safety stock levels without fear of rapid degradation or instability.
- Scalability and Environmental Compliance: The design of this process is inherently scalable, moving away from batch-limited techniques like chromatography to unit operations like filtration and distillation that are easily expanded. The avoidance of thionyl chloride in excess and the elimination of hydrogen gas usage significantly improve the safety profile of the plant, reducing the regulatory burden and insurance costs associated with handling hazardous materials. From an environmental standpoint, the reduction in solvent usage and the potential for solvent recovery loops align with green chemistry principles, helping manufacturers meet increasingly stringent environmental regulations. The generation of fewer byproducts and the ability to treat waste streams more easily due to the absence of heavy metals simplify wastewater treatment processes, ensuring compliance with local discharge standards and supporting corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of cis-2-aminocyclohexanol using this advanced methodology. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation to ensure accuracy and relevance for industry professionals. Understanding these details is crucial for evaluating the feasibility of integrating this intermediate into your specific drug substance or agrochemical manufacturing workflows.
Q: What are the key advantages of this new synthesis route over conventional methods?
A: The new process eliminates the need for column chromatography and expensive palladium-catalyzed hydrogenation steps required in prior art. It utilizes common raw materials like cyclohexene and N-halosuccinimides, resulting in a shorter reaction sequence and simplified purification via crystallization.
Q: How is stereochemical control achieved in this process?
A: Stereocontrol is initially established during the cyclization step which forms a cis-fused oxazoline ring system. Final enantiomeric purity is secured through a classical diastereomeric salt resolution using L-DBTA, yielding the target (1R,2S) isomer with high optical purity (e.g., 98.3% ee).
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is designed for scalability. It avoids hazardous reagents like thionyl chloride in excess and removes the bottleneck of column chromatography. The use of recyclable resolving agents and standard Lewis acid catalysts facilitates safe and efficient commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable cis-2-Aminocyclohexanol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality chiral intermediates play in the development of next-generation therapeutics. Our team of expert chemists has thoroughly analyzed this patented route and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this efficient synthesis to life. We are committed to delivering products that meet stringent purity specifications, utilizing our rigorous QC labs to verify enantiomeric excess and chemical purity at every stage of the manufacturing process. Our state-of-the-art facilities are equipped to handle the specific catalytic requirements of this synthesis, ensuring that the benefits of this novel process are fully realized in the final product delivered to your facility.
We invite you to collaborate with us to leverage this cost-effective and scalable technology for your supply chain. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our manufacturing capabilities can support your project timelines and budgetary goals effectively.
