Advanced Catalytic Synthesis of TMPMgCl·LiCl for Scalable Pharmaceutical Manufacturing
Advanced Catalytic Synthesis of TMPMgCl·LiCl for Scalable Pharmaceutical Manufacturing
The landscape of organometallic chemistry is constantly evolving to meet the rigorous demands of modern pharmaceutical synthesis, particularly in the realm of regioselective functionalization. Patent CN103435635A introduces a transformative preparation method for magnesium chloride (2,2,6,6-tetramethyl piperidine) lithium salt, commonly known as TMPMgCl·LiCl, which serves as a critical reagent for the metallation of aromatic hydrocarbons. This innovation addresses long-standing challenges associated with the stability and cost of traditional lithiation agents by employing a catalytic amount of 1,10-phenanthroline to facilitate a highly efficient one-pot synthesis. By utilizing inexpensive raw materials such as isopropyl magnesium chloride and anhydrous lithium chloride, this process not only enhances reaction yields but also simplifies the operational complexity typically required for generating such potent bases. For a reliable pharma intermediate supplier, understanding and implementing such catalytic advancements is essential for maintaining competitiveness in the global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the metallation of aromatic hydrocarbons has relied heavily on lithium alkylides or lithium amides, which are characterized by their extremely high reactivity and inherent instability. These traditional reagents often necessitate cryogenic reaction conditions, typically ranging from -70°C to -90°C, to mitigate uncontrollable side reactions and ensure regioselectivity, thereby imposing severe energy burdens and equipment constraints on manufacturing facilities. Furthermore, the tetrahydrofuran solutions of these lithium bases are notoriously unstable at room temperature, frequently requiring in-situ generation which complicates process control and limits the scope of applicable solvents primarily to THF or hexane mixtures. The reliance on pre-formed, commercially available i-PrMgCl·LiCl solutions also presents a significant economic bottleneck, as these specialized reagents are substantially more expensive than their individual components and often difficult to source in high concentrations. Consequently, the conventional pathways for generating TMP-bases are fraught with high operational costs, safety risks associated with cryogenic handling, and limited scalability for industrial applications.
The Novel Approach
The patented methodology offers a robust alternative by enabling the direct synthesis of TMPMgCl·LiCl from cheap, readily available starting materials through a catalytic enhancement strategy. By introducing 1,10-phenanthroline into the reaction mixture containing anhydrous lithium chloride, 2,2,6,6-tetramethyl piperidine, and isopropyl magnesium chloride, the process achieves a remarkable improvement in transformation efficiency without the need for expensive pre-complexed reagents. This approach allows the reaction to proceed at mild temperatures between 0°C and 50°C, effectively eliminating the need for energy-intensive cryogenic cooling systems while maintaining high product purity and yield. The versatility of this method is further demonstrated in its application for the functionalization of complex aromatic systems, such as the iodination of isoquinoline, showcasing its compatibility with sensitive heterocycles. 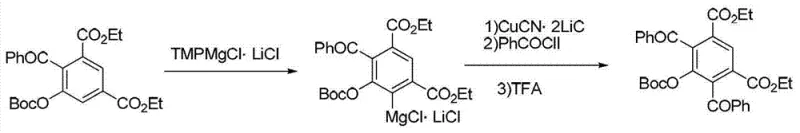 This strategic shift from stoichiometric reliance on expensive reagents to a catalytic one-pot synthesis represents a paradigm shift in cost reduction in organometallic manufacturing, making high-performance bases accessible for large-scale production.
This strategic shift from stoichiometric reliance on expensive reagents to a catalytic one-pot synthesis represents a paradigm shift in cost reduction in organometallic manufacturing, making high-performance bases accessible for large-scale production.
Mechanistic Insights into 1,10-Phenanthroline Catalyzed Metallation
The core innovation of this synthesis lies in the specific coordination chemistry facilitated by the 1,10-phenanthroline catalyst, which fundamentally alters the solubility and reactivity profile of the reaction components. Mechanistically, the nitrogen atoms within the phenanthroline structure coordinate strongly with the lithium ions present in the anhydrous lithium chloride, thereby significantly increasing the solubility of the lithium salt within the organic solvent medium. Simultaneously, this coordination environment interacts with the magnesium center of the isopropyl magnesium chloride, effectively activating the Grignard reagent and lowering the activation energy required for the deprotonation of the 2,2,6,6-tetramethyl piperidine. This dual coordination effect creates a highly reactive yet controlled species that drives the equilibrium towards the formation of the desired TMPMgCl·LiCl complex with exceptional efficiency. The result is a homogeneous reaction system that avoids the heterogeneous limitations often seen in non-catalyzed mixtures, ensuring consistent reagent quality and performance across different batches.
From an impurity control perspective, the ability to operate at elevated temperatures (0-50°C) rather than cryogenic conditions plays a pivotal role in minimizing the formation of unwanted byproducts. Traditional low-temperature processes often suffer from incomplete mixing or localized cold spots that can lead to uneven reagent distribution and subsequent side reactions upon warming. In contrast, the catalytic method ensures a uniform thermal profile throughout the reaction vessel, promoting complete conversion of raw materials and reducing the presence of unreacted starting materials that could complicate downstream purification. Moreover, the precise control over the catalyst loading, optimized between 0.1% and 1% molar weight, prevents the introduction of excessive organic impurities that might arise from ligand degradation. This meticulous balance ensures that the resulting high-purity TMP base meets the stringent specifications required for the synthesis of complex active pharmaceutical ingredients, where trace metal or organic contaminants can be detrimental to final drug safety.
How to Synthesize TMPMgCl·LiCl Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for producing this valuable organometallic reagent with high reproducibility and yield. The process begins with the dispersion of solid anhydrous lithium chloride and the liquid amine in a dry ether solvent, followed by the crucial addition of the phenanthroline catalyst to activate the system. Subsequent dropwise addition of the Grignard reagent under strict temperature control ensures that the exothermic nature of the reaction is managed safely while maximizing the formation of the mixed magnesium-lithium amide complex.
- Disperse anhydrous lithium chloride, 2,2,6,6-tetramethyl piperidine, and a catalytic amount of 1,10-phenanthroline in an organic solvent like THF under inert gas.
- Dropwise add isopropyl magnesium chloride solution at a controlled temperature between 0°C and 50°C while stirring.
- Maintain the reaction temperature for heat preservation until completion, then filter and wash to obtain the final TMPMgCl·LiCl solution.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this catalytic synthesis route offers profound strategic benefits that extend far beyond simple reagent substitution. The primary advantage lies in the drastic simplification of the raw material supply chain, as the process utilizes commodity chemicals like isopropyl magnesium chloride and lithium chloride instead of relying on niche, high-cost specialty reagents that are subject to volatile market pricing and limited availability. This shift significantly mitigates supply risk and enhances the overall resilience of the manufacturing operation, ensuring continuous production even during periods of global chemical shortages. Furthermore, the elimination of cryogenic requirements translates directly into substantial capital expenditure savings, as facilities no longer need to invest in or maintain complex low-temperature cooling infrastructure capable of reaching minus seventy degrees Celsius.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the replacement of expensive pre-formed i-PrMgCl·LiCl solutions with inexpensive, bulk-available starting materials combined with a catalytic amount of ligand. By avoiding the premium pricing associated with specialized organometallic complexes and reducing the energy consumption required for extreme cooling, the overall cost of goods sold for the final intermediate is significantly lowered. Additionally, the higher reaction yields reported in the patent mean that less raw material is wasted, further optimizing the material balance and reducing the cost per kilogram of the active reagent produced. This efficiency gain allows manufacturers to offer more competitive pricing for downstream pharmaceutical intermediates without compromising on quality or margin.
- Enhanced Supply Chain Reliability: Relying on commodity-grade raw materials such as anhydrous lithium chloride and standard Grignard reagents ensures a stable and diversified supply base that is less susceptible to disruptions than single-source specialty chemicals. The robustness of the one-pot synthesis method also reduces the dependency on complex multi-step logistics for reagent delivery, as the base can be generated on-demand at the point of use with minimal storage requirements. This flexibility allows supply chain planners to respond more agilely to fluctuating demand schedules, reducing inventory holding costs and minimizing the risk of reagent degradation during long-term storage. Consequently, the lead time for high-purity organometallic reagents can be effectively shortened, supporting faster time-to-market for new drug development programs.
- Scalability and Environmental Compliance: The operational simplicity of running reactions at near-ambient temperatures greatly facilitates the commercial scale-up of complex organometallics from laboratory benchtop to multi-ton production scales. The reduced energy footprint associated with avoiding cryogenic cooling aligns perfectly with modern sustainability goals and environmental regulations, lowering the carbon intensity of the manufacturing process. Moreover, the high selectivity of the catalytic system minimizes the generation of hazardous waste streams and byproducts, simplifying effluent treatment and disposal procedures. This combination of scalability and environmental stewardship makes the technology highly attractive for large-scale CDMO operations seeking to expand their capacity for advanced intermediate synthesis.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method in industrial settings. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on performance metrics and operational parameters. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this catalytic route into existing manufacturing workflows.
Q: What is the primary advantage of using 1,10-phenanthroline in TMPMgCl·LiCl synthesis?
A: The addition of 1,10-phenanthroline acts as a catalyst that coordinates with lithium ions, significantly increasing the solubility of lithium chloride and enhancing the reactivity of the Grignard reagent, leading to higher yields in a one-pot process.
Q: How does this method compare to traditional lithiation reagents regarding temperature control?
A: Unlike traditional lithium alkylides which often require cryogenic temperatures between -70°C and -90°C to prevent side reactions, this catalytic method operates efficiently at mild temperatures ranging from 0°C to 50°C, drastically simplifying thermal management.
Q: Can this reagent tolerate sensitive functional groups during metallation?
A: Yes, the TMPMgCl·LiCl reagent generated via this method exhibits superior functional group tolerance compared to standard lithium bases, allowing for the regioselective deprotonation of aromatic hydrocarbons containing esters, nitro groups, and ketones without degradation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable TMPMgCl·LiCl Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced organometallic reagents play in the efficient synthesis of next-generation pharmaceutical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of innovative technologies like the 1,10-phenanthroline catalyzed synthesis are fully realized at an industrial level. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical techniques to verify every batch. Our capability to handle sensitive organometallic chemistry under inert atmospheres guarantees the stability and performance of the reagents we supply, providing our partners with the confidence needed to advance their drug discovery pipelines.
We invite you to collaborate with us to leverage these technological advancements for your specific project needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your current manufacturing processes. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized synthesis methods can enhance your supply chain efficiency and reduce overall production costs.
