Advanced ZnCl2 Catalyzed Synthesis of Chiral Pinanediol Esters for Commercial Scale-up
The pharmaceutical and fine chemical industries continuously seek robust methodologies for synthesizing chiral building blocks, particularly boron-containing intermediates essential for asymmetric synthesis. Patent CN101220048B introduces a transformative approach to preparing chiral pinanediol esters, a critical class of compounds utilized extensively in the production of high-value active pharmaceutical ingredients such as Velcade. This technology addresses long-standing challenges in organoboron chemistry by replacing hazardous and expensive lithium reagents with a more manageable Grignard-based system catalyzed by anhydrous zinc chloride. By shifting the paradigm from cryogenic lithium chemistry to a zinc-mediated process, the patent outlines a pathway that not only enhances reaction yields to over 90 percent but also drastically simplifies the operational complexity required for industrial manufacturing. For R&D directors and process chemists, this represents a significant opportunity to optimize impurity profiles and streamline scale-up protocols without compromising the stereochemical integrity of the final product.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of these valuable chiral esters relied heavily on the use of dihalomethyl lithium reagents, specifically LiCHCl2, generated in situ at extreme cryogenic temperatures around minus 100 degrees Celsius. As illustrated in the prior art reaction scheme, this conventional pathway presents severe logistical and safety hurdles for large-scale production. The lithium reagent is notoriously sensitive to both air and moisture, necessitating rigorous exclusion of atmospheric conditions during transfer and reaction, which increases capital expenditure on specialized equipment. Furthermore, the subsequent step involving the addition of solid zinc chloride often introduces trace amounts of water due to the hygroscopic nature of the salt, leading to the decomposition of the sensitive lithium intermediate. This degradation results in inconsistent yields and complicates the purification process, as the formation of oligomeric boronic acid anhydrides and other hydrolysis byproducts creates a difficult-to-separate impurity profile that burdens downstream processing teams.
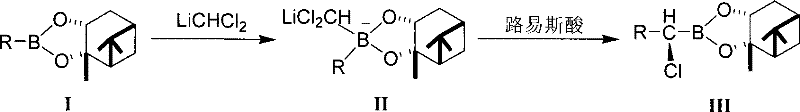
The Novel Approach
In stark contrast to the legacy lithium methodology, the novel approach detailed in the patent utilizes a Grignard reagent (RMgX) coupled with a specifically prepared anhydrous zinc chloride catalyst to drive the transformation efficiently. This new reaction route eliminates the need for ultra-low temperature lithium generation, instead operating at more accessible temperatures ranging from minus 60 to minus 78 degrees Celsius. The strategic use of an ether solution of zinc chloride, rather than the solid powder, ensures that no exogenous moisture is introduced into the reaction vessel, thereby preserving the integrity of the organoboron intermediate. This modification allows the reaction to proceed with exceptional smoothness, naturally warming to room temperature for the final equilibration step. The result is a robust process that delivers total yields exceeding 90 percent, providing a reliable supply of high-purity material suitable for immediate use in subsequent asymmetric coupling reactions without extensive chromatographic purification.
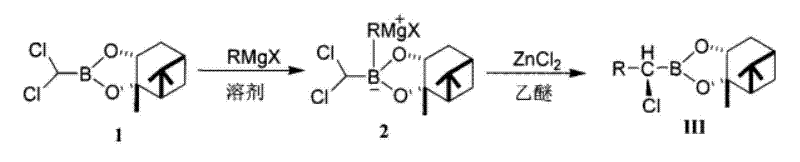
Mechanistic Insights into ZnCl2-Catalyzed Substitution
The core mechanistic advantage of this process lies in the dual role of the zinc species as both a transmetallation mediator and a Lewis acid activator. When the Grignard reagent reacts with the dichloromethylboronic acid pinanediol ester, it forms a transient boronate complex. The introduction of anhydrous ZnCl2 facilitates the displacement of the remaining chlorine atom through a coordinated transition state that lowers the activation energy for the substitution. Crucially, the patent emphasizes the preparation of the zinc chloride catalyst via vacuum dehydration at 150 degrees Celsius, often in the presence of dehydrating agents like phosphorus pentoxide. This pre-treatment step is vital because even trace water can hydrolyze the boron-carbon bond, leading to the formation of boric acid derivatives that act as dead-end impurities. By maintaining a strictly anhydrous environment through the use of vacuum-dried catalysts and inert gas protection, the reaction selectively favors the formation of the desired carbon-boron bond while suppressing competitive hydrolysis pathways.
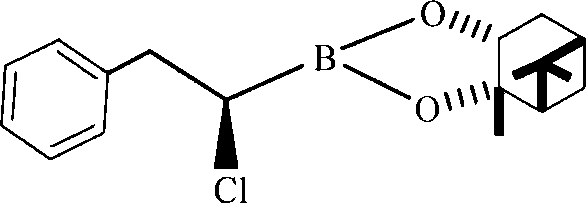
Furthermore, the steric environment provided by the pinanediol ligand plays a pivotal role in stabilizing the boron center against racemization. The bulky bicyclic structure of the pinanediol moiety shields the boron atom, ensuring that the stereochemical information encoded in the starting material is retained throughout the substitution process. This is exemplified by the successful synthesis of specific derivatives such as 3-phenyl-2-chloro-propylboronic acid-alpha-pinanediol ester, where the structural integrity is maintained with high fidelity. The ability to control the reaction kinetics by adjusting the addition rate of the Grignard reagent and the concentration of the zinc catalyst allows process chemists to fine-tune the impurity profile, minimizing the formation of homocoupling byproducts or over-alkylated species that often plague organometallic reactions. This level of control is essential for meeting the stringent purity specifications required by regulatory bodies for pharmaceutical intermediates.
How to Synthesize Chiral Pinanediol Esters Efficiently
Implementing this synthesis requires careful attention to the preparation of reagents, specifically the dehydration of the zinc chloride catalyst and the standardization of the Grignard solution. The protocol dictates that the zinc chloride must be processed under vacuum to remove lattice water before being dissolved in anhydrous ether, creating a homogeneous catalyst solution that can be dosed precisely. Following the initial addition of the Grignard reagent to the boron substrate at low temperature, the catalyst solution is introduced, and the mixture is allowed to warm slowly to ambient temperature over a period of 15 to 24 hours. This slow warming phase is critical for ensuring complete conversion while managing the exotherm safely. Detailed standardized operating procedures regarding stoichiometry, solvent drying, and workup protocols are essential for reproducibility.
- Preparation of anhydrous ZnCl2 catalyst via vacuum dehydration at 150°C to ensure moisture-free conditions.
- Generation of Grignard reagent (RMgX) in ether solvent under inert gas protection at controlled temperatures.
- Reaction of dichloromethylboronic acid-pinanediol ester with Grignard reagent followed by ZnCl2 addition and standard aqueous workup.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the transition from lithium-based chemistry to this zinc-catalyzed Grignard protocol offers profound advantages in terms of cost structure and supply chain resilience. The reliance on n-butyllithium and dichloromethane at cryogenic temperatures in the old method incurs significant costs related to specialized storage, handling safety, and energy consumption for cooling. By replacing these with magnesium turnings and alkyl halides, which are commodity chemicals available from multiple global suppliers, the raw material cost base is significantly reduced. Moreover, the elimination of the need for minus 100 degrees Celsius cooling infrastructure lowers the barrier to entry for contract manufacturing organizations, allowing for broader sourcing options and reducing the risk of supply bottlenecks associated with single-source specialty reagents.
- Cost Reduction in Manufacturing: The replacement of expensive, pyrophoric lithium reagents with stable Grignard reagents leads to a drastic reduction in raw material expenditure. Additionally, the simplified workup procedure, which involves simple filtration of magnesium salts followed by aqueous washing, eliminates the need for complex quenching steps and extensive chromatographic purification, thereby reducing solvent consumption and waste disposal costs substantially.
- Enhanced Supply Chain Reliability: Utilizing widely available magnesium and organic halides ensures a stable supply of starting materials, mitigating the risks associated with the volatile market for organolithium compounds. The robustness of the reaction conditions, which tolerate a wider range of operational parameters compared to the sensitive lithium route, ensures consistent batch-to-batch quality and reduces the likelihood of production delays caused by reagent instability or strict environmental controls.
- Scalability and Environmental Compliance: The process generates fewer hazardous byproducts and avoids the use of large volumes of chlorinated solvents required for lithium quenches, aligning better with green chemistry principles. The ability to run the reaction at higher concentrations and warmer temperatures facilitates easier heat transfer management in large reactors, making the commercial scale-up of complex pharmaceutical intermediates more feasible and environmentally sustainable.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this zinc-catalyzed synthesis method. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, focusing on practical considerations for process development and quality assurance teams evaluating this technology for adoption.
Q: Why is anhydrous ZnCl2 critical for this synthesis?
A: Conventional solid ZnCl2 often contains absorbed moisture which decomposes sensitive intermediates. The patent specifies vacuum-dehydrated ZnCl2 or ether solutions to prevent hydrolysis, ensuring yields exceed 90%.
Q: How does this method improve upon Lithium-based routes?
A: Traditional methods utilize LiCHCl2 generated at -100°C, which is extremely air-sensitive and costly. The ZnCl2 catalyzed Grignard route operates at milder temperatures (-60°C to -78°C) using stable, cost-effective reagents.
Q: What represents the key cost advantage for procurement?
A: The elimination of cryogenic lithium reagents and the use of common magnesium turnings significantly reduce raw material costs. Additionally, the simplified workup reduces processing time and waste disposal expenses.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Pinanediol Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality chiral intermediates play in the development of next-generation therapeutics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to pilot plant is seamless. We adhere to stringent purity specifications and utilize rigorous QC labs to verify that every batch of pinanediol ester meets the exacting standards required for GMP manufacturing. Our facility is equipped to handle the specific dehydration and anhydrous processing requirements outlined in the patent, guaranteeing the delivery of material with superior optical purity and minimal impurity levels.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the potential economic benefits of switching to this zinc-catalyzed method for your specific project needs. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your target molecules, ensuring a reliable and cost-effective supply of these essential chiral building blocks for your drug development programs.
