Advanced Iron-Catalyzed Synthesis of Cyanoalkyl Tetra-Substituted Alkenes for Commercial Scale-Up
Introduction to Patent CN111253293A and Technological Breakthroughs
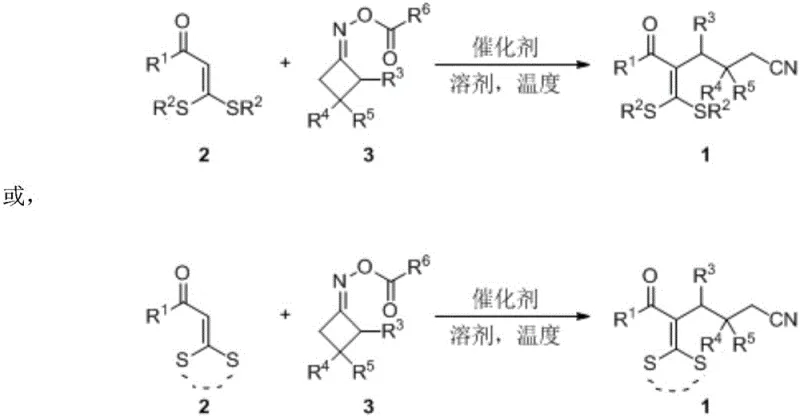
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex molecular architectures efficiently. Patent CN111253293A introduces a significant advancement in the synthesis of cyanoalkyl-substituted tetra-substituted alkene derivatives, which serve as critical scaffolds in medicinal chemistry and organic functional materials. This technology leverages a novel intermolecular radical addition strategy, utilizing readily available 3,3-dialkylthio-2-propen-1-one compounds and cyclobutanone oxime esters as starting materials. By employing an inexpensive iron salt catalyst, the process achieves a one-step construction of carbon-carbon bonds under mild conditions, bypassing the need for costly noble metals. For R&D directors and procurement specialists, this represents a pivotal shift towards more sustainable and economically viable manufacturing processes for high-purity pharmaceutical intermediates.
The structural diversity offered by this synthetic route is particularly noteworthy for supply chain planners looking to secure reliable sources of complex building blocks. The ability to tune various substituents (R1 through R5) allows for the generation of a wide library of derivatives from a common platform, enhancing the flexibility of production lines. Furthermore, the presence of multiple functional groups such as carbonyls and cyano groups in the final product facilitates downstream diversification, enabling the rapid synthesis of bioactive candidates. This patent not only addresses the technical challenges of forming tetra-substituted alkenes but also aligns with modern green chemistry principles by minimizing waste and utilizing earth-abundant catalysts.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of polysubstituted alkenes has relied heavily on transition metal-catalyzed cross-coupling reactions, which often necessitate the use of precious metals such as palladium, rhodium, ruthenium, or iridium. These conventional pathways typically require extensive pre-functionalization of the starting materials, involving steps like halogenation, silylation, boronation, or trifluoromethanesulfonylation to activate the carbon centers for coupling. Such multi-step sequences not only increase the overall production time and labor costs but also result in poor atom economy due to the generation of stoichiometric amounts of metal salts and leaving group byproducts. For large-scale manufacturing, the reliance on expensive noble metals poses significant supply chain risks and cost volatility, while the removal of trace metal residues to meet stringent pharmaceutical purity standards adds further complexity and expense to the purification process.
The Novel Approach
In stark contrast, the methodology disclosed in CN111253293A offers a streamlined, one-pot solution that eliminates the need for pre-functionalized substrates and expensive catalysts. By utilizing cyclobutanone oxime esters as radical precursors, the reaction generates cyanoalkyl radicals in situ which directly add to the double bond of 3,3-dialkylthio-2-propen-1-one compounds. This radical addition mechanism proceeds efficiently under the catalysis of simple iron salts, such as ferric chloride, which are orders of magnitude cheaper and more environmentally benign than their noble metal counterparts. The process operates under relatively mild thermal conditions, typically around 110°C in solvents like trifluorotoluene, ensuring high reaction efficiency with yields reaching up to 84%. This approach drastically simplifies the operational workflow, reduces the environmental footprint, and provides a cost-effective route for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into FeCl3-Catalyzed Radical Addition
The core of this technological innovation lies in the iron-catalyzed generation of iminyl radicals from cyclobutanone oxime esters, which subsequently undergo ring-opening to form distal cyanoalkyl radicals. Upon heating in the presence of FeCl3, the oxime ester undergoes homolytic cleavage of the N-O bond, initiating a radical cascade that is highly selective for the formation of the desired carbon-carbon bond. The generated cyanoalkyl radical attacks the electron-deficient double bond of the 3,3-dialkylthio-2-propen-1-one synthon, leading to the formation of a new carbon-centered radical intermediate. This intermediate then abstracts a hydrogen atom or undergoes further oxidation to terminate the cycle, yielding the stable tetra-substituted alkene product. Understanding this mechanistic pathway is crucial for R&D teams aiming to optimize reaction parameters or adapt the chemistry to novel substrate classes, as it highlights the tolerance of the system to various functional groups and steric environments.
From an impurity control perspective, the radical nature of this reaction offers distinct advantages over ionic pathways that might be prone to side reactions like elimination or rearrangement. The use of iron salts minimizes the risk of heavy metal contamination, a critical factor for API intermediate manufacturing where residual metal limits are strictly regulated. Furthermore, the reaction conditions are sufficiently robust to accommodate a variety of substituents on both the oxime ester and the alkene synthon, as evidenced by the successful synthesis of derivatives with aryl, alkyl, and heteroatom groups. This mechanistic robustness ensures consistent product quality and simplifies the purification strategy, typically requiring only standard silica gel column chromatography to achieve high-purity isolates suitable for subsequent biological evaluation or process development.
How to Synthesize Cyanoalkyl-Substituted Tetra-Substituted Alkenes Efficiently
The practical implementation of this synthesis is designed for straightforward execution in standard laboratory or pilot plant settings, requiring minimal specialized equipment beyond basic heating and inert atmosphere capabilities. The protocol involves mixing the alkene synthon and the oxime ester in a polar aprotic solvent, followed by the addition of the iron catalyst and heating to the specified temperature. Detailed standardized synthesis steps, including precise molar ratios, solvent volumes, and workup procedures, are outlined in the guide below to ensure reproducibility and safety during scale-up operations. Adhering to these optimized conditions allows manufacturers to maximize yield while maintaining the structural integrity of the sensitive functional groups present in the molecule.
- Combine 3,3-dialkylthio-2-propen-1-one compound and cyclobutanone oxime ester in a reaction vessel under an inert atmosphere.
- Add iron salt catalyst (preferably FeCl3 at 10 mol%) and trifluorotoluene solvent to the mixture.
- Heat the reaction mixture to 110°C for 24 hours, then purify the resulting cyanoalkyl-substituted alkene via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this iron-catalyzed technology translates into tangible strategic benefits regarding cost stability and operational resilience. By shifting away from precious metal catalysts to abundant iron salts, companies can insulate their production costs from the volatile market fluctuations associated with palladium and rhodium pricing. Additionally, the elimination of pre-functionalization steps reduces the total number of unit operations required, thereby lowering labor costs, energy consumption, and solvent usage. This streamlined process not only accelerates the time-to-market for new intermediates but also enhances the overall sustainability profile of the manufacturing site, aligning with increasingly strict environmental regulations and corporate social responsibility goals.
- Cost Reduction in Manufacturing: The substitution of expensive noble metal catalysts with inexpensive iron salts significantly lowers the direct material cost per kilogram of the final product. Furthermore, the one-step nature of the reaction eliminates the need for separate halogenation or boronation stages, reducing the consumption of reagents and the generation of hazardous waste streams. This consolidation of synthetic steps leads to substantial savings in utility costs and waste disposal fees, contributing to a more lean and efficient production model that improves overall profit margins without compromising product quality.
- Enhanced Supply Chain Reliability: Relying on earth-abundant iron catalysts mitigates the supply risk associated with geopolitically sensitive precious metals, ensuring a more stable and predictable supply chain for critical raw materials. The starting materials, 3,3-dialkylthio-2-propen-1-one compounds and cyclobutanone oxime esters, are structurally diverse and can be sourced or synthesized from readily available commodity chemicals, reducing dependency on single-source suppliers. This flexibility allows procurement teams to negotiate better terms and secure long-term contracts, safeguarding production schedules against potential disruptions in the global chemical market.
- Scalability and Environmental Compliance: The mild reaction conditions and high atom economy of this process make it inherently scalable from gram-scale laboratory synthesis to multi-ton commercial production. The reduced generation of toxic byproducts and the absence of heavy metal residues simplify the wastewater treatment and exhaust gas handling requirements, facilitating easier compliance with environmental protection standards. This environmental compatibility not only reduces regulatory burdens but also enhances the company's reputation as a responsible manufacturer, which is increasingly valued by downstream partners in the pharmaceutical and agrochemical sectors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the patent specifications and experimental data. These insights are intended to clarify the operational feasibility and strategic value of adopting this iron-catalyzed route for your specific manufacturing needs. For further technical details or custom feasibility studies, please refer to the comprehensive documentation provided by our technical team.
Q: What are the primary advantages of this iron-catalyzed method over traditional transition metal catalysis?
A: Unlike conventional methods relying on expensive palladium, rhodium, or iridium catalysts which require pre-functionalized substrates like halides or boronates, this method utilizes inexpensive and relatively non-toxic iron salts (FeCl3). It achieves direct C-C bond construction via radical addition without the need for prior halogenation or silicization, significantly improving atom economy and reducing raw material costs.
Q: What is the expected yield range and stereoselectivity for this synthesis?
A: According to the patent data, the reaction demonstrates high efficiency with isolated yields ranging from 42% to 84% depending on the specific substituents. The process also exhibits good stereoselectivity, producing tetra-substituted alkene derivatives with defined structural integrity suitable for further functionalization into bioactive molecules.
Q: Can the resulting alkene derivatives be further functionalized for drug discovery?
A: Yes, the synthesized cyanoalkyl-substituted tetra-substituted alkenes contain multiple reactive handles including carbonyl, alkylthio, and cyano groups. These functional groups allow for diverse downstream transformations, such as cyclization into pyrazole derivatives, making them highly valuable scaffolds for medicinal chemistry and the development of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cyanoalkyl-Substituted Tetra-Substituted Alkene Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the iron-catalyzed radical addition technology described in CN111253293A for the production of high-value pharmaceutical intermediates. As a seasoned CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of cyanoalkyl-substituted alkene derivatives delivered meets the highest standards of quality and consistency required by global regulatory bodies.
We invite you to collaborate with us to leverage this cost-effective and sustainable synthesis route for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline constraints. By partnering with us, you gain access to specific COA data and route feasibility assessments that will empower you to make informed decisions about your supply chain strategy. Contact us today to discuss how we can support your growth with reliable, high-quality chemical solutions.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
