Advanced One-Pot Synthesis of 2-Methylpyridine Intermediates for Pharmaceutical Manufacturing
The pharmaceutical and agrochemical industries continuously seek robust, scalable, and environmentally benign pathways for constructing nitrogen-containing heterocycles. Patent CN108314642B introduces a groundbreaking methodology for the synthesis of 2-methylpyridine compounds, a critical structural motif found in numerous bioactive molecules including long-acting sulfonamides, antihistamines like chlorpheniramine, and herbicides such as paraquat. This innovative approach leverages a transition-metal-free, iodine-catalyzed oxidative cyclization strategy that transforms simple, commercially available triethylamine and oxime acetates into valuable 2-methylpyridine derivatives. By utilizing molecular oxygen as the terminal oxidant, this protocol not only aligns with green chemistry principles but also circumvents the severe safety hazards and energy-intensive requirements associated with legacy manufacturing processes. The ability to access these scaffolds through a direct one-pot tandem reaction represents a significant leap forward in process chemistry, offering a reliable pharmaceutical intermediates supplier pathway that prioritizes both efficiency and sustainability.
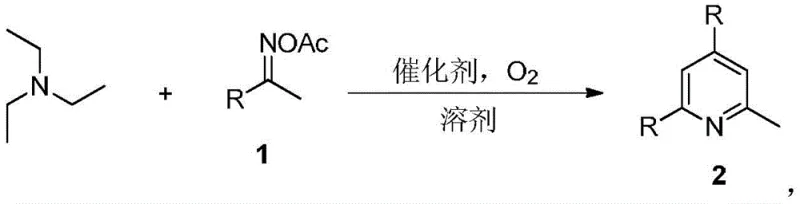
Historically, the industrial production of 2-methylpyridine has relied heavily on archaic methodologies such as the acetaldehyde condensation method, the acetylene method, or the acrylonitrile route. These conventional techniques are plagued by inherent limitations that severely impact operational expenditure and safety profiles. Specifically, they necessitate extreme reaction environments characterized by high temperatures and elevated pressures, which demand specialized, costly reactor infrastructure and rigorous safety protocols. Furthermore, these older pathways often suffer from poor atom economy, generating substantial quantities of hazardous waste and by-products that require complex, multi-step purification sequences to isolate the desired product. The low selectivity and frequent occurrence of side reactions in these high-energy processes lead to inconsistent yields, creating supply chain volatility and inflating the final cost of goods. For procurement managers and supply chain heads, these factors translate into unpredictable lead times and increased regulatory burdens regarding waste disposal and environmental compliance.
In stark contrast, the novel approach detailed in the patent data offers a paradigm shift towards streamlined manufacturing. By employing triethylamine as a readily accessible nitrogen source and coupling it with diverse oxime acetates, the reaction proceeds through a highly efficient one-pot series mechanism. This eliminates the need for isolating unstable intermediates, thereby reducing solvent consumption and processing time. The reaction conditions are remarkably mild, operating effectively between 120-160°C under a standard oxygen atmosphere, which removes the necessity for high-pressure equipment. The use of inexpensive iodine-based catalysts, such as elemental iodine, N-iodosuccinimide, or ammonium iodide, further drives down raw material costs while avoiding the contamination issues associated with precious transition metals like palladium or platinum. This transition-metal-free characteristic is particularly advantageous for pharmaceutical applications, where residual metal limits are strictly regulated, thus simplifying the downstream purification workflow and ensuring high-purity pharmaceutical intermediates.
Mechanistic Insights into Iodine-Catalyzed Oxidative Cyclization
The mechanistic underpinning of this transformation involves a sophisticated radical-mediated cascade initiated by the iodine catalyst under aerobic conditions. Initially, the iodine species activates the oxime acetate moiety, facilitating the generation of reactive iminyl radicals upon thermal decomposition. Concurrently, triethylamine serves as a two-carbon synthon and a nitrogen donor, undergoing oxidative dealkylation to provide the necessary fragments for pyridine ring construction. The presence of molecular oxygen is crucial, acting as a green oxidant to regenerate the active iodine catalyst and drive the thermodynamic equilibrium towards the aromatized pyridine product. This oxidative cyclization proceeds through a series of radical additions and eliminations that construct the six-membered heterocyclic core with high regioselectivity. The tolerance of the catalytic system to various functional groups suggests a robust radical mechanism that does not interfere with sensitive substituents on the aromatic ring, allowing for the late-stage functionalization of complex molecules.
From an impurity control perspective, this mechanism offers distinct advantages over acid-catalyzed condensations. The specificity of the radical pathway minimizes the formation of polymeric tars and oligomeric by-products that typically contaminate high-temperature acetaldehyde routes. The use of stoichiometric sodium thiosulfate during the workup phase effectively quenches residual iodine, preventing halogenation side reactions and ensuring a clean crude profile. This high level of chemoselectivity reduces the burden on purification teams, as the crude reaction mixtures often require only standard silica gel chromatography to achieve analytical purity. For R&D directors, understanding this mechanism validates the scalability of the process, as the radical chain propagation is efficient and does not rely on sensitive organometallic species that might degrade upon scale-up. The broad substrate scope demonstrated in the patent data confirms that electronic variations on the phenyl ring, whether electron-donating or withdrawing, do not significantly impede the radical cyclization efficiency.
How to Synthesize 2-Methylpyridine Compounds Efficiently
The practical execution of this synthesis is designed for operational simplicity, making it ideal for both laboratory optimization and pilot plant campaigns. The standard protocol involves charging a reaction vessel with triethylamine and the specific oxime acetate derivative in a molar ratio of approximately 2:1. A solvent such as toluene, acetonitrile, or 1,4-dioxane is added to facilitate mixing and heat transfer, with a typical loading of 4mL per mmol of substrate. The iodine catalyst is then introduced, and the system is pressurized with oxygen or maintained under an oxygen balloon. Heating the mixture to the optimal temperature range of 120-160°C initiates the cascade, with reaction times typically around 12 hours to ensure full conversion. Upon completion, the reaction is quenched with water, and the organic layer is washed with sodium thiosulfate to remove iodine residues, followed by standard drying and concentration. Detailed standardized synthesis steps are provided in the guide below.
- Dissolve triethylamine and oxime acetate compounds in a suitable solvent such as toluene, acetonitrile, or 1,4-dioxane.
- Add an iodine-based catalyst (Iodine, N-iodosuccinimide, or Ammonium iodide) to the reaction mixture.
- Heat the mixture to 120-160°C under an oxygen atmosphere for approximately 12 hours, then purify via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and supply continuity, this technology presents a compelling value proposition centered on cost efficiency and risk mitigation. The substitution of expensive, hazardous, or hard-to-source reagents with commodity chemicals like triethylamine and iodine drastically simplifies the procurement landscape. Triethylamine is a bulk chemical produced globally in massive quantities, ensuring a stable supply chain that is immune to the geopolitical fluctuations often seen with specialty catalysts. The elimination of transition metals removes the need for expensive scavenging resins and extensive analytical testing for heavy metal residues, which are significant cost drivers in API manufacturing. Furthermore, the one-pot nature of the reaction consolidates multiple unit operations into a single vessel, reducing labor costs, energy consumption, and facility occupancy time. This intensification of the process directly contributes to substantial cost savings in fine chemical intermediates manufacturing without compromising on quality or yield.
- Cost Reduction in Manufacturing: The economic benefits of this route are derived primarily from the use of low-cost starting materials and the avoidance of precious metal catalysts. Traditional methods often require palladium or rhodium complexes that are not only expensive to purchase but also difficult to recover and recycle. By utilizing elemental iodine or ammonium iodide, the catalyst cost becomes negligible relative to the overall batch cost. Additionally, the mild reaction conditions reduce energy expenditures associated with heating and cooling cycles. The high atom economy of the tandem reaction means less raw material is wasted as by-product, maximizing the yield per kilogram of input. This efficiency translates into a lower cost of goods sold (COGS), allowing manufacturers to maintain healthy margins even in competitive markets. The simplified workup procedure also reduces solvent usage and waste treatment fees, further enhancing the financial viability of the process.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the reliance on widely available, non-regulated reagents. Unlike specialized organometallic catalysts that may have long lead times or single-source suppliers, iodine and triethylamine are stocked by numerous chemical distributors worldwide. This redundancy ensures that production schedules are not disrupted by raw material shortages. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in reagent quality, reducing the rate of batch failures. For supply chain heads, this reliability is critical for maintaining just-in-time delivery commitments to downstream pharmaceutical clients. The ability to scale this reaction from gram to tonnage without changing the fundamental chemistry provides a seamless path from clinical trial material to commercial launch, mitigating the risks associated with technology transfer between different manufacturing sites.
- Scalability and Environmental Compliance: Environmental regulations are becoming increasingly stringent, and this synthetic route is well-positioned to meet future compliance standards. The use of molecular oxygen as the oxidant generates water as the primary by-product, avoiding the generation of stoichiometric amounts of toxic heavy metal waste or chlorinated organic by-products common in other oxidation methods. The absence of high-pressure requirements simplifies the engineering controls needed for scale-up, allowing the reaction to be run in standard glass-lined or stainless steel reactors. This ease of scale-up facilitates rapid capacity expansion to meet market demand. Moreover, the reduced E-factor (mass of waste per mass of product) aligns with corporate sustainability goals, enhancing the brand reputation of manufacturers who adopt this green chemistry approach. The process inherently supports the principles of green chemistry by designing out hazards and minimizing waste at the source.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this iodine-catalyzed synthesis platform. These insights are derived directly from the experimental data and scope evaluation presented in the patent literature, providing a clear understanding of the technology's capabilities and limitations. Understanding these details is essential for project managers evaluating the feasibility of integrating this route into existing production lines. The answers reflect the consensus on reaction performance, safety profiles, and applicability across different chemical spaces.
Q: What are the primary advantages of this iodine-catalyzed method over traditional acetaldehyde routes?
A: This method operates under milder conditions (120-160°C) without high pressure, avoids toxic transition metals, and utilizes cheap triethylamine, significantly reducing environmental impact and purification costs compared to high-temperature acetaldehyde or acetylene methods.
Q: What is the substrate scope for this 2-methylpyridine synthesis?
A: The reaction tolerates a wide range of substituents on the oxime acetate, including electron-donating groups like methyl and methoxy, electron-withdrawing groups like halogens and nitro, as well as heterocycles like thiophene and naphthyl groups.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the one-pot nature eliminates intermediate isolation steps, and the use of molecular oxygen as the oxidant alongside inexpensive reagents makes it highly scalable and economically viable for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Methylpyridine Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient intermediate synthesis in the global pharmaceutical supply chain. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative academic discoveries like this iodine-catalyzed method are successfully translated into industrial reality. Our state-of-the-art facilities are equipped to handle the specific requirements of oxidative cyclizations, including dedicated oxygen handling systems and corrosion-resistant reactor trains. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2-methylpyridine compounds meets the highest international standards for pharmaceutical use. Our commitment to quality assurance ensures that impurity profiles are tightly controlled, facilitating smoother regulatory filings for our clients.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthetic methodology can be tailored to your specific project needs. Whether you require custom synthesis of novel analogs or large-scale production of established intermediates, we are prepared to offer a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this greener route. We encourage you to contact us to request specific COA data and route feasibility assessments for your target molecules. By collaborating with us, you gain access to a supply chain partner dedicated to driving innovation, reducing costs, and ensuring the reliable delivery of high-purity pharmaceutical intermediates essential for developing the next generation of life-saving therapies.
