Advanced Synthetic Routes for Substituted Pyrazoles: Scaling High-Purity Agrochemical Intermediates
The global demand for high-performance insecticides continues to drive innovation in the synthesis of key heterocyclic intermediates. Patent CN111825653A, filed in late 2020, presents a significant technological breakthrough in the preparation of substituted pyrazole compounds, specifically targeting the efficient production of precursors for anthranilamide insecticides like chlorantraniliprole. This intellectual property addresses long-standing challenges in the industrial synthesis of Formula I compounds, such as 3-bromo-1-(3-chloro-2-pyridyl)-5-methyl-1H-pyrazole. By introducing novel pathways for constructing the pyrazole-pyridine scaffold, the patent offers a robust framework for manufacturers seeking to optimize their production lines. The disclosed methods not only improve reaction yields but also streamline the purification processes, making them highly attractive for large-scale operations. For R&D teams and procurement strategists, understanding these synthetic advancements is crucial for securing a competitive edge in the agrochemical supply chain.
Furthermore, the technical depth of CN111825653A extends beyond simple molecule construction; it provides a comprehensive solution for generating high-purity intermediates essential for next-generation crop protection agents. The patent details multiple embodiments, ranging from direct alkylation strategies to oxidative conversions, allowing flexibility in process design depending on available raw materials and equipment capabilities. This versatility is particularly valuable for contract development and manufacturing organizations (CDMOs) aiming to diversify their service offerings. As the industry moves towards more sustainable and cost-effective manufacturing practices, the methodologies outlined in this patent serve as a benchmark for modern intermediate production. Implementing these techniques can significantly enhance the reliability of supply for critical active pharmaceutical ingredients (APIs) and agrochemical actives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of pyrazole precursors for anthranilamides has been plagued by inefficiencies that hinder industrial scalability. Prior art, including various PCT publications, often describes transformations that are theoretically sound but practically limited when applied to industrial-scale batch synthesis. A major bottleneck in conventional methods is the lack of effective industrial-scale ways to prepare starting methylpyrazole compounds. Many existing routes rely on complex multi-step sequences that suffer from low overall yields and generate substantial waste streams. Additionally, traditional alkylation and oxidation steps frequently require harsh conditions or expensive catalysts that are difficult to remove, leading to impurity profiles that complicate downstream processing. These limitations result in higher production costs and inconsistent supply quality, posing significant risks for procurement managers responsible for maintaining continuous manufacturing lines.
The Novel Approach
In contrast, the approach detailed in CN111825653A introduces a streamlined pathway that directly addresses these scalability issues. The patent discloses a method for preparing Formula I compounds by reacting Formula II compounds with halogen-substituted pyridines under optimized conditions. This novel route eliminates several intermediate isolation steps found in older methodologies, thereby reducing material loss and processing time. By utilizing accessible reagents such as 2,3-dichloropyridine and specific pyrazole derivatives, the process achieves superior conversion rates. The invention also provides alternative routes involving the oxidation of methyl groups to carboxylic acids, offering flexibility in synthetic strategy. This adaptability allows manufacturers to select the most cost-effective route based on their specific infrastructure, ultimately facilitating cost reduction in agrochemical manufacturing through improved process efficiency and reduced waste generation.
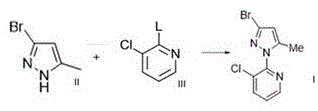
The core of this innovation lies in the precise control of reaction parameters during the coupling of the pyrazole and pyridine rings. As illustrated in the reaction schemes, the process leverages specific base and solvent systems to drive the nucleophilic substitution to completion. This level of control is essential for minimizing side reactions that typically plague heterocyclic coupling. The ability to produce Formula I compounds with high fidelity ensures that downstream synthesis of chlorantraniliprole proceeds smoothly, reducing the burden on purification units. For supply chain heads, this translates to a more predictable production schedule and reduced risk of batch failures. The patent's emphasis on industrial applicability means that these methods are not just laboratory curiosities but viable solutions for ton-scale production.
Mechanistic Insights into Nucleophilic Substitution and Oxidation
The mechanistic foundation of the disclosed synthesis relies on a carefully orchestrated nucleophilic aromatic substitution followed by selective oxidation. In the primary embodiment, the nitrogen atom of the pyrazole ring acts as a nucleophile, attacking the electron-deficient carbon of the chloropyridine ring. This reaction is facilitated by the presence of strong bases such as potassium carbonate or sodium hydroxide, which deprotonate the pyrazole nitrogen, increasing its nucleophilicity. The choice of solvent plays a critical role in stabilizing the transition state; polar aprotic solvents like N,N-dimethylacetamide (DMAc) or dimethyl sulfoxide (DMSO) are preferred for their ability to solvate cations while leaving the anionic nucleophile reactive. Furthermore, the addition of catalysts like potassium fluoride or crown ethers enhances the reaction rate by improving the solubility of inorganic salts in the organic phase, a phenomenon known as phase transfer catalysis.
Impurity control is another critical aspect addressed by the mechanistic design of this process. The patent specifies strict temperature controls, typically maintaining reaction temperatures between 100°C and 150°C, to prevent thermal degradation of the sensitive pyrazole ring. Post-reaction workup involves precise pH adjustments, often acidifying the mixture to pH less than 4 to precipitate the desired carboxylic acid products while keeping soluble impurities in the aqueous phase. This selective precipitation is a powerful tool for achieving purities exceeding 90% without the need for extensive chromatography. For R&D directors, understanding these mechanistic nuances is vital for troubleshooting potential scale-up issues. By adhering to the specified molar ratios and addition rates, manufacturers can ensure consistent product quality and minimize the formation of regioisomers or over-oxidized byproducts.
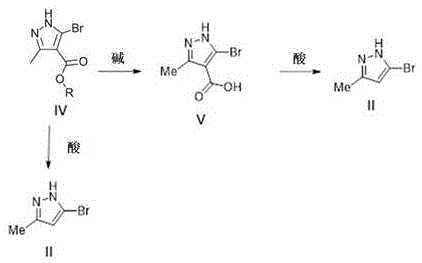
How to Synthesize 3-Bromo-5-methyl-1H-pyrazole Efficiently
The synthesis of the key intermediate 3-bromo-5-methyl-1H-pyrazole (Formula II) serves as the foundational step for the entire value chain described in the patent. This process begins with the bromination of ethyl 3-methyl-1H-pyrazole-4-carboxylate, followed by hydrolysis to the corresponding acid and subsequent decarboxylation. The patent provides detailed experimental conditions for each step, emphasizing the importance of controlling exothermic reactions during bromination and managing gas evolution during decarboxylation. These operational details are critical for ensuring safety and reproducibility in a plant setting. The detailed standardized synthesis steps see the guide below for a comprehensive breakdown of the procedure.
- Prepare the pyrazole precursor (Formula II) via bromination of ethyl 3-methyl-1H-pyrazole-4-carboxylate followed by hydrolysis and decarboxylation.
- React the Formula II compound with 2,3-dichloropyridine (Formula III) in the presence of a base like potassium carbonate and a catalyst system such as KF/18-crown-6 in DMAc at 150°C.
- Isolate the final Formula I product through filtration and drying, achieving high purity suitable for downstream chlorantraniliprole synthesis.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of the technologies described in CN111825653A offers tangible strategic benefits beyond mere technical feasibility. The primary advantage lies in the simplification of the supply chain for critical raw materials. By enabling the use of widely available starting materials like ethyl acetoacetate and hydrazine hydrate, the process reduces dependency on specialized or scarce reagents. This shift significantly enhances supply chain reliability, ensuring that production schedules are not disrupted by raw material shortages. Moreover, the robustness of the reaction conditions allows for the use of standard stainless steel reactors, eliminating the need for exotic metallurgy that drives up capital expenditure. These factors collectively contribute to a more resilient and cost-efficient manufacturing ecosystem.
- Cost Reduction in Manufacturing: The elimination of complex purification steps and the use of inexpensive inorganic bases directly lower the variable costs associated with production. By avoiding expensive transition metal catalysts that require rigorous removal protocols, the process simplifies the downstream workflow. This reduction in processing complexity translates to lower energy consumption and reduced labor hours per kilogram of product. Furthermore, the high yields reported in the patent examples mean that less raw material is wasted, maximizing the return on investment for every batch produced. These efficiencies accumulate to provide substantial cost savings over the lifecycle of the product.
- Enhanced Supply Chain Reliability: The versatility of the synthetic routes allows manufacturers to pivot between different precursors based on market availability. If one specific pyrazole derivative becomes scarce, the process can be adapted to use alternative starting materials without compromising the final product quality. This flexibility is a crucial risk mitigation strategy in the volatile chemical market. Additionally, the stability of the intermediates ensures that they can be stored and transported safely, reducing the pressure for just-in-time delivery and allowing for strategic stockpiling. This reliability is essential for meeting the demanding delivery timelines of global agrochemical companies.
- Scalability and Environmental Compliance: The processes described are inherently scalable, having been designed with industrial batch synthesis in mind. The use of aqueous workups and standard organic solvents facilitates easy waste treatment and solvent recovery, aligning with modern environmental regulations. The minimization of hazardous byproducts reduces the burden on waste management systems and lowers disposal costs. As regulatory scrutiny on chemical manufacturing intensifies, adopting greener and more efficient processes becomes a competitive necessity. This patent provides a pathway to achieve high production volumes while maintaining a minimal environmental footprint.
Frequently Asked Questions (FAQ)
The following questions address common inquiries regarding the technical implementation and commercial viability of the patented processes. These insights are derived directly from the experimental data and claims within CN111825653A, providing clarity for stakeholders evaluating this technology. Understanding these details helps in making informed decisions about process adoption and partnership opportunities. The answers reflect the practical realities of scaling these chemical transformations from the laboratory to the production floor.
Q: What are the key advantages of the new pyrazole synthesis route described in CN111825653A?
A: The patent outlines methods that overcome the limited industrial utility of prior art by providing scalable routes for producing starting methylpyrazoles and their subsequent conversion to carboxylic acid precursors efficiently.
Q: How does this process ensure high purity for agrochemical applications?
A: The process utilizes specific workup procedures including pH adjustment, extraction, and recrystallization steps which optimize purity to exceed 90%, critical for downstream insecticide synthesis.
Q: Is this method suitable for large-scale commercial production?
A: Yes, the patent explicitly describes embodiments suitable for industrial-scale batch synthesis, utilizing common reagents and solvents that facilitate commercial scale-up of complex agrochemical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Bromo-5-methyl-1H-pyrazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the production of advanced agrochemicals. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. By leveraging the advanced synthetic methodologies outlined in patents like CN111825653A, we can offer customized manufacturing solutions that optimize both cost and quality for our global partners.
We invite you to engage with our technical procurement team to discuss how we can support your specific project requirements. Whether you need a Customized Cost-Saving Analysis for your current supply chain or require specific COA data and route feasibility assessments for new developments, we are here to assist. Partnering with us ensures access to reliable agrochemical intermediate supplies backed by deep technical expertise and a commitment to excellence. Contact us today to explore how we can drive value and efficiency in your manufacturing operations.
