Advanced Copper-Catalyzed Synthesis of 4-Polyfluoroalkyl-3,5-Dicarbonylpyrazoles for Commercial Scale-Up
The pharmaceutical and agrochemical industries are constantly seeking robust methodologies to construct fluorinated heterocycles, given their profound impact on metabolic stability and bioavailability. Patent CN110272389B introduces a transformative approach for synthesizing 4-polyfluoroalkyl-3,5-dicarbonylpyrazole compounds, a critical scaffold found in numerous bioactive molecules such as Celecoxib. This innovation leverages a copper-catalyzed tandem reaction that merges readily available aryl ethyl ketones or aryl methylene acetones with ethyl diazo polyfluoroacetoacetates. By shifting away from traditional noble metal catalysis, this protocol offers a streamlined, one-step pathway that operates under remarkably mild conditions, typically around 50°C in polar aprotic solvents. For R&D directors and procurement specialists, this represents a significant opportunity to optimize supply chains for high-purity pharmaceutical intermediates, ensuring both cost efficiency and consistent quality in the production of complex fluorinated building blocks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of polyfluoroalkyl-substituted pyrazole cores has been fraught with synthetic challenges that hinder efficient commercial manufacturing. Traditional routes often rely on expensive noble metal catalysts, such as rhodium or palladium complexes, which not only inflate the raw material costs but also introduce stringent requirements for metal residue removal to meet regulatory standards for active pharmaceutical ingredients. Furthermore, many existing methodologies suffer from narrow substrate scope, requiring harsh reaction conditions like extreme temperatures or strong acidic environments that can degrade sensitive functional groups. These limitations frequently result in multi-step sequences with poor overall yields and significant waste generation, creating bottlenecks in the supply chain for reliable agrochemical intermediate suppliers who need to deliver large volumes consistently without compromising on purity or environmental compliance.
The Novel Approach
In stark contrast, the methodology disclosed in CN110272389B utilizes an earth-abundant copper catalyst, specifically cuprous cyanide, to drive the tandem cyclization efficiently. This novel approach eliminates the dependency on precious metals, thereby drastically simplifying the downstream purification process and reducing the overall cost of goods sold. The reaction proceeds smoothly in common solvents like N,N-dimethylformamide (DMF) with organic bases such as DBU, demonstrating exceptional tolerance to a wide array of substituents including electron-withdrawing nitro groups and electron-donating methoxy groups. As illustrated in the general reaction scheme below, this one-pot transformation directly converts simple ketones and diazo compounds into the desired dicarbonyl pyrazoles with high regioselectivity, offering a scalable solution for cost reduction in pharmaceutical intermediate manufacturing.
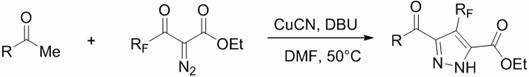
Mechanistic Insights into Copper-Catalyzed Tandem Cyclization
The success of this synthesis lies in the unique ability of the copper catalyst to activate the diazo compound, generating a reactive copper-carbene intermediate in situ. This electrophilic species undergoes a selective insertion or addition reaction with the enolizable ketone substrate, initiating a cascade that ultimately forms the pyrazole ring. The presence of the polyfluoroalkyl group on the diazo reagent plays a crucial role in stabilizing the transition states and directing the regiochemistry of the cyclization, ensuring that the fluorinated moiety is installed precisely at the 4-position of the pyrazole core. This mechanistic pathway avoids the formation of unwanted regioisomers, a common pitfall in pyrazole synthesis, thereby simplifying the isolation of the target molecule and enhancing the overall purity profile required for downstream drug development applications.
Furthermore, the reaction conditions are meticulously optimized to balance reactivity and selectivity, preventing the decomposition of the sensitive diazo reagent while ensuring complete conversion of the starting materials. The use of mild heating at 50°C provides sufficient energy to overcome the activation barrier without promoting side reactions such as dimerization of the carbene or hydrolysis of the ester functionality. This precise control over the reaction environment allows for the accommodation of diverse substrates, as evidenced by the broad scope of aryl ketones shown in the substrate diversity image below, making it a versatile tool for generating libraries of fluorinated heterocycles for medicinal chemistry campaigns.
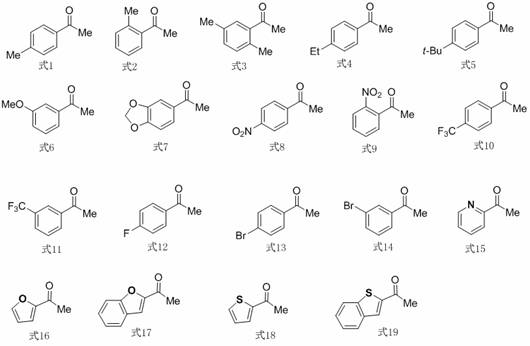
How to Synthesize 4-Polyfluoroalkyl-3,5-Dicarbonylpyrazoles Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires strict adherence to the optimized parameters regarding reagent stoichiometry and atmospheric conditions. The process begins with the preparation of a reaction vessel under an inert nitrogen atmosphere to prevent moisture-induced decomposition of the diazo component. Operators must carefully weigh the aryl ketone substrate and the ethyl diazo polyfluoroacetoacetate, ensuring the molar ratios align with the patent specifications to maximize yield. The addition of the copper catalyst and the organic base must be timed correctly to initiate the catalytic cycle effectively, followed by a controlled heating period that allows the tandem reaction to reach completion before proceeding to the workup phase.
- Mix aryl ethyl ketone or aryl methylene acetone with ethyl diazo polyfluoroacetoacetate, copper catalyst (e.g., CuCN), base (e.g., DBU), and solvent (e.g., DMF) under nitrogen.
- Heat the reaction mixture to 50°C and stir for 12 hours to facilitate the tandem cyclization.
- Dilute with ethyl acetate, wash with saturated ammonium chloride, dry over MgSO4, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this copper-catalyzed protocol offers tangible strategic benefits that extend beyond mere chemical efficiency. The primary advantage lies in the substantial cost optimization achieved by replacing scarce and volatile noble metal catalysts with inexpensive copper salts, which are globally available and subject to far less price fluctuation. This shift not only lowers the direct material costs but also mitigates supply risk, ensuring a more stable and predictable sourcing strategy for critical raw materials needed in the production of high-value pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The elimination of expensive rhodium or palladium catalysts removes a significant cost driver from the bill of materials, while the simplified one-pot nature of the reaction reduces labor and utility consumption associated with multi-step processes. Additionally, the avoidance of complex metal scavenging steps lowers the cost of purification reagents and waste disposal, contributing to a leaner and more economically viable manufacturing model for complex fluorinated compounds.
- Enhanced Supply Chain Reliability: By utilizing commodity chemicals such as aryl ethyl ketones and standard solvents like DMF, the process relies on a robust and diversified global supply base rather than specialized reagents with long lead times. This accessibility ensures that production schedules can be maintained without interruption, reducing the risk of delays caused by raw material shortages and enabling faster response times to market demands for new drug candidates.
- Scalability and Environmental Compliance: The mild reaction conditions, operating at moderate temperatures without the need for high pressure or cryogenic cooling, make this process inherently safer and easier to scale from gram to ton quantities. Furthermore, the reduced use of hazardous heavy metals aligns with increasingly stringent environmental regulations, facilitating smoother regulatory approvals and minimizing the ecological footprint of the manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the process capabilities and limitations for potential partners and licensees.
Q: What are the key advantages of this copper-catalyzed method over traditional noble metal catalysis?
A: This method utilizes inexpensive copper salts like CuCN instead of costly rhodium or palladium catalysts, significantly reducing raw material costs while maintaining high regioselectivity and yield under mild conditions.
Q: What is the substrate scope for this pyrazole synthesis?
A: The process demonstrates excellent functional group tolerance, accommodating various aryl ethyl ketones and aryl methylene acetones with electron-donating or withdrawing groups, as well as heterocyclic substrates.
Q: Is this process suitable for large-scale manufacturing of pharmaceutical intermediates?
A: Yes, the reaction operates at mild temperatures (50°C) in common solvents like DMF, avoiding extreme pressures or cryogenic conditions, which facilitates safe and efficient commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Polyfluoroalkyl-3,5-Dicarbonylpyrazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthetic routes in accelerating drug discovery and development timelines. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and robust. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities to guarantee batch-to-batch consistency.
We invite you to collaborate with us to leverage this advanced copper-catalyzed technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to help you optimize your supply chain and achieve your commercial goals efficiently.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
