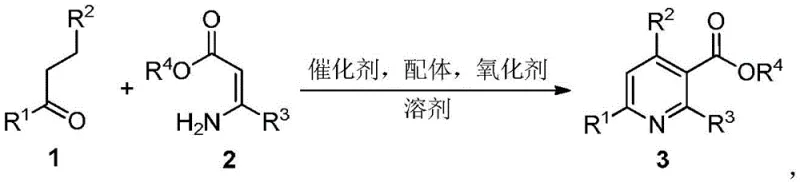
Scalable Copper-Catalyzed Synthesis of Pyridine-3-Carboxylates for Advanced Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries continuously seek robust methodologies for constructing nitrogen-containing heterocycles, particularly pyridine derivatives, which serve as indispensable scaffolds in active pharmaceutical ingredients (APIs) and agrochemicals. Patent CN107739332B discloses a significant advancement in this domain by presenting a highly efficient synthetic method for pyridine-3-carboxylate compounds. This innovation addresses critical bottlenecks in traditional manufacturing by employing a one-pot multi-step tandem reaction strategy that utilizes readily available alpha,beta-saturated ketones or aldehydes coupled with aminoalkenoates. The technical breakthrough lies in the strategic combination of a copper-based catalyst system with a nitroxyl radical oxidant, enabling the direct assembly of the pyridine core under relatively mild thermal conditions. For R&D directors and process chemists, this patent represents a pivotal shift towards more sustainable and atom-economical processes, eliminating the need for pre-functionalized starting materials that often drive up costs and waste generation in complex molecule synthesis.
Furthermore, the versatility of this synthetic route is underscored by its exceptional tolerance to diverse functional groups, allowing for the rapid generation of structural libraries essential for drug discovery programs. The ability to synthesize these valuable intermediates using simple reagents like propiophenone derivatives and ethyl 3-amino-3-phenylacrylate in the presence of air as the terminal oxidant marks a departure from energy-intensive and hazardous legacy protocols. By leveraging this technology, manufacturers can achieve significant improvements in process safety and operational simplicity, directly translating to enhanced supply chain reliability for high-purity pharmaceutical intermediates. The following analysis delves into the mechanistic nuances and commercial implications of adopting this copper-catalyzed oxidative cyclization for industrial applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the pyridine-3-carboxylate skeleton has relied on classical condensation reactions such as the Hantzsch pyridine synthesis or modifications thereof, which frequently suffer from significant drawbacks that hinder their utility in modern green chemistry contexts. Traditional routes often necessitate the use of harsh reaction conditions, including extremely high temperatures or strong acidic/basic environments, which can lead to the decomposition of sensitive functional groups and result in complex impurity profiles that are difficult to separate. Moreover, many conventional strategies exhibit poor atom economy, generating substantial amounts of stoichiometric byproducts that require extensive downstream processing and waste treatment, thereby inflating the overall cost of goods sold (COGS). The reliance on expensive transition metal catalysts, such as palladium or rhodium, in some cross-coupling approaches further exacerbates cost pressures and introduces risks related to heavy metal residue limits in final drug substances. Additionally, the multi-step nature of older syntheses often involves isolating unstable intermediates, increasing the potential for yield loss and operational hazards during scale-up.
The Novel Approach
In stark contrast, the methodology outlined in patent CN107739332B introduces a streamlined one-pot protocol that merges bond formation and oxidative aromatization into a single operational sequence, drastically reducing the number of unit operations required. This novel approach utilizes an inexpensive copper salt, such as copper(II) acetate, in conjunction with a bidentate nitrogen ligand like 2,2'-bipyridine and a stable nitroxyl radical oxidant like TEMPO to facilitate the transformation. The reaction proceeds efficiently in polar aprotic solvents such as N,N-dimethylformamide (DMF) at temperatures ranging from 100°C to 140°C, utilizing molecular oxygen from air as the ultimate oxidant, which is both economically and environmentally superior to stoichiometric chemical oxidants. This strategy not only simplifies the workflow by avoiding intermediate isolation but also ensures high atom efficiency, aligning perfectly with the principles of green chemistry. The broad substrate scope demonstrated in the patent, accommodating various substituted ketones and aldehydes, confirms the robustness of this method for producing diverse pyridine-3-carboxylate derivatives needed for specialized chemical applications.
Mechanistic Insights into Copper-Catalyzed Oxidative Cyclization
The core of this synthetic innovation relies on a sophisticated copper-catalyzed oxidative cyclization mechanism that orchestrates the assembly of the pyridine ring from simple precursors. The reaction initiates with the activation of the alpha,beta-saturated ketone by the copper catalyst, likely forming an enolate or copper-enolate species that undergoes nucleophilic attack on the aminoalkenoate substrate. This initial condensation step generates a dihydropyridine intermediate, which is subsequently subjected to oxidative dehydrogenation to yield the fully aromatic pyridine system. The TEMPO radical plays a crucial role in this oxidation step, acting as a hydrogen atom abstractor that regenerates the active copper(II) species while being reduced to its hydroxylamine form, which is then re-oxidized by molecular oxygen. This catalytic cycle ensures that the copper remains in its active oxidation state throughout the process, minimizing catalyst loading requirements and preventing the accumulation of inactive copper(I) species that could stall the reaction.
Understanding the impurity control mechanisms within this system is vital for ensuring the production of high-purity pharmaceutical intermediates. The mild reaction conditions and the specific selectivity of the copper/TEMPO system help suppress side reactions such as over-oxidation or polymerization of the reactive enamine intermediates. The use of air as the oxidant, rather than aggressive peroxides or hypervalent iodine reagents, further minimizes the formation of oxidative byproducts that could complicate purification. Furthermore, the choice of solvent and ligand creates a coordination environment that stabilizes the transition states leading to the desired pyridine product, effectively outcompeting potential pathways that lead to regioisomers or oligomeric impurities. This inherent selectivity reduces the burden on downstream purification processes, such as column chromatography or recrystallization, thereby improving the overall throughput and yield of the manufacturing process.
How to Synthesize Pyridine-3-Carboxylates Efficiently
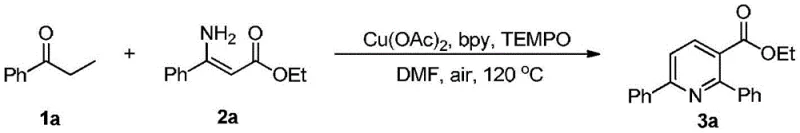
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to the stoichiometry and reaction parameters to maximize yield and reproducibility. The general procedure involves charging a reaction vessel with the alpha,beta-saturated ketone, the aminoalkenoate, the copper catalyst, the ligand, and the oxidant in the chosen solvent, followed by heating under an air atmosphere. For instance, a typical optimization might involve using a molar ratio of ketone to aminoalkenoate of roughly 1.2:1 to ensure complete consumption of the more valuable amine component, with catalyst loadings kept low to maintain cost efficiency. The reaction temperature is typically maintained between 100°C and 140°C, with 120°C often providing an optimal balance between reaction rate and thermal stability of the reagents. Upon completion, the workup is straightforward, involving aqueous quenching and organic extraction, which facilitates the isolation of the crude product for final purification.
- Mix alpha,beta-saturated ketone (e.g., propiophenone), aminoalkenoate (e.g., ethyl 3-amino-3-phenylacrylate), Cu(OAc)2 catalyst, bipyridine ligand, and TEMPO oxidant in DMF solvent.
- Seal the reaction vessel in the presence of air and heat the mixture to 100-140°C (optimally 120°C) with stirring for approximately 20 hours.
- Quench the reaction with water, extract with ethyl acetate, wash the organic phase with brine, dry over sodium sulfate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this copper-catalyzed methodology offers tangible benefits that extend beyond mere technical feasibility, directly impacting the bottom line through cost reduction in pharmaceutical intermediate manufacturing. The elimination of expensive noble metal catalysts in favor of abundant copper salts significantly lowers the raw material costs, while the use of air as the oxidant removes the need for purchasing and handling hazardous stoichiometric oxidizing agents. This shift not only reduces direct material expenses but also simplifies regulatory compliance and safety protocols, as the process avoids the generation of toxic heavy metal waste streams that require specialized disposal. The simplified one-pot nature of the reaction reduces the number of processing steps, leading to shorter cycle times and lower labor and utility costs associated with multiple isolation and purification stages. These factors collectively contribute to a more resilient and cost-effective supply chain for critical pyridine building blocks.
- Cost Reduction in Manufacturing: The substitution of precious metal catalysts with inexpensive copper salts and the utilization of atmospheric oxygen as the terminal oxidant drastically reduce the cost of goods. By avoiding complex multi-step sequences and intermediate isolations, the process minimizes solvent usage and energy consumption, leading to substantial operational savings without compromising product quality.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable starting materials, such as simple ketones and amino esters, ensures a consistent and secure supply of raw materials, mitigating the risk of shortages associated with exotic or custom-synthesized reagents. The robustness of the reaction conditions allows for flexible manufacturing schedules and reduces the likelihood of batch failures due to sensitive reagent degradation.
- Scalability and Environmental Compliance: The high atom economy and minimal waste generation of this one-pot tandem reaction align with stringent environmental regulations, facilitating easier permitting and scale-up to commercial production volumes. The absence of toxic byproducts and the use of greener reagents simplify waste treatment processes, supporting sustainable manufacturing practices and enhancing the corporate sustainability profile.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this pyridine synthesis technology, providing clarity for stakeholders evaluating its adoption. These insights are derived directly from the experimental data and scope limitations detailed in the patent documentation, ensuring accurate expectations for process performance. Understanding these aspects is crucial for integrating this method into existing production workflows and optimizing the supply of high-value heterocyclic intermediates.
Q: What are the primary advantages of this copper-catalyzed method over traditional pyridine synthesis?
A: This method utilizes commercially available and safe reagents under mild conditions (100-140°C) with air as the terminal oxidant, avoiding harsh conditions and expensive noble metal catalysts often required in conventional routes.
Q: What is the substrate scope for this pyridine-3-carboxylate synthesis?
A: The process demonstrates broad applicability, tolerating various substituents on the ketone and aminoalkenoate substrates, including electron-donating groups (methyl, methoxy), electron-withdrawing groups (fluoro, chloro, trifluoromethyl), and heterocycles like thiophene.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the one-pot tandem reaction design simplifies operation by eliminating intermediate purification steps, and the use of inexpensive copper catalysts and air oxidation significantly reduces production costs and environmental impact, making it ideal for scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyridine-3-Carboxylates Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient synthetic routes like the one described in CN107739332B for securing the supply of critical pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent quality regardless of volume. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of pyridine-3-carboxylates delivered adheres to the highest industry standards for drug substance manufacturing. We are committed to translating innovative academic and patent technologies into robust, GMP-compliant commercial processes that drive value for our global partners.
We invite you to collaborate with our technical team to explore how this copper-catalyzed synthesis can be tailored to your specific project needs, offering a pathway to Customized Cost-Saving Analysis for your supply chain. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can accelerate your development timelines while optimizing production costs.
